Abstract
The human lower respiratory tract is considered sterile in normal healthy individuals (Flanagan et al., 2007; Speert, 2006) despite the fact that every day we breathe in multiple microorganisms present in the air and aspirate thousands of organisms from the mouth and nasopharynx. This apparent sterility is maintained by numerous interrelated components of the lung physical structures such as the mucociliary elevator and components of the innate and adaptive immune systems (discussed below) (reviewed in (Diamond et al., 2000; Gerritsen, 2000)). However, it is possible that the observed sterility might be a result of the laboratory practices applied to study the flora of the lungs. Historically, researchers faced with a set of diseases characterized by a changing and largely cryptic lung microbiome have lacked tools to study lung ecology as a whole and have concentrated on familiar, cultivatable candidate pathogens.
You have full access to this open access chapter, Download chapter PDF
Similar content being viewed by others
Keywords
- Human Immunodeficiency Virus
- Chronic Obstructive Pulmonary Disease
- Cystic Fibrosis
- Respiratory Syncytial Virus
- Cystic Fibrosis Patient
These keywords were added by machine and not by the authors. This process is experimental and the keywords may be updated as the learning algorithm improves.
The Human Lung
Lung Microbiome Overview
The human LOWER respiratory tract is considered sterile in normal healthy individuals (Flanagan et al., 2007; Speert, 2006) despite the fact that every day we breathe in multiple microorganisms present in the air and aspirate thousands of organisms from the mouth and nasopharynx. This apparent sterility is maintained by numerous interrelated components of the lung physical structures such as the mucociliary elevator and components of the innate and adaptive immune systems (discussed below) (reviewed in (Diamond et al., 2000; Gerritsen, 2000)). However, it is possible that the observed sterility might be a result of the laboratory practices applied to study the flora of the lungs. Historically, researchers faced with a set of diseases characterized by a changing and largely cryptic lung microbiome have lacked tools to study lung ecology as a whole and have concentrated on familiar, cultivatable candidate pathogens. With the availability of new technologies for cultivation-independent analysis of microbial populations, it is now possible to follow individuals by sampling their lung microbiome sequentially during episodes of disease and recovery in order to identify associations between the lung microbiome and health and disease.
Any respired contaminating particles or pathogens that evade the lung’s physical and immune barriers are usually eliminated by dendritic cells and alveolar macrophages that deliver them into local draining lymph nodes. Macrophages kill invading microorganisms while en route to the draining lymph nodes, and in some cases at the nodes themselves (Bozza et al., 2002; Kirby et al., 2009). Thus, it is not unusual to isolate viable bacteria or fungi from “normal” lung tissue (Lass-Florl et al., 1999), and the term “sterile” should be applied with caution. It is perhaps more accurate to say that there is no resident flora that permanently colonizes normal lungs.
Even normal healthy lungs are not microbe free all the time. Lower airway infections by bacteria, viruses, or fungi are among the most prevalent causes of transmissible disease in humans, with two to three million community-acquired (non-hospital-acquired) cases per year in the United States (Segreti et al., 2005). In 2006, the number of deaths attributed to pneumonia (bacterial and viral) and influenza in the United States was 60,000 (Gao et al., 2008) http://www.cdc.gov/nchs/fastats/deaths.htm). In 2009, nearly 9.3 million new cases of tuberculosis were reported around the world (http://www.who.int/mediacentre/factsheets/fs104/en/index.html). With proper treatment, the lungs of individuals with these infectious diseases will revert to their normal “sterile” state.
Little is known about the composition of the microbial population of the upper and lower airways in health or disease. It is likely, given the multiple microorganisms already implicated in chronic lung diseases such as chronic obstructive pulmonary disease (COPD), that there are other undetected organisms and that there are complex relationships between multiple pathogens involved that are not currently understood. A few studies have examined microbial species in limited numbers of normal subjects and patients with various respiratory disorders. One study using 16S rDNA clone libraries and microarrays did not detect any bacteria in the lungs of patients without respiratory disease who were briefly intubated for surgery (Flanagan et al., 2007). The same study reported that all patients intubated for longer periods had detectable 16S rDNA and that the bacterial diversity present decreased during antibiotic usage. Another study used 16S rDNA amplification to identify bacterial species in 16 patients with ventilator-associated pneumonia (Bahrani-Mougeot et al., 2007). This study identified bacterial pathogens not seen using conventional culture techniques, especially anaerobes, and found that oral bacteria could be detected in the lung. In one study, sputum samples from 25 cystic fibrosis (CF) patients were analyzed using 16S gene profiling and the authors identified an average of 7.2 species present per subject (Bittar et al., 2008). Viruses have also been examined in nasal lavages in both asthmatic and normal subjects with cold symptoms using the Virochip microarray (Kistler et al., 2007). The microarray technology identified more viruses than conventional culture methods and had excellent sensitivity and specificity compared with pathogen-specific polymerase chain reaction (PCR). An unexpected diversity of human coronavirus and rhinovirus strains was discovered in the subjects.
So if the lung is generally sterile, why do some individuals become chronically colonized? What organisms colonize the lungs? Those with physically compromised airways or immune system deficiencies are subject to chronic microbial colonization of their airways and to high-frequency episodes of viral, bacterial, or fungal lower respiratory infections. Perhaps in no other body site is the direct relationship between disease and microbiome more explicit than in the lungs where there is a distinct and obvious microbial difference between normal and diseased individuals.
Anatomical and Immunological Setting
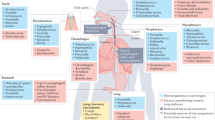
The lower respiratory tract, composed of the trachea and lungs, is quite different in structure and function from the upper respiratory tract, which is highly colonized by microorganisms. The lungs themselves are divided into different sections according to their function and structures: the bronchi, bronchioles, and alveoli. Bronchi and bronchioles are primarily conductive airways surrounded by thick cartilage that allow easy airflow into the parenchyma (or alveolar tissue) of the lungs, where gas exchange occurs. Conductive airways are covered in ciliated epithelium interspersed with different types of secretory cells that release mucins, immunomodulatory proteins, surfactants, and proteases. Together, the physical and chemical barriers protect against physical and biological damage by establishing a mucociliary elevator, which brings about an upward transport of a mucus stream for the lungs (Fraser, 2005). The secretory cells decrease in proportion from 20 to 30% in the trachea to less than 1% in the distal and alveolar parts of the lungs. In addition to the physicochemical protection provided by cilia and mucus, the epithelium is also protected by several immune cells, including dendritic, Langerhans, T lymphocytes, and mast cells, that respond to inhaled antigens establishing a robust immunity (Fraser, 2005). It is thought that in conjunction with the physical barriers provided by the nose and upper respiratory mucosa, these defenses are enough to maintain sterility in the lower respiratory tract.
The vast majority of the lung surface epithelium, however, is alveolar. It is estimated that 87% of the total volume of the lungs is alveolar, with only 6% of this being tissue and the remainder gas (Stone et al., 1992). The primary role of this tissue is to carry out gas exchange. The epithelium is mostly a continuous single layer of cells overlying a thin interstitium, which contains numerous capillaries that supply ample blood for gas exchange (Fraser, 2005; Stone et al., 1992). Unlike the epithelium in the conducting airways, the respiratory epithelium is not ciliated or protected by mucus. Instead, it is covered by surfactant proteins that maintain the surface tension for efficient gas exchange. The lack of mucus or secretory cells is compensated by the presence of alveolar macrophages, mast cells, lymphocytes, dendritic cells, and other monocyte-like cells that protect the epithelium from potential pathogens and help maintain sterility.
Diseases of the Lower Respiratory Tract and Their Impact on the Lung Microbiome
Recent World Health Organisation (WHO) figures rank lower respiratory diseases second in an assessment of the burden of disease worldwide (http://www.who.int/respiratory/en/). In 2006, 124, 500 people died in the US due to chronic lower respiratory disease (http://www.cdc.gov/nchs/fastats/deaths.htm). Chronic respiratory diseases include: asthma, COPD, CF, and bronchiectasis. These diseases generally lead to impaired clearance and function of the mucociliary elevator and/or the immune protection of the lung. In addition, immune deficiency such as that caused by the human immunodeficiency virus (HIV) also disrupts the typical immune homeostasis in the lungs. Without the normal protective barriers, the lungs fall victim to persistent and severe colonization that can ultimately lead to death, particularly for CF patients. As discussed below, the lung microbiome in each of these diseases is very different from normal individuals. The data discussed in the following sections demonstrate a clear link between microbial colonization and severity of disease symptoms. It is unclear, however, what exact role these different microbial populations play in initiating and enhancing the progress of such chronic respiratory diseases. Lastly, in some cases, the data hint that some population structures might actually be protective against further decline, but much more research needs to be conducted in this area to make a definitive claim.
This chapter discusses the methods for sampling and characterizing the microbiome of the lungs. In addition, it reviews the current status of our understanding of the lung microbiome in asthma, idiopathic bronchiectasis, CF, COPD, and during immune deficiency due to HIV infection.
Microbiome Characterization
Human Lung Sampling Methods
Several procedures have been developed for sampling the microbial population of the human lung econiche. In order of increasing invasiveness they are sputum induction, bronchoalveolar lavage (BAL), bronchial brushing, endobronchial biopsy, and transbronchial biopsy. Sputum induction by inhalation of hypertonic saline is a non-invasive method to obtain samples from the lower respiratory tract for cell and microbial analysis (Bickerman et al., 1958). The quality of samples varies and can be scored on the volume of the obtained sputum plugs and the level of salivary contamination as measured by squamous cells observed by microscopy.
BAL is a procedure in which a bronchoscope is passed through the mouth or nose into the lungs and saline is instilled into a segment of the lung and then recollected for examination (Henderson, 1994; Reynolds and Chretien, 1984). BAL is most commonly used to diagnose infections in both immunocompetent and immunosuppressed patients. BAL is the most common procedure for sampling the lower respiratory system microbial colonization/infection status, to sample the components of the epithelial lining fluid, and to determine the protein composition of the airways. It is often used in evaluating the patient’s lung immunological status by sampling cells and pathogen levels. BAL is an invasive procedure and thus is less ideal for research purposes.
Bronchial brushing provides access to cells and microbes that are adherent to the luminal surfaces of the lower airways. In this procedure, a flexible fiber optic bronchoscope is used for brushing a targeted lesion or site (Fennessy, 1967; Zavala et al., 1973), where induced sputum and BAL procedures will allow sampling of cells and microbes that can be washed from the lumen surface, brushing will recover adherent cells (e.g., bronchial epithelial cells) and microbes. Recently, brushing techniques have been developed to sample distal lung (i.e., small airway) epithelial cells and associated microbes (Ammous et al., 2008). This technique will enable investigators to further study the microbiome in lung diseases such as COPD.
Endobronchial biopsy involves using the fiber optic bronchoscope to identify appropriate target sites in the lung and obtain large airway tissue samples using inserted alligator forceps, cup forceps, or curette passed through the endoscope’s central channel. This procedure poses a higher risk than BAL but allows sampling the invasive microbes within the airway tissue (Scott et al., 1991; Trulock et al., 1992).
Transbronchial biopsy, the most invasive of these sampling procedures, is routinely performed for clinical care and allows clinicians and researchers to obtain distal (small) airway tissues as well as alveolar tissues. This procedure has been safely done by several research groups (Balzar et al., 2005) and will likely further our understanding of the microbiome in human distal lung tissues, but carries a significant risk of complications.
High-Throughput Sequencing of Bacterial Ribosomal RNA Subunits
Standard microbiological and virologic methods detect only a small proportion of the bacteria and viruses present in various body sites because the great majority of these organisms are uncharacterized or uncultivable. To understand the real diversity, culture-independent methods, such as sequencing, are thus a necessity. Sequenced-based identification of microbial species is facilitated by decreased costs of sequencing, and the availability of next-generation sequencing technologies, further enhances the capacity to generate large amounts of data.
For the identification of bacterial species within an environment, the amplification of 16S rRNA genes (or 16S rDNA) using universal primers are useful for diversity characterization because this genetic locus is present in all bacterial species (Relman et al., 1991). The nine hypervariable regions of the 16S rDNA can be used for bacterial species identification (Chakravorty et al., 2007; Rokas et al., 2007) with some regions having better discriminatory value than others. The sequencing and phylogenetic analysis of bacterial 16S rRNA derived from microbiome samples has been the primary method used to investigate bacterial diversity in the human body (Bik et al., 2006; Dekio et al., 2005; Eckburg et al., 2005; Gao et al., 2007; Hugenholtz et al., 1998; Hyman et al., 2005; Zhang et al., 2006). These studies have revealed a far higher level of diversity than conventional culture techniques (Aly et al., 1976; Bik et al., 2006; Dekio et al., 2005; Kazor et al., 2003; Korting et al., 1988; Kroes et al., 1999; Paster et al., 2001). These studies revealed that the majority of bacterial sequences correspond to uncultivated species and novel organisms. There was significant intersubject variability and variability between stool and mucosal microbial populations. For example, recent studies by Blaser and colleagues at New York University have demonstrated substantial changes in the ratio of the genus Streptococcus to Propionibacterium in skin samples from healthy persons and in normal skin of patients with psoriasis \({\textrm{(ratio}} = 0.4{\textrm{;}}\;n = 2,649\;{\textrm{clones)}}\), and from psoriatic lesion samples \({\textrm{(ratio}} = 5.0{\textrm{;}}\;n = 1,314\;{\textrm{clones; }}P = 0.01{\textrm{)}}\) (Gao et al., 2008).
In a lung study, bacterial diversity was analyzed in the endotrachael aspirates from seven intubated patients colonized with Pseudomonas aeruginosa using both sequencing from 16S rRNA clone libraries and an oligonucleotide microarray termed as the PhyloChip (Flanagan et al., 2007). Controls were subjects briefly intubated for elective surgery. Bacteria were not detected by either method in samples from the controls. Sequencing from the clone libraries detected the presence of many orally, nasally, and gastrointestinal associated bacteria including known pathogens. The Phylochip detected the same organisms and many additional bacterial groups present at low abundance. Following antibiotic therapy, the bacterial populations’ diversity decreased and was dominated by a single respiratory pathogen. In six of the seven patients, the dominant species was P. aeruginosa in spite of targeting this organism with antibiotics to which it was reportedly sensitive. The authors hypothesize that the loss of population diversity may directly contribute to pathogenicity, persistence, and development of pneumonia.
Ribosomal RNA ITS Typing of Fungal Populations
Similarly, amplification of regions from the 18S and internal transcribed spacer (ITS) regions of the rRNA, a conserved fungal gene, allows discrimination of fungal species (Fujita et al., 2001; Makimura, 2001). A preliminary study was undertaken to examine the efficacy of a community sequencing method to identify the fungal species in BAL lung samples from 23 human subjects. The fungal ITS1-5.8S-ITS2 region was amplified and the results showed that 4 of 23 patients (17%) had fungal DNA levels that could be reproducibly detected by PCR. The detected fungi included Aspergillus fumigatus, Candida tropicalis, and Penicillium digitatum, among others (Denning, unpublished). These data agree with a culture-based study that showed that 63% of their sample population had evidence of pulmonary fungal colonization (Lass-Florl et al., 1999), most commonly with A. fumigatus and other Candida species, and also Zygomycetes. The results also demonstrate that rRNA sequencing is a viable platform for characterization of fungal communities in the human body.
Viral Identification via Genome Sequencing for Population Analysis
Although there are no conserved genes that can be targeted for determination of viral diversity, whole genome shotgun of a sample enriched for viruses (such as by filtering) can lead to an effective characterization of viral communities (Angly et al., 2006). Hundreds of viral genome sequences can be completed in a single sequencing reaction run using the GS-FLX (454/Roche) sequencing platform. Using this technology and a random priming-based method, referred to as sequence independent single primer amplification (SISPA), near-full-length genomes of RNA or DNA viruses can be sequenced. SISPA can be used to sequence known and unknown viral genomes (Djikeng et al., 2008). This viral sequencing methodology can potentially be adapted for the determination of viruses within BAL by enrichment using nuclease treatment and filtration followed by taking the extracted total RNA and DNA through the SISPA process followed by sequence comparisons to known viruses.
Metagenomics
The initial studies of small 16S rRNA described above hinted at great diversity within the human microbiome, yet it left important questions unanswered such as the identity of the nondominant community members and their biological roles. The applications of shotgun techniques to the study of the human microbiome (Kurokawa et al., 2007; Manichanh et al., 2006; Zhang et al., 2006) again highlight the extent of microbial diversity associated with the human body while revealing much more of the identity and biology of nonculturable microorganisms. As a result of reduced costs and improved sequencing technologies, it is possible to perform in-depth metagenomic surveys of the human body’s microbial diversity beyond the 16S rRNA surveys. Metagenomics, a term introduced in 1998, describes the functional and sequence-based analysis of total microbial genomes from environmental samples (Handelsman et al., 1998). Metagenomics uses techniques that resemble the “Whole Genome Shotgun” approach of single genome sequencing, but it is not limited to a single species.
Human metagenomics has provided insight into the complex composition of the microbiome of these several body sites, and this information has allowed us to draw tentative conclusions about the relationship between specific microbiomes and health. The human microbiome is composed of multiple “ecological niches”, including the mouth (Kroes et al., 1999; Paster et al., 2001), esophagus (Zhang et al., 2006), stomach (Bik et al., 2006), intestine (Gill et al., 2006), skin (Gao et al., 2007), and vagina (Zhou et al., 2004). Our understanding of the overlap and the degree of communication between them is rudimentary at best. Perhaps the most extensively studied has been the human gut microbiome where the interaction of the gut microflora, independently or through interaction with the genetic makeup of the host plays a role in obesity, Crohn’s disease, and ulcerative colitis (Frank et al., 2007; Gophna et al., 2006; Turnbaugh et al., 2006).
The Lung Microbiome in Asthma
Asthma is a complex disease characterized by chronic inflammation in the lungs and reversible narrowing of the airways. Symptoms include dyspnea, coughing, wheezing, airway hyper–reactivity, chronic eosinophilic atopy, and mucus hyper secretion (Busse and Lemanske, 2001). About 20 million people in the US have been diagnosed with asthma; 9 million of them are children. Asthma causes 4,000 deaths per year in the US and 11 million exacerbations. Asthma is caused by environmental and genetic factors (Martinez, 2007), with asthma attacks resulting from immune responses to inhaled allergens. The majority of asthma exacerbations are caused by viral infections (Krishnan et al., 2006). Atypical bacterial infections have also been associated with asthma exacerbations and with chronic asthma (Johnston and Martin, 2005; Martin et al., 2001). In susceptible individuals, the development of asthma has been associated with bacterial colonization in neonates (Bisgaard et al., 2007) and viral and bacterial infections (Wu and Chu, 2009). There is abundant evidence testifying to the importance of microbes to the development and maintenance of asthma. A recent publication using a bacterial gene sequencing method suggests a disordered microbiome in asthmatic airways (Hilty et al., 2010).
In the developed world there has been an increased focus on predisposing factors for asthma due to its rapidly increasing prevalence, now affecting up to a quarter of urban children (Lilly, 2005). Asthma is known to be caused by environmental and genetic factors (Martinez, 2007). These factors determine asthma severity and how easily it can be treated (Martinez, 2007). Many associations with asthma have been detected including exposure to cigarette smoke (Thomson et al., 2004), caesarean section birth relative to natural birth (Thavagnanam et al., 2008), early viral respiratory infections (Gold and Wright, 2005; Harju et al., 2006), early in life antibiotic use (Marra et al., 2006), and living in the US (Gold and Wright, 2005). One theory for the cause of the increase in asthma incidence is the hygiene hypothesis (Strachan, 1989), that the rise in prevalence of asthma is a direct consequence of the success of modern hygienic practices in preventing childhood infections. This hypothesis is supported by numerous studies that have shown that children coming from a less hygienic environment have less asthma and other allergenic diseases (Ball et al., 2000; Celedon et al., 1999; Jarvis et al., 1997). In addition, alterations in innate immune system genes have been shown to be associated with the inception and development of asthma. These genes include the toll-like receptors and other genes such as MBL, MYLK, DEFB1, JUN, INF-α5, and NOS2A reviewed in Wu and Chu (2009).
The Role of Infections in Asthma Exacerbations
Asthma exacerbations have long been associated with viral infections (Pattemore et al., 1992). More recently, the use of reverse transcriptase PCR has greatly facilitated the identification of the exacerbation-associated virus. Studies using this tool have suggested that 75–80% of asthma exacerbations are caused by virus infections (Wark et al., 2002).
Rhinovirus (RV) infections during early childhood are associated with the development of asthma, lower respiratory tract infections, and wheezing (Jackson et al., 2008; Lemanske et al., 2005). They are also associated with hospitalization for asthma in adults (Venarske et al., 2006). A separate study revealed that patients with allergic asthma infected with RV had increased admissions to hospitals and that dust mite allergen was the primary allergen when these patients were skin tested with a panel of aeroallergens (Green et al., 2002). Respiratory syncytial virus (RSV) in infants causes lower respiratory infection leading to pneumonia and bronchiolitis. RSV bronchiolitis is the leading cause of wheezing in infants and young children, and children infected with RSV resulting in bronchiolitis are more likely to develop wheezing and asthma later in childhood (Peebles, 2004). Similarly, the human metapneumovirus (hMPV) was first isolated from children in 2001 and has been found to be associated with asthma exacerbations in both children under 5 years of age and adults (Foulongne et al., 2006; Williams et al., 2004).
Mycoplasma pneumoniae and Chlamydia pneumoniae are bacteria that attach to airway epithelial cells and cause cell damage. Infections by these bacteria have been shown to be associated with asthma exacerbations (Johnston and Martin, 2005; Lieberman et al., 2003; Martin et al., 2001). Using a PCR assay, 31 of 55 patients with asthma were positive for either of these bacteria in lung tissue or BAL, suggesting that some level of colonization by these bacteria may be common in asthma patients (Martin et al., 2001). Studies in a mouse model suggest that preexisting allergic inflammation impairs the ability to upregulate TLR-2 and IL-6 in the lungs, leading to decreased clearance of M. pneumoniae and an increase in airway inflammation (Kraft et al., 2008).
The Role of Infections and Microbial Colonization in Asthma Development
Evidence is accumulating that infections are associated with the induction and development of asthma. First, long-term cohort studies on the development of asthma show that most childhood asthma begins in infancy. The first episode of wheezing begins before the age of 3 and is frequently associated with lower respiratory tract viral infections, usually RSV, but also RV (Gern et al., 2000; Sigurs et al., 2005). These infectious episodes and associated wheezing are strong predictors for the development of childhood asthma and atopy (Devulapalli et al., 2008; Kusel et al., 2007; Martinez et al., 1995; Singh et al., 2007). Second, many studies have associated viral infections with asthma prevalence in children (Devulapalli et al., 2008; Jackson et al., 2008; Kusel et al., 2007; Papadopoulos and Kalobatsou, 2007; Sigurs et al., 2005; Singh et al., 2007; Williams et al., 2004). Lastly, Wu et al. have provided evidence to suggest that viral infections have a causal role in asthma initiation and development where they show that viral infection during the first 4 months of age is strongly correlated with the development of asthma by age 5 (Wu et al., 2008). Only one-third of children with childhood wheezing and asthma, however, will develop persistent asthma symptoms in adulthood (Gerritsen, 2002; Taylor et al., 2005; Vonk et al., 2004). Management of the symptoms with corticosteroid therapy is effective but may not alter the asthma progression (Guilbert et al., 2006).
The role of infections in asthma induction and development will likely be shown to be mediated through the effect of these infections on the chronic inflammatory response in the airways of asthmatics. Microbial infections can generate either a Th2- or a Th1-biased response that could exacerbate or attenuate asthma, respectively. In asthmatics, a pro-inflammatory Th2 response persists even in the absence of allergens involving CD4+ Th2 cells, eosinophils, mast cells, and the Th2 cytokines IL-4, IL-5, IL-9, and IL-13 (Holgate, 2008).
Bacterial infections have been shown to contribute to asthma development. In a longitudinal prospective birth cohort study of 411 infants born to mothers with current or previous asthma, neonates colonized in the hypopharyngeal regions with Streptococcus pneumoniae, Haemophilus influenzae, or Moraxella catarrhalis or a combination of these organisms were found to be at increased risk for recurrent wheezing in early childhood and asthma at age 5 (Bisgaard et al., 2007). A protective role for some bacteria has been reported (Blaser et al., 2008). Several studies have found a protective effect of mycobacterial exposure on atopy and airway inflammation (Camporota et al., 2003; Shirakawa et al., 1997; Yang et al., 2002). These exposures include Bacillus Calmette-Guerin vaccination or heat-killed Mycobacterium vaccae. Early exposure to bacterial endotoxins may reduce future allergies or asthma (von Mutius et al., 2000), although endotoxins associated with house dust are associated with more asthma symptoms and worse lung function (Dales et al., 2006; Michel et al., 1996; Park et al., 2001).
Thus, the role of bacteria in asthma initiation and development appears to be complex. The causative interaction is likely to prove to be the interaction of bacteria and bacterial components in modulating the Th1 and Th2 innate immune system responses. The characterization of these interactions will be complicated by timing, dose, anatomical site, and duration of the bacterial exposure as well as the host genetic and environmental factors influencing the immune inflammatory response (Holt, 1996).
The Role of Fungi in Severe Asthma
It has recently been demonstrated that patients with severe asthma who are also atopic or sensitized to environmental fungi may benefit from treatment with the antifungal azole itraconazole (Denning et al., 2009). This observation has raised questions about the relationships among asthma severity, fungal sensitization, and fungal exposure. The issue is complicated by more than 1.5 million species of fungi that are thought to exist (Hawksworth and Rossman, 1997) and more than 80 species of fungi that have been associated with symptoms of airway allergy (Horner et al., 1995). For one species, A. fumigatus, 20 allergens are thought to participate in human airway allergies (http://www.allergome.org). Determining the clinical relevance of fungal allergens is confounded further by extensive cross-reactivity among fungal allergens (Crameri et al., 2009).
Fungal allergens can induce a number of different human bronchopulmonary disorders, each with a distinct immune pathogenesis. In allergic bronchopulmonary aspergillosis (ABPA), the respiratory system is chronically colonized typically with A. fumigatus. Evidence now suggests that severe asthmatics without ABPA are more likely to be atopic to fungi than patients with milder disease. The diagnostic label “severe asthma with fungal sensitization (SAFS)” has recently been applied to this group (Denning et al., 2006). In these patients, the fungal sensitization is most commonly A. fumigatus, Candida albicans, and Penicillium notatum (O’Driscoll et al., 2009).
The association between severe asthma and fungi has been identified in numerous studies. Atopy to environmental fungi has been associated with severe asthma (O’Driscoll et al., 2005). Many population studies have shown an association between local fungal spore counts and medical emergencies due to asthma exacerbations (Atkinson et al., 2006). Furthermore, studies have shown that fungus exposure in fungal-sensitized individuals induces asthma symptoms (Malling, 1986; Matheson et al., 2005; Pulimood et al., 2007; Salo et al., 2006; Woodcock et al., 2006). Treatment of patients with SAFS with antifungal drugs has generally led to improvement of asthma symptoms (Denning et al., 2006) concurrent with improvements in several markers of atopy such as reduced IgE values, reduced eosinophils counts, and reductions in the level of dose of oral and systemic steroids required (Pasqualotto et al., 2009).
These findings lead to the considerations of the fungal composition of the lung microbiome in asthmatic individuals and indeed in normal individuals. Environmental fungi colonize the lungs of otherwise healthy people (Lass-Florl et al., 1999; Okudaira et al., 1977). These studies were dependent on cultivation-based methods for the detection and identification of these fungi. As a cultivation-independent method, gas chromatography/mass spectroscopy on exhaled breath has revealed the presence of fungus specific biomarkers in patients with CF with and without fungal colonization by A. fumigatus (Syhre et al., 2008). This approach was limited to analyzing for known A. fumigatus markers. The application of sequencing-based approaches for studying the lung microbiome will be essential for revealing the role of fungi in the lung microbiome and the role of the lung microbiome on asthma.
The Lung Microbiome in Cystic Fibrosis (CF)
Introduction
CF is the most common inherited lung disease in the world. It is a severe autosomal recessive disease with an incidence of 1:2000 at birth in populations of northwestern European origin, with a mutant gene carrier frequency of 1:23 in these populations. The genetic defect occurs in the cystic fibrosis transmembrane regulator (CFTR) protein, which acts to transport chloride across cell membranes. Patients with CF are the archetype population with chronic bronchial colonization. Symptoms include permanent bacterial colonization of the lower airways, with a formation of a biofilm, fat maldigestion, male infertility, and elevated levels of chloride in the sweat (Knowles and Durie, 2002). The thick pulmonary system mucus in CF patients minimizes the effectiveness of the mucociliary elevator in clearing the lung of mucus-trapped microorganisms and other debris. As a consequence, microbes chronically colonize these patients’ lungs and they suffer bouts of infection, requiring frequent hospital admission. Cultures reveal a wide range of bacteria, including P. aeruginosa, Mycobacteria, A. fumigatus, and sometimes viruses.
The Chronically Colonized CF Airways Represent a Surprising Complex and Diverse Ecosystem
The precise contributions of different microbes to patient morbidity, and the importance of inter-specific interactions remain largely unclear [reviewed in (Harrison, 2007)]. The complexity of this ecosystem is difficult to overstate. As an example of this complexity, the lungs of CF patients contain large numbers of neutrophils that migrate to this location in response to microbial colonization. These neutrophils secrete granule antimicrobial proteins called defensins that kill microbes. By analysis of CF sputum samples, the levels of extracellular defensins are sufficiently abundant that they may damage the airway epithelium (Soong et al., 1997). As another example of this inter-specific complexity, P. aeruginosa in CF lungs produces copious amounts of a tricyclic compound pyocyanin that kills competing microbes and eukaryotic cells. This compound was shown to specifically inactivate a human lung epithelial cell line vacuolar ATPase (Ran et al., 2003).
The Microbiome of the CF Lung Contains Microbial Diversity Not Evaluable by Standard Culture Techniques
A study of the microbiome of the lungs was conducted to explore the hypothesis that organisms not routinely identified by culture occur in the lungs of CF patient airways and may contribute to disease. To test this hypothesis, 16S rRNA sequence analysis was performed on BAL samples from 42 subjects, 28 CF patients, and 14 other disease controls (Harris et al., 2007). The findings of this analysis were that, for CF subjects, a single rRNA type was dominantly represented in the clone libraries prepared from lung microbiome genomic DNA. This was not found in the controls. Thirteen of the CF subjects’ samples contained bacteria not routinely assessed by culture. Candidate pathogens were identified in four CF subjects. Candidate pathogens were also identified in the controls. This study documented the power of culture-independent molecular techniques to provide a broader view of the airway bacteria than standard clinical culture methods.
The CF viral metagenome was explored in a recent study using five CF individuals and five individuals without disease (Willner et al., 2009). In both cohorts, the overall viral diversity was low. The CF bacteriophage communities were highly similar to each other, whereas the non-CF individual had more distinct phage communities. CF eukaryotic viral communities were dominated by a few viruses, including human herpes viruses and retroviruses.
Fastidious or Noncultivatable Bacteria: An Emerging Concept in CF
The significance of fastidious or noncultivatable organisms in the airway of CF patients is beginning to be explored. Application of specific culture conditions to favor the growth of anaerobes coupled with molecular identification techniques have focused attention in CF on bacteria not routinely detected by standard culture and biochemical identification techniques (Harris et al., 2007; Tunney et al., 2008; Worlitzsch et al., 2009). Direct, culture-independent detection techniques have identified much larger numbers of bacterial species in CF airways and have demonstrated the ability to identify likely pathogenic bacteria occurring during exacerbations when routine cultures are negative (Harris et al., 2007). These molecular identification and detection methods have identified bacteria with different antibiotic susceptibilities relative to conventional pathogens and will undoubtedly lead to novel antimicrobial intervention trials in CF (Worlitzsch et al., 2009). Similar methodology to detect anaerobes or noncultivatable bacteria has not been applied systematically to patients with idiopathic bronchiectasis.
The Lung Microbiome in Idiopathic Bronchiectasis
Introduction
Bronchiectasis is characterized by chronic dilation and inflammation of the conducting airways associated with recurring infections (Barker, 2002). It is the pathologic manifestation of several genetic disorders, including CF and primary ciliary dyskinesia (PCD). However, many patients have no identifiable causes. Idiopathic bronchiectasis is estimated to affect approximately 110,000 US adults (Weycker et al., 2005). Symptoms include cough and chronic sputum production, recurring airway infection, dyspnea, wheezing, and chest pain (Barker, 2002). Microbial infections are central to the pathogenesis and progression of disease.
Bronchiectasis Phenotype and Microbial Flora
Much of the research characterizing the composition and significance of the lower airway microbial flora has been done in CF and relatively little is known about the microbial contribution to disease pathogenesis in idiopathic bronchiectasis. However, recent observations suggest a link between the lower airway microbial flora and host disease characteristics. For example, the prevalence of idiopathic bronchiectasis associated with nontuberculous mycobacteria (NTM) appears to be increasing (Billinger et al., 2009; Marras et al., 2007). Both familial clustering and a characteristic “tall asthenic” phenotype (scoliosis, pectus excavatum, mitral valve abnormalities) in postmenopausal women with bronchiectasis associated with NTM infection (Colombo et al., 2009; Kim et al., 2008) have been reported. It is unknown whether the age, female sex, and unique body morphotype associations are seen in idiopathic bronchiectasis unassociated with NTM. Correlating disease phenotype with microbial flora is dependent upon accurately categorizing the microbial status of the patients. For environmental organisms like NTM, it is important that this categorization include both accurate speciation and determination that the organism likely represents true infection rather than contamination or transient colonization. The American Thoracic Society and the Infectious Diseases Society of America (ATS/IDSA) microbiologic diagnostic criteria for pulmonary disease based on sputum specimens call for at least two positive sputum specimens for the same species (Kim et al., 2008).
Significance of Filamentous Fungi: A Novel Consideration in Bronchiectasis
Concomitant recovery of filamentous fungi from airway specimens is also common in bronchiectasis, but the pathophysiologic consequences are not known. A recent study in CF patients found that A. fumigatus, like NTM, was commonly present in older patients: 75% of patients aged 16–20 years and in 60% of patients over age 20 (Valenza et al., 2008). Amin and colleagues further noted that CF patients who were chronically infected with A. fumigatus (defined as two positive cultures in a given year) had significantly worse airway obstruction as evidenced by a lower forced expiratory volume in one second (FEV1) and significantly higher risk of pulmonary exacerbations during subsequent follow-up than patients without A. fumigatus (Amin et al., 2009). This potential negative impact on the course of bronchiectasis and a possible benefit from antifungal treatment for chronic infection in CF have prompted initiation of a multicenter clinical trial of itraconazole in CF patients in Canada (Amin et al., 2009; Shoseyov et al., 2006). In non-CF bronchiectasis, a recent study suggested that Aspergillus is more common in patients infected with NTM than in those without NTM, and that it is commonly associated with fungal lung disease manifestations in NTM-infected patients (Kunst et al., 2006). However, outside the relatively small numbers of idiopathic bronchiectasis patients with allergic bronchopulmonary aspergillosis (ABPA) or chronic necrotizing aspergillosis, the pathologic significance of these fungi has not been systematically explored in large numbers of patients and very few data are available for Aspergillus species other than fumigatus or filamentous fungi other than Aspergillus (Kobashi et al., 2006; Kunst et al., 2006; Raju et al., 2008).
Serologic Assessment of Fungal Response
ABPA is well described in association with bronchiectasis occurring in asthmatics and patients with CF (Malde and Greenberger, 2004). The diagnostic criteria rely on an elevated total IgE as well as elevated A. fumigatus-specific IgE and IgG in the setting of episodic bronchial obstruction, pulmonary infiltrates, and central bronchiectasis. Kunst and colleagues assessed the prevalence of positive serologic markers for A. fumigatus [IgE by radioallergosorbent test (RAST) and precipitins] among idiopathic bronchiectasis patients and found these markers to be commonly present especially in the setting of concomitant NTM disease (Kunst et al., 2006). Patients with these serologic markers more commonly had radiographic manifestations suggesting Aspergillus-associated disease. While other filamentous fungi such as Scedosporium species have been commonly recovered from the airways of both CF and non-CF bronchiectasis patients, the role these fungi play in disease pathogenesis remains controversial (Cooley et al., 2007). While specific IgE antibody RAST and precipitin assays can be prepared using allergen prepared from the isolated species and correlated with the clinical presentation of allergic bronchopulmonary mycoses, these assays have not been commonly used to characterize the clinical significance of these fungal species recovered from the lower airway (Fedorova et al., 2008; Lake et al., 1991).
The Lung Microbiome in COPD
Introduction
COPD is the fourth leading cause of death in the US (Petty, 2000) and is expected to rank third in the world by 2020 (Lopez and Murray, 1998). Despite efforts aimed at smoking cessation, little impact has been made on COPD incidence, and current treatments are ineffective in slowing progression of the disease. COPD has been defined by the Global Initiative for Chronic Obstructive Lung Disease (GOLD) as “a disease state characterized by airflow limitation that is not fully reversible”. The diagnosis of COPD can also encompass those with chronic obstructive bronchiolitis and emphysema. Tissue inflammation in COPD is characterized by a predominant neutrophil, CD8+ lymphocyte, and macrophage infiltration (Keatings et al., 1996; Lacoste et al., 1993; O’Shaughnessy et al., 1997; Saetta et al., 1998). It has been proposed that the mechanism of tissue damage involves the recruitment and activation of neutrophils, macrophages, and CD8+ T cells with concomitant upregulation of several cellular proteases and inflammatory cytokines.
Role of Infections in COPD
Although smoking is clearly the leading risk factor for COPD, not all smokers develop disease (Buist and Connett, 1993). While smoking can stimulate inflammation in the lungs, smokers with COPD have an increased inflammatory response than smokers without COPD, and inflammation can persist despite smoking cessation (Keatings et al., 1996; Lacoste et al., 1993; O’Shaughnessy et al., 1997; Saetta et al., 1998). These observations suggest that some other factor or factors contribute to development and perpetuation of the inflammatory response in COPD. Infection might be one such factor critical in triggering and perpetuating the inflammatory response in COPD. The mechanism by which infections might act to promote COPD progression has been termed the “vicious circle” hypothesis (Sethi, 2000a; Sethi and Murphy, 2008). In this scenario, smoking causes structural remodeling that renders smokers more likely to become colonized and/or less able to clear subclinical infection. Defects in mucociliary clearance and surfactant abnormalities caused by smoking also contribute to the tendency to develop chronic infection (Finley and Ladman, 1972; Honda et al., 1996; Raju et al., 2008; Vastag et al., 1985; Verra et al., 1995). Once colonization is established, the organism or organisms recruit white blood cells to the lungs, stimulating release of inflammatory cytokines and chemokines as well as proteases. Inability to clear the inciting organism perpetuates the cycle, ultimately resulting in tissue destruction, airway thickening, and clinical COPD.
Bacteria, Viruses, and Fungi Have All Been Linked to COPD
The most commonly implicated bacteria are H. influenzae, M. catarrhalis, S. pneumoniae, and P. aeruginosa (Sethi, 2004). Viruses that seem to be important in COPD include adenovirus, influenzae viruses, rhinovirus, respiratory syncytial virus, and human metapneumovirus (Mallia et al., 2006; Martinello et al., 2006; Retamales et al., 2001; Seemungal et al., 2001). These pathogens can be found in patients with COPD in the stable state and during exacerbations (Sethi, 2004). The colonization seen in patients with COPD is likely playing a role in disease and is not just an innocent bystander. For example, as bacterial load increases, FEV1 falls, and colonization has been associated with greater sputum purulence, increased sputum neutrophils, and increased levels of interleukin (IL)-8, tumor necrosis factor (TNF)-α, and neutrophil elastase (Obrian et al., 2007; Patel et al., 2002; Sethi, 2000b; Stockley et al., 2000). Exacerbations associated with viruses are more severe and last longer than those without a viral trigger (Papi et al., 2006; Seemungal et al., 2001). In addition, exacerbations associated with both bacteria and viruses may be more severe than those associated with single organisms (Obrian et al., 2007), suggesting the usefulness of metagenomic techniques in this disease.
Colonization with the fungus Pneumocystis jirovecii (Pc, formerly Pneumocystis carinii f. sp. hominis) has recently been implicated in COPD pathogenesis. This organism generally causes acute Pneumocystis pneumonia (PCP) in patients with immunosuppression such as those infected with HIV, but colonization with the organism occurs in both HIV+ and HIV− individuals and may be important in COPD. Colonization with Pc is increased in HIV patients with COPD and correlates with disease severity (Morris et al., 2004b; Probst et al., 2000). Animal models also support the role of Pc colonization in COPD. Christensen and colleagues recently reported that in immunocompetent mice, exposure to cigarette smoke and Pc colonization resulted in pulmonary function deficits and airspace enlargement characteristic of emphysema (Christensen et al., 2008). In a model of Pc colonization in simian/human-immunodeficiency virus (SHIV)-infected nonhuman primates (Norris et al., 2006), Pc-colonized animals developed airway obstruction and radiographic emphysema while animals infected with SHIV alone did not develop these changes (Shipley et al., 2010).
The Lung Microbiome and Human Immunodeficiency Virus Infections
Lung Diseases Remain a Leading Cause of Morbidity and Mortality in HIV Infection
Although pulmonary infections and neoplasms associated with HIV have decreased since the availability of highly active antiretroviral therapy (HAART) (Palella et al., 1998), some pulmonary conditions may actually be increasing in persons with HIV. Diseases such as COPD, asthma, and bronchiectasis were reported to be increased in those with HIV before the introduction of antiretroviral therapy, and a similar decrease in these conditions as seen in the opportunistic infections has not occurred after antiretroviral treatment of HIV. In fact, in a recent study of HIV+ patients, almost 4% of deaths were due to obstructive airway disease in 1998, a threefold increase from the pre-HAART era (Louie et al., 2002). Before the HAART era, HIV+ subjects were noted to have an accelerated form of emphysema with significant emphysematous disease seen in subjects less than 40 years old (Diaz et al., 1992, 2000). Both emphysema and airflow obstruction have been reported in HIV infection. Unlike many of the acquired immunodeficiency syndrome (AIDS)-defining opportunistic infections, HIV-associated COPD may actually be more common in the current era of HIV as it is frequently reported in those without a history of AIDS-related pulmonary complications and the now aging HIV+ population has a longer exposure to smoking and HIV.
Role of the Microbiome in Obstructive Lung Diseases in HIV
Given the immunological defects seen with HIV, it is quite possible that HIV+ subjects, especially those who smoke, are more prone to develop subclinical pulmonary infections, even if successfully treated with HAART. The changes that occur in the lung microbiome have not been studied in HIV, but microbial colonization is a likely factor in the accelerated COPD seen in this population. The vicious circle hypothesis of COPD could be further worsened in HIV+ patients by upregulation of HIV levels in the lung stimulated by pulmonary colonization. Several studies have shown that pulmonary infections increase lung levels of HIV. Koziel and colleagues reported that HIV RNA was detected in 62% of patients with active lung disease compared to 16% of asymptomatic subjects, independent of clinical stage of HIV and serum HIV RNA levels (Koziel et al., 1999). The lung appears to be an independent compartment for HIV replication as drug mutations found in BAL differ from those in blood (White et al., 2004). HIV in the lungs is associated with a lymphocytic alveolitis, particularly in those subjects with CD4 cell counts between 200 and 500 cells/μl, suggesting that the virus might act independently to stimulate pulmonary inflammation (Twigg et al., 1999). The relationship of HIV pulmonary viral levels, infections, inflammation, and COPD has not been examined.
Role of Pneumocystis Colonization in HIV-Associated COPD
Pneumocystis colonization is likely important in the pathogenesis of COPD in those with HIV as well as in the HIV- population. In HIV+ subjects, the prevalence of colonization is high, particularly if subjects smoke, and colonization is seen even in patients with high CD4 cell counts receiving HAART (Morris et al., 2004a). Anatomic emphysema is also more common in HIV+ patients with Pc colonization (Morris, unpublished data). It has recently been shown that Pneumocystis colonization in HIV+ subjects is associated with worse airway obstruction and an increased likelihood of clinical diagnosis of COPD, independent of smoking history and CD4 cell count (Morris et al., 2009). In addition, the SHIV-infected nonhuman primates described above serve as a model for the development of COPD in the setting of Pc colonization and HIV-like immunodeficiency (Norris et al., 2006; Shipley et al., in press).
Conclusions
Microorganisms including bacteria, fungi, and viruses play a central role in development, exacerbation, and progress of lung diseases. Even though normal lungs do not have a permanent resident microbiome, diseased lungs are acutely infected and/or chronically colonized. Standard laboratory practices have not properly reflected the entirety of the microbiomes, either in health or in disease, and thus newer sequence-based technologies have begun to reveal the true complexity of the lung microbiomes. Much more research still needs to be conducted in order to fully understand the microbial burden of the lungs, and how this burden relates to health and disease.
It is apparent that conditions that compromise the physical and immune system barriers to lung colonization by microbes result in chronic colonization and recurrent infections. These conditions include chronic inflammation as seen in HIV, asthma, and bronchiecstasis, or physical obstruction observed in CF and bronchiecstasis. The lung microbiomes in each of these conditions has not been properly explored to date, which limits our ability to make definitive conclusions on how to best manage these diseases. Our understanding of the fundamental role of viruses in the initial establishment and progress of asthma underscores how little knowledge exists on the role of viral infection in other chronic respiratory diseases. In addition, the fact that some bacterial populations and/or components seem to be protective against further and severe exacerbations in asthma opens the door to questions about the role of microorganisms in protecting against other diseases. The exact nature of this protection is not clearly understood, and great benefit would come from studies that further clarify these intriguing results. Furthermore, if some population structures aid in preventing disease progression, it is likely that other population structures may predispose episodes of acute acerbations and progression of the underlying disease condition. A comprehensive understanding of the dynamics of these microbiome interactions would likely result in better, more efficient therapies for these and other respiratory diseases.
Our developing microbiome analysis technology coupled with our increasing awareness of the potential positive and negative impact of lung population structure on respiratory system health and disease strongly supports the initiation of aggressive projects to characterize the human lung microbiome and its influence on health and disease.
References
Aly R, Maibach HE, Mandel A (1976) Bacterial flora in psoriasis. Br J Dermatol 95:603–606
Amin R, Dupuis A, Aaron SD, Ratjen F (2009) The effect of chronic infection with Aspergillus fumigatus on lung function and hospitalization in cystic fibrosis patients. Chest 137:171–176
Ammous Z, Hackett NR, Butler MW, Raman T, Dolgalev I, O’Connor TP, Harvey BG, Crystal RG (2008) Variability in small airway epithelial gene expression among normal smokers. Chest 133:1344–1353
Angly FE, Felts B, Breitbart M, Salamon P, Edwards RA, Carlson C, Chan AM, Haynes M, Kelley S, Liu H, Mahaffy JM, Mueller JE, Nulton J, Olson R, Parsons R, Rayhawk S, Suttle CA, Rohwer F (2006) The marine viromes of four oceanic regions. PLOS Biol 4:e368
Atkinson RW, Strachan DP, Anderson HR, Hajat S, Emberlin J (2006) Temporal associations between daily counts of fungal spores and asthma exacerbations. Occup Environ Med 63:580–590
Bahrani-Mougeot FK, Paster BJ, Coleman S, Barbuto S, Brennan MT, Noll J, Kennedy T, Fox PC, Lockhart PB (2007) Molecular analysis of oral and respiratory bacterial species associated with ventilator-associated pneumonia. J Clin Microbiol 45:1588–1593
Ball TM, Castro-Rodriguez JA, Griffith KA, Holberg CJ, Martinez FD, Wright AL (2000) Siblings, day-care attendance, and the risk of asthma and wheezing during childhood. N Engl J Med 343:538–543
Balzar S, Chu HW, Strand M, Wenzel S (2005) Relationship of small airway chymase-positive mast cells and lung function in severe asthma. Am J Respir Crit Care Med 171:431–439
Barker AF (2002) Bronchiectasis. N Engl J Med 346:1383–1393
Bickerman HA, Sproul EE, Barach AL (1958) An aerosol method of producing bronchial secretions in human subjects: a clinical technic for the detection of lung cancer. Dis Chest 33:347–362
Bik EM, Eckburg PB, Gill SR, Nelson KE, Purdom EA, Francois F, Perez-Perez G, Blaser MJ, Relman DA (2006) Molecular analysis of the bacterial microbiota in the human stomach. Proc Natl Acad Sci USA 103:732–737
Billinger ME, Olivier KN, Viboud C, de Oca RM, Steiner C, Holland SM, Prevots DR (2009) Nontuberculous mycobacteria-associated lung disease in hospitalized persons, United States, 1998–2005. Emerg Infect Dis 15:1562–1569
Bisgaard H, Hermansen MN, Buchvald F, Loland L, Halkjaer LB, Bønnelykke K, Brasholt M, Heltberg A, Vissing NH, Thorsen SV, Stage M, Pipper CB (2007) Childhood asthma after bacterial colonization of the airway in neonates. N Engl J Med 357:1487–1495
Bittar F, Richet H, Dubus JC, Reynaud-Gaubert M, Stremler N, Sarles J, Raoult D, Rolain JM (2008). Molecular detection of multiple emerging pathogens in sputa from cystic fibrosis patients. PLOS One 3:e2908
Blaser MJ, Chen Y, Reibman J (2008) Does Helicobacter pylori protect against asthma and allergy? Br Med J 57:561
Bozza S, Gaziano R, Spreca A, Bacci A, Montagnoli C, di Francesco P, Romani L (2002) Dendritic cells transport conidia and hyphae of Aspergillus fumigatus from the airways to the draining lymph nodes and initiate disparate Th responses to the fungus. J Immunol 168:1362
Buist AS, Connett JE (1993) The lung health study. Baseline characteristics of randomized participants. Chest 103:1644
Busse WW, Lemanske RF Jr (2001) Asthma. N Engl J Med 344:350–362
Camporota L, Corkhill A, Long H et al (2003) The effects of Mycobacterium vaccae on allergen-induced airway responses in atopic asthma. Eur Respir J 21:287–293
Celedon JC, Litonjua AA, Weiss ST, Gold DR (1999) Day-care attendance in the first year of life and illnesses of the upper and lower respiratory tract in children with a familial history of atopy. Pediatrics 104:495–500
Chakravorty S, Helb D, Burday M, Connell N, Alland D (2007) A detailed analysis of 16S ribosomal RNA gene segments for the diagnosis of pathogenic bacteria. J Microbiol Methods 69:330–339
Christensen PJ, Preston AM, Ling T, Du M, Fields WB, Curtis JL, Beck JM (2008) Pneumocystis murina infection and cigarette smoke exposure interact to cause increased organism burden, development of airspace enlargement, and pulmonary inflammation in mice. Infect Immun 76:3481–3490
Colombo RE, Hill SC, Claypool RJ, Holland SM, Olivier KN (2009) Familial clustering of pulmonary nontuberculous mycobacterial disease. Chest 137:629–634
Cooley L, Spelman D, Thursky K, Slavin M (2007) Infection with Scedosporium apiospermum and S. prolificans, Australia. Emerg Infect Dis 13:1170–1177
Crameri R, Zeller S, Glaser AG, Vilhelmsson M, Rhyner C (2009) Cross-reactivity among fungal allergens: a clinically relevant phenomenon? Mycoses 52:99–106
Dales R, Miller D, Ruest K, Guay M, Judek S (2006) Airborne endotoxin is associated with respiratory illness in the first 2 years of life. Environ Health Perspect 114:610–614
Dekio I, Hayashi H, Sakamoto M, Kitahara M, Nishikawa T, Suematsu M, Benno Y (2005) Detection of potentially novel bacterial components of the human skin microbiota using culture-independent molecular profiling. J Med Microbiol 54:1231–1238
Denning DW, O’Driscoll BR, Hogaboam CM, Bowyer P, Niven RM (2006) The link between fungi and severe asthma: a summary of the evidence. Eur Respir J 27:615–626
Denning DW, O’Driscoll BR, Powell G, Chew F, Atherton GT, Vyas A, Miles J, Morris J, Niven RM (2009) Randomized controlled trial of oral antifungal treatment for severe asthma with fungal sensitization: the fungal asthma sensitization trial (FAST) study. Am J Respir Crit Care Med 179:11–18
Devulapalli CS, Carlsen KC, Haland G, Munthe-Kaas MC, Pettersen M, Mowinckel P, Carlsen KH (2008) Severity of obstructive airways disease by age 2 years predicts asthma at 10 years of age. Thorax 63:8–13
Diamond G, Legarda D, Ryan LK (2000) The innate immune response of the respiratory epithelium. Immunol Rev 173:27–38
Diaz PT, Clanton TL, Pacht ER (1992) Emphysema-like pulmonary disease associated with human immunodeficiency virus infection. Ann Intern Med 116:124–128
Diaz PT, King MA, Pacht ER, Wewers MD, Gadek JE, Nagaraja HN, Drake J, Clanton TL (2000) Increased susceptibility to pulmonary emphysema among HIV-seropositive smokers. Ann Intern Med 132:369–372
Djikeng A, Halpin R, Kuzmickas R, DePasse J, Feldblyum J, Sengamalay N, Afonso C, Zhang X, Anderson NG, Ghedin E, Spiro DJ (2008) Viral genome sequencing by random priming methods. BMC Genomics 9:5
Eckburg PB, Bik EM, Bernstein CN, Purdom E, Dethlefsen L, Sargent M, Gill SR, Nelson KE, Relman DA (2005) Diversity of the human intestinal microbial flora. Science 308:1635–1638
Fedorova ND, Khaldi N, Joardar VS, Maiti R, Amedeo P, Anderson MJ, Crabtree J, Silva JC, Badger JH, Albarraq A, Angiuoli S, Bussey H, Bowyer P, Cotty PJ, Dyer PS, Egan A, Galens K, Fraser-Liggett CM, Haas BJ, Inman JM, Kent R, Lemieux S, Malavazi L, Orvis J, Roemer T, Ronning CM, Sundaram JP, Sutton G, Turner G, Venter JC, White OR, Whitty BR, Youngman P, Wolfe KH, Goldman GH, Wortman JR, Jiang B, Denning DW, Nierman WC (2008) Genomic islands in the pathogenic filamentous fungus Aspergillus fumigatus. PLOS Genet 4:e1000046
Fennessy JJ (1967) Transbronchial biopsy of peripheral lung lesions. Radiology 88:878–882
Finley TN, Ladman AJ (1972) Low yield of pulmonary surfactant in cigarette smokers. N Engl J Med 286:223–227
Flanagan JL, Brodie EL, Weng L, Lynch SV, Wiener-Kronish JP, Bristow J (2007) Loss of bacterial diversity during antibiotic treatment of intubated patients colonized with Pseudomonas aeruginosa. J Clin Microbiol 45:1954–1962
Foulongne V, Guyon G, Rodiere M, Segondy M (2006) Human metapneumovirus infection in young children hospitalized with respiratory tract disease. Pediatr Infect Dis J 25:354–359
Frank DN, St Amand AL, Feldman RA, Boedeker EC, Harpaz N, Pace NR (2007) Molecular-phylogenetic characterization of microbial community imbalances in human inflammatory bowel diseases. Proc Natl Acad Sci USA 104:13780–13785
Fraser RS (2005) Histology and gross anatomy of the respiratory tract. In: Hamid Q, Shannon J, Martin J (eds) Physiologic basis of respiratory disease. BK Decker, Hamilton, ON, pp 133–141
Fujita SI, Senda Y, Nakaguchi S, Hashimoto T (2001) Multiplex PCR using internal transcribed spacer 1 and 2 regions for rapid detection and identification of yeast strains. J Clin Microbiol 39:3617–3622
Gao Z, Tseng CH, Pei Z, Blaser MJ (2007) Molecular analysis of human forearm superficial skin bacterial biota. Proc Natl Acad Sci USA 104:2927–2932
Gao Z, Tseng CH, Strober BE, Pei Z, Blaser MJ (2008) Substantial alterations of the cutaneous bacterial biota in psoriatic lesions. PLOS One 3:e2719
Gern JE, Vrtis R, Grindle KA, Swenson C, Busse WW (2000) Relationship of upper and lower airway cytokines to outcome of experimental rhinovirus infection. Am J Respir Crit Care Med 162:2226–2231
Gerritsen J (2000) Host defence mechanisms of the respiratory system. Paediatr Respir Rev 1:128–134
Gerritsen J (2002) Follow-up studies of asthma from childhood to adulthood. Paediatr Respir Rev 3:184–192
Gill SR, Pop M, Deboy RT, Eckburg PB, Turnbaugh PJ, Samuel BS, Gordon JI, Relman DA, Fraser-Liggett CM, Nelson KE (2006) Metagenomic analysis of the human distal gut microbiome. Science NY 312:1355–1359
Gold DR, Wright R (2005) Population disparities in asthma. Annu Rev Public Health 26:89–113
Gophna U, Sommerfeld K, Gophna S, Doolittle WF, Veldhuyzen van Zanten SJ (2006) Differences between tissue-associated intestinal microfloras of patients with Crohn’s disease and ulcerative colitis. J Clin Microbiol 44:4136–4141
Green RM, Custovic A, Sanderson G, Hunter J, Johnston SL, Woodcock A (2002) Synergism between allergens and viruses and risk of hospital admission with asthma: case-control study. BMJ 324:763
Guilbert TW, Morgan WJ, Zeiger RS et al (2006) Long-term inhaled corticosteroids in preschool children at high risk for asthma. N Engl J Med 354:1985–1997
Handelsman J, Rondon MR, Brady SF, Clardy J, Goodman RM (1998) Molecular biological access to the chemistry of unknown soil microbes: a new frontier for natural products. Chem Biol 5:R245–249
Harju TH, Leinonen M, Nokso-Koivisto J et al (2006). Pathogenic bacteria and viruses in induced sputum or pharyngeal secretions of adults with stable asthma. Thorax 61:579–584
Harris JK, De Groote MA, Sagel SD et al (2007) Molecular identification of bacteria in bronchoalveolar lavage fluid from children with cystic fibrosis. Proc Natl Acad Sci USA 104:20529–20533
Harrison F (2007) Microbial ecology of the cystic fibrosis lung. Microbiology 153:917–923
Hawksworth DL, Rossman AY (1997) Where are all the undescribed fungi? Phytopathology 87:888–891
Henderson AJ (1994) Bronchoalveolar lavage. Arch Dis Child 70:167–169
Hilty M, Burke C, Pedro H et al (2010) Disordered microbial communities in asthmatic airways. PLOS One 5:e8578
Holgate ST (2008) Pathogenesis of asthma. Clin Exp Allergy 38:872–897
Holt PG (1996) Infections and the development of allergy. Toxicol Lett 86:205–210
Honda Y, Takahashi H, Kuroki Y, Akino T, Abe S (1996) Decreased contents of surfactant proteins A and D in BAL fluids of healthy smokers. Chest 109:1006–1009
Horner WE, Helbling A, Salvaggio JE, Lehrer SB (1995) Fungal allergens. Clin Microbiol Rev 8:161–179
Hugenholtz P, Goebel BM, Pace NR (1998) Impact of culture-independent studies on the emerging phylogenetic view of bacterial diversity. J Bacteriol 180:4765–4774
Hyman RW, Fukushima M, Diamond L, Kumm J, Giudice LC, Davis RW (2005) Microbes on the human vaginal epithelium. Proc Natl Acad Sci USA 102:7952–7957
Jackson DJ, Gangnon RE, Evans MD et al (2008) Wheezing rhinovirus illnesses in early life predict asthma development in high-risk children. Am J Respir Crit Care Med 178:667–672
Jarvis D, Chinn S, Luczynska C, Burney P (1997) The association of family size with atopy and atopic disease. Clin Exp Allergy 27:240–245
Johnston SL, Martin RJ (2005) Chlamydophila pneumoniae and Mycoplasma pneumoniae: a role in asthma pathogenesis? Am J Respir Crit Care Med 172:1078–1089
Kazor CE, Mitchell PM, Lee AM, Stokes LN, Loesche WJ, Dewhirst FE, Paster BJ (2003) Diversity of bacterial populations on the tongue dorsa of patients with halitosis and healthy patients. J Clin Microbiol 41:558–563
Keatings VM, Collins PD, Scott DM, Barnes PJ (1996) Differences in interleukin-8 and tumor necrosis factor-alpha in induced sputum from patients with chronic obstructive pulmonary disease or asthma. Am J Respir Crit Care Med 153:530–534
Kirby AC, Coles MC, Kaye PM (2009) Alveolar macrophages transport pathogens to lung draining lymph nodes. J Immunol 183:1983
Kistler A, Avila PC, Rouskin S et al (2007) Pan-viral screening of respiratory tract infections in adults with and without asthma reveals unexpected human coronavirus and human rhinovirus diversity. J Infect Dis 196:817–825
Knowles MR, Durie PR (2002) What is cystic fibrosis? N Engl J Med 347:439–442
Kobashi Y, Fukuda M, Yoshida K, Miyashita N, Niki Y, Oka M (2006) Chronic necrotizing pulmonary aspergillosis as a complication of pulmonary Mycobacterium avium complex disease. Respirology 11:809–813
Korting HC, Lukacs A, Braun-Falco O (1988) Microbial flora and odor of the healthy human skin. Hautarzt 39:564–568
Koziel H, Kim S, Reardon C, Li X, Garland R, Pinkston P, Kornfeld H (1999) Enhanced in vivo human immunodeficiency virus-1 replication in the lungs of human immunodeficiency virus-infected persons with Pneumocystis carinii pneumonia. Am J Respir Crit Care Med 160:2048–2055
Kraft M, Adler KB, Ingram JL, Crews AL, Atkinson TP, Cairns CB, Krause DC, Chu HW (2008) Mycoplasma pneumoniae induces airway epithelial cell expression of MUC5AC in asthma. Eur Respir J 31:43–46
Krishnan V, Diette GB, Rand CS, Bilderback AL, Merriman B, Hansel NN, Krishnan JA (2006) Mortality in patients hospitalized for asthma exacerbations in the United States. Am J Respir Crit Care Med 174:633–638
Kroes I, Lepp PW, Relman DA (1999) Bacterial diversity within the human subgingival crevice. Proc Natl Acad Sci USA 96:14547–14552
Kunst H, Wickremasinghe M, Wells A, Wilson R (2006) Nontuberculous mycobacterial disease and aspergillus-related lung disease in bronchiectasis. Eur Respir J 28:352–357
Kurokawa K, Itoh T, Kuwahara T et al (2007) Comparative metagenomics revealed commonly enriched gene sets in human gut microbiomes. DNA Res 14:169–181
Kusel MM, de Klerk NH, Kebadze T, Vohma V, Holt PG, Johnston SL, Sly PD (2007) Early-life respiratory viral infections, atopic sensitization, and risk of subsequent development of persistent asthma. J Allergy Clin Immunol 119:1105–1110
Lacoste JY, Bousquet J, Chanez P et al (1993) Eosinophilic and neutrophilic inflammation in asthma, chronic bronchitis, and chronic obstructive pulmonary disease. J Allergy Clin Immunol 92:537–548
Lake FR, Froudist JH, McAleer R, Gillon RL, Tribe AE, Thompson PJ (1991) Allergic bronchopulmonary fungal disease caused by bipolaris and curvularia. Aust N Z J Med 21:871–874
Lass-Florl C, Salzer GM, Schmid T, Rabl W, Ulmer H, Dierichi, MP (1999) Pulmonary aspergillus colonization in humans and its impact on management of critically ill patients. Br J Haematol 104:745–747
Lemanske RF Jr, Jackson DJ, Gangnon RE et al (2005) Rhinovirus illnesses during infancy predict subsequent childhood wheezing. J Allergy Clin Immunol 116:571–577
Lieberman D, Printz S, Ben-Yaakov M, Lazarovich Z, Ohana B, Friedman MG, Dvoskin B, Leinonen M, Boldur, I (2003) Atypical pathogen infection in adults with acute exacerbation of bronchial asthma. Am J Respir Crit Care Med 167:406–410
Lilly CM (2005) Diversity of asthma: evolving concepts of pathophysiology and lessons from genetics. J Allergy Clin Immunol 115:S526–531
Lopez AD, Murray CC (1998) The global burden of disease, 1990–2020. Nat Med 4:1241–1243
Louie JK, Hsu LC, Osmond DH, Katz MH, Schwarcz SK (2002) Trends in causes of death among persons with acquired immunodeficiency syndrome in the era of highly active antiretroviral therapy, San Francisco, 1994–1998. J Infect Dis 186:1023–1027
Makimura K (2001) Species identification system for dermatophytes based on the DNA sequences of nuclear ribosomal internal transcribed spacer 1. Nihon Ishinkin Gakkai zasshi = Jpn J Med Mycol 42:61–67
Malde B, Greenberger PA (2004) Allergic bronchopulmonary aspergillosis. Allergy Asthma Proc 25:S38–39
Mallia P, Message SD, Kebadze T, Parker HL, Kon OM, Johnston SL (2006) An experimental model of rhinovirus induced chronic obstructive pulmonary disease exacerbations: a pilot study. Respir Res 7:116
Malling HJ (1986) Diagnosis and immunotherapy of mould allergy. IV. Relation between asthma symptoms, spore counts and diagnostic tests. Allergy 41:342–350
Manichanh C, Rigottier-Gois L, Bonnaud E et al (2006) Reduced diversity of faecal microbiota in Crohn’s disease revealed by a metagenomic approach. Gut 55:205–211
Marra F, Lynd L, Coombes M, Richardson K, Legal M, Fitzgerald JM, Marra CA (2006) Does antibiotic exposure during infancy lead to development of asthma?: a systematic review and metaanalysis. Chest 129:610–618
Marras TK, Chedore P, Ying AM, Jamieson F (2007) Isolation prevalence of pulmonary nontuberculous mycobacteria in Ontario, 1997–2003. Thorax 62:661–666
Martin RJ, Kraft M, Chu HW, Berns EA, Cassell GH (2001) A link between chronic asthma and chronic infection. J Allergy Clin Immunol 107:595–601
Martinello RA, Esper F, Weibel C, Ferguson D, Landry ML, Kahn JS (2006) Human metapneumovirus and exacerbations of chronic obstructive pulmonary disease. J Infect 53:248–254
Martinez FD, Wright AL, Taussig LM, Holberg CJ, Halonen M, Morgan WJ (1995) Asthma and wheezing in the first six years of life. The group health medical associates. N Engl J Med 332:133–138
Martinez FD (2007) Genes, environments, development and asthma: a reappraisal. Eur Respir J 29:179–184
Matheson MC, Abramson MJ, Dharmage SC, Forbes AB, Raven JM, Thien FC, Walters, EH (2005) Changes in indoor allergen and fungal levels predict changes in asthma activity among young adults. Clin Exp Allergy 35:907–913
Michel O, Kips J, Duchateau J, Vertongen F, Robert L, Collet H, Pauwels R, Sergysels R (1996) Severity of asthma is related to endotoxin in house dust. Am J Respir Crit Care Med 154:1641–1646
Morris A, Kingsley LA, Groner G, Lebedeva IP, Beard CB, Norris KA (2004a) Prevalence and clinical predictors of pneumocystis colonization among HIV-infected men. AIDS 18:793–798
Morris A, Sciurba FC, Lebedeva IP, Githaiga A, Elliott WM, Hogg JC, Huang L, Norris KA (2004b) Association of chronic obstructive pulmonary disease severity and pneumocystis colonization. Am J Respir Crit Care Med 170:408–413
Morris A, Alexander T, Radhi S, Lucht L, Sciurba FC, Kolls JK, Srivastava R, Steele C, Norris KA (2009) Airway obstruction is increased in pneumocystis-colonized human immunodeficiency virus-infected outpatients. J Clin Microbiol 47:3773–3776
Nierman WC, Pain A, Anderson MJ et al (2005) Genomic sequence of the pathogenic and allergenic filamentous fungus Aspergillus fumigatus. Nature 438:1151–1156
Norris KA, Morris A, Patil S, Fernandes E (2006) Pneumocystis colonization, airway inflammation, and pulmonary function decline in acquired immunodeficiency syndrome. Immunol Res 36:175–187
O’Driscoll BR, Hopkinson LC, Denning DW (2005) Mold sensitization is common amongst patients with severe asthma requiring multiple hospital admissions. BMC Pulm Med 5:4
O’Driscoll BR, Powell G, Chew F, Niven RM, Miles JF, Vyas A, Denning DW (2009) Comparison of skin prick tests with specific serum immunoglobulin E in the diagnosis of fungal sensitization in patients with severe asthma. Clin Exp Allergy 39:1677–1683
O’Shaughnessy TC, Ansari TW, Barnes NC, Jeffery PK (1997) Inflammation in bronchial biopsies of subjects with chronic bronchitis: inverse relationship of CD8+ T lymphocytes with FEV1. Am J Respir Crit Care Med 155:852–857
Obrian GR, Georgianna DR, Wilkinson JR, Yu J, Abbas HK, Bhatnagar D, Cleveland TE, Nierman W, Payne GA (2007) The effect of elevated temperature on gene transcription and aflatoxin biosynthesis. Mycologia 99:232
Okudaira M, Kurata H, Sakabe F (1977) Studies on the fungal flora in the lung of human necropsy cases. A critical survey in connection with the pathogenesis of opportunistic fungus infections. Mycopathologia 61:3–18
Palella FJ Jr, Delaney KM, Moorman AC, Loveless MO, Fuhrer J, Satten GA, Aschman DJ, Holmberg SD (1998) Declining morbidity and mortality among patients with advanced human immunodeficiency virus infection. HIV outpatient study Investigators. N Engl J Med 338:853–860
Papadopoulos NG, Kalobatsou A (2007) Respiratory viruses in childhood asthma. Curr Opin Allergy Clin Immunol 7:91–95
Papi A, Bellettato CM, Braccioni F, Romagnoli M, Casolari P, Caramori G, Fabbri LM, Johnston SL (2006) Infections and airway inflammation in chronic obstructive pulmonary disease severe exacerbations. Am J Respir Crit Care Med 173:1114–1121
Park JH, Gold DR, Spiegelman DL, Burge HA, Milton DK (2001) House dust endotoxin and wheeze in the first year of life. Am J Respir Crit Care Med 163:322–328
Pasqualotto AC, Powell G, Niven R, Denning DW (2009) The effects of antifungal therapy on severe asthma with fungal sensitization and allergic bronchopulmonary aspergillosis. Respirology 14:1121–1127
Paster BJ, Boches SK, Galvin JL, Ericson RE, Lau CN, Levanos VA, Sahasrabudhe A, Dewhirst FE (2001) Bacterial diversity in human subgingival plaque. J Bacteriol 183:3770–3783
Patel IS, Seemungal TA, Wilks M, Lloyd-Owen SJ, Donaldson GC, Wedzicha JA (2002) Relationship between bacterial colonisation and the frequency, character, and severity of COPD exacerbations. Thorax 57:759–764
Pattemore PK, Johnston SL, Bardin PG (1992) Viruses as precipitants of asthma symptoms. I. Epidemiology. Clin Exp Allergy 22:325–336
Peebles RS Jr (2004) Viral infections, atopy, and asthma: is there a causal relationship? J Allergy Clin Immunol 113:S15–18
Petty TL (2000) Scope of the COPD problem in North America: early studies of prevalence and NHANESIII data: basis for early identification and intervention. Chest 117:326S–331S
Probst M, Ries H, Schmidt-Wieland T, Serr A (2000) Detection of Pneumocystis carinii DNA in patients with chronic lung diseases. Eur J Clin Microbiol Infect Dis 19:644–645
Pulimood TB, Corden JM, Bryden C, Sharples L, Nasser SM (2007) Epidemic asthma and the role of the fungal mold Alternaria alternata. J Allergy Clin Immunol 120:610–617
Raju B, Hoshino Y, Belitskaya-Lévy I et al (2008) Gene expression profiles of bronchoalveolar cells in pulmonary TB. Tuberculosis 88:39–51
Ran H, Hassett DJ, Lau GW (2003) Human targets of Pseudomonas aeruginosa pyocyanin. Proc Natl Acad Sci USA 100:1315–14320
Relman DA, Falkow S, LeBoit PE, Perkocha LA, Min KW, Welch DF, Slater LN (1991).The organism causing bacillary angiomatosis, peliosis hepatis, and fever and bacteremia in immunocompromised patients. N Engl J Med 324:1514
Retamales I, Elliott WM, Meshi B, Coxson HO, Pare PD, Sciurba FC, Rogers RM, Hayashi S, Hogg JC (2001) Amplification of inflammation in emphysema and its association with latent adenoviral infection. Am J Respir Crit Care Med 164:469–473
Reynolds HY, Chretien J (1984) Respiratory tract fluids: analysis of content and contemporary use in understanding lung diseases. Dis Mon 30:1–103
Rokas A, Payne G, Fedorova ND et al (2007) What can comparative genomics tell us about species concepts in the genus Aspergillus? Stud Mycol 59:11
Saetta M, Di Stefano A, Turato G, Facchini FM, Corbino L, Mapp CE, Maestrelli P, Ciaccia A, Fabbri LM (1998) CD8+ T lymphocytes in peripheral airways of smokers with chronic obstructive pulmonary disease. Am J Respir Crit Care Med 157:822–826
Salo PM, Arbes SJ Jr, Sever M, Jaramillo R, Cohn RD, London SJ, Zeldin DC (2006) Exposure to Alternaria alternata in US homes is associated with asthma symptoms. J Allergy Clin Immunol 118:892–898
Scott JP, Fradet G, Smyth RL, Mullins P, Pratt A, Clelland CA, Higenbottam T, Wallwork J (1991) Prospective study of transbronchial biopsies in the management of heart–lung and single lung transplant patients. J Heart Lung Transplant 10:626–636;Discussion 636–627
Seemungal T, Harper-Owen R, Bhowmik A et al (2001) Respiratory viruses, symptoms, and inflammatory markers in acute exacerbations and stable chronic obstructive pulmonary disease. Am J Respir Crit Care Med 164:1618–1623
Segreti J, House HR, Siegel RE (2005) Principles of antibiotic treatment of community-acquired pneumonia in the outpatient setting. Am J Med 118:21–28
Sethi S (2000a) Bacterial infection and the pathogenesis of COPD. Chest 117:286S–291S
Sethi S (2000b) Infectious etiology of acute exacerbations of chronic bronchitis. Chest 117:380S–385S
Sethi S (2004) Bacteria in exacerbations of chronic obstructive pulmonary disease: phenomenon or epiphenomenon? Proc Am Thorac Soc 1:109–114
Sethi S, Murphy TF (2008) Infection in the pathogenesis and course of chronic obstructive pulmonary disease. N Engl J Med 359:2355–2365
Shirakawa T, Enomoto T, Shimazu S, Hopkin JM (1997) The inverse association between tuberculin responses and atopic disorder. Science 275:77–79
Shipley TW, Kling HM, Morris A, Patil S, Kristoff J, Guyach SE, Murphy JM, Shao X, Sciuraba FC, Rodgers RM, Richards T, Thompson P, Montelaro RC, Coxson HO, Higg JC, Norris KA (2010) Persistent pneumocystis colonization leads to development of chronic obstructive pulmonary disease in a non-human primate model of AIDS. J Inf Dis 202:302–312
Shoseyov D, Brownlee KG, Conway SP, Kerem E (2006) Aspergillus bronchitis in cystic fibrosis. Chest 130:222–226
Sigurs N, Gustafsson PM, Bjarnason R, Lundberg F, Schmidt S, Sigurbergsson F, Kjellman B (2005) Severe respiratory syncytial virus bronchiolitis in infancy and asthma and allergy at age 13. Am J Respir Crit Care Med 171:137–141
Singh AM, Moore PE, Gern JE, Lemanske RF Jr, Hartert TV (2007) Bronchiolitis to asthma: a review and call for studies of gene–virus interactions in asthma causation. Am J Respir Crit Care Med 175:108–119
Soong LB, Ganz T, Ellison A, Caughey GH (1997) Purification and characterization of defensins from cystic fibrosis sputum. Inflamm Res 46:98–102
Speert DP (2006) Bacterial infections of the lung in normal and immunodeficient patients. Novartis Found Symp 279:42–51
Stockley RA, Hill AT, Hill SL, Campbell EJ (2000). Bronchial inflammation: its relationship to colonizing microbial load and alpha(1)-antitrypsin deficiency. Chest 117:291S–293S
Stone KC, Mercer RR, Freeman BA, Chang LY, Crapo JD (1992) Distribution of lung cell numbers and volumes between alveolar and nonalveolar tissue. Am Rev Respir Dis 146:454
Strachan DP (1989) Hay fever, hygiene, and household size. Br Med J 299:1259–1260
Syhre M, Scotter JM, Chambers ST (2008) Investigation into the production of 2-Pentylfuran by Aspergillus fumigatus and other respiratory pathogens in vitro and human breath samples. Med Mycol 46:209–215
Taylor DR, Cowan JO, Greene JM, Willan AR, Sears MR (2005) Asthma in remission: can relapse in early adulthood be predicted at 18 years of age? Chest 127:845–850
Thavagnanam S, Fleming J, Bromley A, Shields MD, Cardwell CR (2008) A meta-analysis of the association between caesarean section and childhood asthma. Clin Exp Allergy 38:629–633
Thomson NC, Chaudhuri R, Livingston E (2004) Asthma and cigarette smoking. Eur Respir J 24:822–833
Trulock EP, Ettinger NA, Brunt EM, Pasque MK, Kaiser LR, Cooper JD (1992) The role of transbronchial lung biopsy in the treatment of lung transplant recipients. An analysis of 200 consecutive procedures. Chest 102:1049–1054
Tunney MM, Field TR, Moriarty TF et al (2008) Detection of anaerobic bacteria in high numbers in sputum from patients with cystic fibrosis. Am J Respir Crit Care Med 177:995–1001
Turnbaugh PJ, Ley RE, Mahowald MA, Magrini V, Mardis ER, Gordon JI (2006) An obesity-associated gut microbiome with increased capacity for energy harvest. Nature 444:1027–1031
Twigg HL, Soliman DM, Day RB, Knox KS, Anderson RJ, Wilkes DS, Schnizlein-Bick CT (1999) Lymphocytic alveolitis, bronchoalveolar lavage viral load, and outcome in human immunodeficiency virus infection. Am J Respir Crit Care Med 159:1439–1444
Valenza G, Tappe D, Turnwald D, Frosch M, Konig C, Hebestreit H, Abele-Horn M (2008) Prevalence and antimicrobial susceptibility of microorganisms isolated from sputa of patients with cystic fibrosis. J Cyst Fibros 7:123–127
Vastag E, Matthys H, Kohler D, Gronbeck L, Daikeler G (1985) Mucociliary clearance and airways obstruction in smokers, ex-smokers and normal subjects who never smoked. Eur J Respir Dis Suppl 139:93–100
Venarske DL, Busse WW, Griffin MR et al (2006). The relationship of rhinovirus-associated asthma hospitalizations with inhaled corticosteroids and smoking. J Infect Dis 193:1536–1543
Verra F, Escudier E, Lebargy F, Bernaudin JF, De Cremoux H, Bignon J (1995) Ciliary abnormalities in bronchial epithelium of smokers, ex-smokers, and nonsmokers. Am J Respir Crit Care Med 151:630–634
von Mutius E, Braun-Fahrlander C, Schierl R, Riedler J, Ehlermann S, Maisch S, Waser M, Nowak D (2000) Exposure to endotoxin or other bacterial components might protect against the development of atopy. Clin Exp Allergy 30:1230–1234
Vonk JM, Postma DS, Boezen HM, Grol MH, Schouten JP, Koeter GH, Gerritsen J (2004) Childhood factors associated with asthma remission after 30 year follow up. Thorax 59:925–929
Wark PA, Johnston SL, Moric I, Simpson JL, Hensley MJ, Gibson PG (2002) Neutrophil degranulation and cell lysis is associated with clinical severity in virus-induced asthma. Eur Respir J 19:68–75
Weycker D, Edelsberg J, Oster G, Tino G (2005) Prevalence and economic burden of bronchiectasis. Clin Pulm Med 12:205–209
White NC, Israel-Biet D, Coker RJ, Mitchell DM, Weber JN, Clarke JR (2004) Different resistance mutations can be detected simultaneously in the blood and the lung of HIV-1 infected individuals on antiretroviral therapy. J Med Virol 72:352–357
Williams JV, Harris PA, Tollefson SJ, Halburnt-Rush LL, Pingsterhaus JM, Edwards KM, Wright PF, Crowe JE Jr (2004) Human metapneumovirus and lower respiratory tract disease in otherwise healthy infants and children. N Engl J Med 350:443–450
Willner D, Furlan M, Haynes M et al (2009) Metagenomic analysis of respiratory tract DNA viral communities in cystic fibrosis and non-cystic fibrosis individuals. PLOS One 4:e7370
Woodcock AA, Steel N, Moore CB, Howard SJ, Custovic A, Denning DW (2006) Fungal contamination of bedding. Allergy 61:140–142
Worlitzsch D, Rintelen C, Bohm K, Wollschlager B, Merkel N, Borneff-Lipp M, Doring G (2009) Antibiotic-resistant obligate anaerobes during exacerbations of cystic fibrosis patients. Clin Microbiol Infect 15:454–460
Wu P, Dupont WD, Griffin MR, Carroll KN. Mitchel EF, Gebretsadik T, Hartert TV (2008) Evidence of a causal role of winter virus infection during infancy in early childhood asthma. Am J Respir Crit Care Med 178:1123–1129
Wu Q, Chu HW (2009) Role of infections in the induction and development of asthma: genetic and inflammatory drivers. Expert Rev Clin Immunol 5:97–109
Yang X, Fan Y, Wang S, Han X, Yang J, Bilenki L, Chen L (2002) Mycobacterial infection inhibits established allergic inflammatory responses via alteration of cytokine production and vascular cell adhesion molecule-1 expression. Immunology 105:336–343
Zavala DC, Richardson RH, Mukerjee PK, Rossi NP, Bedell GN (1973) Use of the bronchofiberscope for bronchial brush biopsy. Diagnostic results and comparison with other brushing techniques. Chest 63:889–892
Zhang H, Su YA, Hu P, Yang J, Zheng B, Wu P, Peng J, Tang Y, Zhang L (2006) Signature patterns revealed by microarray analyses of mice infected with influenza virus A and Streptococcus pneumoniae. Microbes Infect 8:2172–2185
Zhou X, Bent SJ, Schneider MG, Davis CC, Islam MR, Forney LJ (2004) Characterization of vaginal microbial communities in adult healthy women using cultivation-independent methods. Microbiology 150:2565–2573
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2011 Springer Science+Business Media, LLC
About this chapter
Cite this chapter
Losada, L., Ghedin, E., Morris, A., Chu, H.W., Nierman, W.C. (2011). The Human Lung Microbiome. In: Nelson, K. (eds) Metagenomics of the Human Body. Springer, New York, NY. https://doi.org/10.1007/978-1-4419-7089-3_7
Download citation
DOI: https://doi.org/10.1007/978-1-4419-7089-3_7
Published:
Publisher Name: Springer, New York, NY
Print ISBN: 978-1-4419-7088-6
Online ISBN: 978-1-4419-7089-3
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)