Abstract
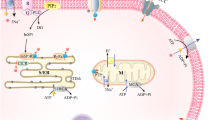
Sustained elevation in cytosolic Ca2+ concentration ([Ca2+]C) is believed to induce premature trypsin activation and initiate acute pancreatitis. Among many triggering factors, ethanol abuse is the most prevalent in provoking acute pancreatitis but with unknown etiological mechanism. Here we show that elevated [Ca2+]C is directly responsible for inducing premature trypsin activation in the zymogen granules (ZGs) of pancreas acinar cells and is exacerbated by ethanol treatment. In vitro, a highly cooperative ion exchange mechanism has been found to regulate elevation in [Ca2+]C induced premature trypsin activation. The sustained rise in [Ca2+]C triggers K+ influx into the ZGs in exchange for the release of matrix bound-Ca2+ and -H+. Such K+/Ca2+ ion-exchange mechanism promotes the amplification of mobile-Ca2+ concentration in the ZGs ([Ca2+]G) while K+ replaces bound-H+ by K+/H+ ion-exchange to decrease the pH within ZGs (pHG). Increasing [Ca2+]G with concomitant lowering of pHG generate premature trypsin activation in ZGs. Hence, our data suggest that ethanol abuse provokes a sustained rise in [Ca2+]C by which K+ influx induced ion-exchange mechanism initiates premature trypsin activation in acinar ZGs during acute pancreatitis.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Preview
Unable to display preview. Download preview PDF.
Similar content being viewed by others
Reference
Apte, M.V., and Wilson, J.S. (2003). Alcohol-induced pancreatic injury, Best. Pract. Res. Clin. Gastroenterol. 17, 593–612.
Colomb, E., Figarella, C., and Guy, O. (1979). The two human trypsinogens: Evidence of complex formation with basic pancreatic trypsin inhibitor-proteolytic activity. Biochim. Biophys. Acta. 570, 397–405.
Criddle, D.N., Raraty, M.G., Neoptolemos, J.P., Tepikin, A.V., Petersen, O.H., and Sutton, R. (2004). Ethanol toxicity in pancreatic acinar cells: mediation by nonoxidative fatty acid metabolites. Proc Natl. Acad. Sci. USA 101, 10738–10743.
Ding, Y.X., Yang, K., and Chin, W.C. (2006). Ethanol augments elevated-[Ca2+]C induced trypsin activation in pancreatic acinar zymogen granules. Biochem. Biophys. Res. Commun. 350(3), 593–597.
Dufour, M.C., and Adamson, M.D. (2003). The epidemiology of alcohol-induced pancreatitis, Pancreas. 27, 286–90.
Figarella, C., Miszcuk-Jamska, B., and Barret, A.J. (1988). Possible lysosomal activation of pancreatic zymogen: activation of both human trypsinogens by cathepsin B ans spontaneous acid activation of human trypsinogen. Biol. Chem. Hoppe-Seyler. 369, 293–298.
Frick, T.W., Fernandez-del-Castillo, C., Bimmler, D., and Warshaw, A.L. (1997). Elevated calcium and activation of trypsinogen in rat pancreatic acini. Gut. 41, 339–343.
Gardner, T.B, Merk, B.S., and Yakshe, P. (2006). Acute pancreatitis. http://www.emedicine.com/med/topic1720.htm.
Gonzalez A., Nunez A.M., Granados M.P., Pariente J.A., and Salido G.M. (2006). Ethanol impairs CCK-8-evoked amylase secretion through Ca2+-mediated ROS generation in mouse pancreatic acinar cells. Alcohol. 38, 51–57.
Gorelick, F.S., Modlin, I.M., Leach, S.D., Carangelo, R. and Katz, M. (1992). Intracellular proteolysis of pancreatic zymogens. Yale J Biol Med. 65, 407–420.
Gorelick, F.S., and Otani, T. (1999). Mechanisms of intracellular zymogen activation. Baillieres. Best. Pract. Res. Clin. Gastroenterol. 13, 227–240.
Halangk, W., and Lerch, M.M. (2004). Early events in acute pancreatitis. Gastroenterol. Clin. North. Am. 33, 717–731.
Kao, J.P.Y. (1994). In: A Practical Guide to the Study of Calcium in Living Cells (ed. Nuccitelle, R.) 155–181 (Academic, San Diego).
Kassell, B., and Kay, J. (1973). Zymogens of proteolytic enzymes. Science 180, 1022–1027.
Katz, M., Carangelo, R., Miller, L.J. and Gorelick, F. (1996). Effect of ethanol on cholecystokinin-stimulated zymogen conversion in pancreatic acinar cells, Am. J. Physiol. 270, G171–G175.
Kruger, B., Lerch, M.M., and Tessenow, W. (1998). Direct detection of premature protease activation in living pancreatic acinar cells. Lab. Invest. 78, 763–764.
Kruger, B., Albrecht, E., and Lerch, M.M. (2000). The role of intracellular calcium signaling in premature protease activation and the onset of pancreatitis. Am. J. Pathol. 157, 43–50.
Leytus, S.P., Melhado, L.L., and Mangel, W.F. (1983). Rhodamine-based compounds as fluorogenic substrates for serine proteinases. Biochem. J. 209, 299–307.
Mergener, K., and Baillie, J. (1998). Acute pancreatitis. B. M. J. 316, 44–48.
Nguyen, T., Chin, W.C., and Verdugo, P. (1998). Role of Ca2+/K+ ion exchange in intracellular storage and release of Ca2+. Nature 395, 908–912.
Parekh, A.B. (2000). Calcium signaling and acute pancreatitis: specific response to a promiscuous messenger. Proc. Natl. Acad. Sci. 97, 12933–12934.
Petersen, O.H., and Sutton, R. (2006). Ca2+ signaling and pancreatitis: effects of alcohol, bile and coffee. Trends. Pharmacol. Sci. 27, 113–120.
Quesada, I., Chin, W.C., Campos-Bedolla, P., Steed, J., and Verdugo, P. (2001). Mouse mast cell secretory granules can function as intracellular ionic oscillators. Biophys. J. 80, 2133–2139.
Raraty, M., Ward, J., Erdemli, G., Vaillant, C., Neoptolemos, J.P., Sutton, R., Petersen. O.H. (2000). Calcium-dependent enzyme activation and vacuole formation in the apical granular region of pancreatic acinar cells. Proc. Natl. Acad. Sci. 24, 13126–13131.
Saluja, A.K., Lu, L., Yamaguchi, Y., Hofbauer, B., Ruenzi, M., Dawra, R., Bhatia, M., and Steer, M.L. (1997). A cholecystokinin-releasing factor mediates ethanol-induced stimulation of rat pancreatic secretion. J. Clin. Invest. 99, 506–512.
Steer, M.L. (1999). Early events in acute pancreatitis. Baillieres. Best. Pract. Res. Clin. Gastroenterol. 13, 213–225.
Verdugo, P. (1990). Goblet cells and mucus secretion. Annu. Rev. Physiol. 52, 157–176.
Yang, K., Ding, Y.X., Chin, W.C. (2007). K+-induced ion-exchanges trigger trypsin activation in pancreas acinar zymogen granules. Arch. Biochem. Biophys. 459, 256–263.
Zhao, L., Karne, S., Kolodecik, T. and Gorelick, F.S. (2002). Alcohols enhance caerulein-induced zymogen activation in pancreatic acinar cells, Am. J. Physiol. Gastrointest. Liver Physiol. 282 G501–G507.
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2008 Springer Science+Business Media B.V.
About this chapter
Cite this chapter
Ding, Y., Chen, E., Yang, K., Chin, WC. (2008). The Role of Ion-Exchange on Trypsin Premature Activation in Zymogen Granules. In: Pollack, G.H., Chin, WC. (eds) Phase Transitions in Cell Biology. Springer, Dordrecht. https://doi.org/10.1007/978-1-4020-8651-9_4
Download citation
DOI: https://doi.org/10.1007/978-1-4020-8651-9_4
Publisher Name: Springer, Dordrecht
Print ISBN: 978-1-4020-8650-2
Online ISBN: 978-1-4020-8651-9
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)