Abstract
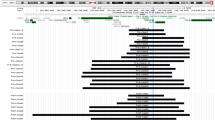
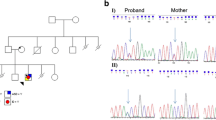
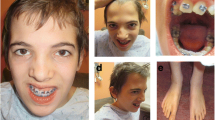
ATP7A duplications are estimated to represent the molecular cause of Menkes disease in 4–10% of affected patients. We identified a novel duplication of ATP7A exons 1–7 discovered in the context of a challenging prenatal diagnostic situation. All other reported ATP7A duplications (n = 24) involved intragenic tandem duplications, predicted to disrupt the normal translational reading frame and produce nonfunctional ATP7A proteins. In contrast, the exon 1–7 duplication occurred at the 5′ end of the ATP7A gene rather than within the gene and did not correspond to any known copy number variants. We hypothesized that, if the exon 1–7 duplication was in tandem, functional ATP7A molecules could be generated depending on promoter selection, mRNA splicing, and the proximal and distal duplication breakpoints and that Menkes disease would be averted. Here, we present detailed molecular characterization of this novel duplication, as well as 2-year postnatal clinical and biochemical correlations. The case highlights the ongoing need for cautious interpretation of prenatal genetic test results.
Competing interests: None declared
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Donsante A, Johnson P, Jansen LA, Kaler SG (2010) Somatic mosaicism in Menkes disease suggests choroid plexus-mediated copper transport to the developing brain. Am J Med Genet A 152A:2529–2534
Dutra AS, Mignot E, Puck JM (1996) Gene localization and syntenic mapping by FISH in the dog. Cytogenet Cell Genet 74:113–117
Haddad MR, Macri CJ, Holmes DS et al (2012) In utero copper treatment for Menkes disease associated with a severe ATP7A mutation. Mol Genet Metab 107:222–228
Kaler SG (1996) Menkes disease mutations and response to early copper histidine treatment. Nat Genet 13:21–22
Kaler SG (2011) The neurology of ATP7A copper transporter disease: emerging concepts and future trends. Nat Rev Neurol 7:15–29
Kaler SG, Packman S (2013) Inherited disorders of human copper metabolism. In: Rimoin DL, Connor JM, Pyeritz RE, Korf BR (eds) Emery and Rimoin’s principles and practice of medical genetics, 6th edn. Churchill Livingstone/Elsevier, New York
Kaler SG, Tumer Z (1998) Invited commentary: the prenatal diagnosis of Menkes disease. Prenat Diagn 18:287–289
Kaler SG, Gahl WA, Berry SA, Holmes CS, Goldstein DS (1993a) Predictive value of plasma catechol levels in neonatal detection of Menkes disease. J Inher Metab Dis 16:907–908
Kaler SG, Goldstein DS, Holmes C, Salerno JA, Gahl WA (1993b) Plasma and cerebrospinal fluid neurochemical pattern in Menkes disease. Ann Neurol 33:171–175
Kaler SG, Westman JA, Bernes SM et al (1993c) Gastrointestinal hemorrhage associated with gastric polyps in Menkes disease. J Pediatr 122:93–95
Kaler SG, Holmes CS, Goldstein DS et al (2008) Neonatal diagnosis and treatment of Menkes disease. N Engl J Med 358:605–614
Mogensen M, Skjørringe T, Kodama H, Silver K, Horn N, Møller LB (2011) Exon duplications in the ATP7A gene: frequency and transcriptional behaviour. Orphanet J Rare Dis 6:73
Moizard MP, Ronce N, Blesson S et al (2011) Twenty-five novel mutations including duplications in the ATP7A gene. Clin Genet 79:243–253
Schoonveld C, Donsante A, Holmes CS, Goldstein DS, Das S, Kaler SG (2013) Prenatal diagnostic conundrum involving a novel ATP7A duplication. Clin Genet 84:97–98
Sheela SR, Manoj L, Liu P-C, Lem KE, Kaler SG (2005) Copper replacement treatment for symptomatic Menkes disease: ethical considerations. Clin Genet 68:278–283
Tümer Z (2013) An overview and update of ATP7A mutations leading to Menkes disease and occipital horn syndrome. Hum Mutat 34:417–429
Yi L, Donsante A, Kennerson ML, Mercer JFB, Garbern JY, Kaler SG (2012) Altered intracellular localization and valosin-containing protein (p97 VCP) interaction underlie ATP7A-related distal motor neuropathy. Hum Mol Genet 21:1794–1807
Acknowledgments
We thank the patient’s parents for their kind cooperation in these studies.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Additional information
Communicated by: Gregory Enns
Compliance with Ethics Guidelines
Compliance with Ethics Guidelines
Informed Consent
All procedures followed were in accordance with the ethical standards of the responsible committee on human experimentation (institutional and national) and with the Helsinki Declaration of 1975, as revised in 2000. Informed consent was obtained from the parents of the patients included in the study.
This article does not contain any studies with animal subjects performed by the any of the authors.
Eun-Young Choi, Keyur Patel, Marie Reine Haddad, and Ling Yi performed the molecular and cell biological experiments described in this article. Courtney Holmes and David S. Goldstein performed the neurochemical analyses. Amalia Dutra and Evgenia Pak performed fluorescence in situ hybridization (FISH) experiments. Eun-Young Choi and Stephen Kaler planned the studies and wrote the manuscript.
All authors (Eun-Young Choi, Keyur Patel, Marie Reine Haddad, Ling Yi, Courtney Holmes, David S. Goldstein, Amalia Dutra, Evgenia Pak, and Stephen Kaler) declare that they have no conflict of interest.
Rights and permissions
Copyright information
© 2014 SSIEM and Springer-Verlag Berlin Heidelberg
About this chapter
Cite this chapter
Choi, EY. et al. (2014). Tandem Duplication of Exons 1–7 Neither Impairs ATP7A Expression Nor Causes a Menkes Disease Phenotype. In: Zschocke, J., Baumgartner, M., Morava, E., Patterson, M., Rahman, S., Peters, V. (eds) JIMD Reports, Volume 20. JIMD Reports, vol 20. Springer, Berlin, Heidelberg. https://doi.org/10.1007/8904_2014_391
Download citation
DOI: https://doi.org/10.1007/8904_2014_391
Received:
Revised:
Accepted:
Published:
Publisher Name: Springer, Berlin, Heidelberg
Print ISBN: 978-3-662-46699-5
Online ISBN: 978-3-662-46700-8
eBook Packages: MedicineMedicine (R0)