Abstract
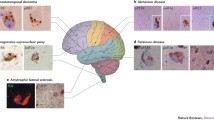
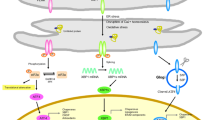
The accumulation of protein aggregates has a fundamental role in the patophysiology of distinct neurodegenerative diseases. This phenomenon may have a common origin, where disruption of intracellular mechanisms related to protein homeostasis (here termed proteostasis) control during aging may result in abnormal protein aggregation. The unfolded protein response (UPR) embodies a major element of the proteostasis network triggered by endoplasmic reticulum (ER) stress. Chronic ER stress may operate as possible mechanism of neurodegenerative and synaptic dysfunction, and in addition contribute to the abnormal aggregation of key disease-related proteins. In this article we overview the most recent findings suggesting a causal role of ER stress in neurodegenerative diseases.
Similar content being viewed by others
References
Abisambra JF, Jinwal UK, Blair LJ et al (2013) Tau accumulation activates the unfolded protein response by impairing endoplasmic reticulum-associated degradation. J Neurosci Off J Soc Neurosci 33(22):9498–9507
Acosta-Alvear D, Zhou Y, Blais A et al (2007) XBP1 controls diverse cell type-and condition-specific transcriptional regulatory networks. Mol Cell 27(1):53–66
Alzheimer’s Association (2015) Alzheimer’s Dementia: J Alzheimer’s Assoc 11(3):332–384
Apodaca J, Kim I, Rao H (2006) Cellular tolerance of prion protein PrP in yeast involves proteolysis and the unfolded protein response. Biochem Biophys Res Commun 347(1):319–326
Atkin JD, Farg MA, Turner BJ et al (2006) Induction of the unfolded protein response in familial amyotrophic lateral sclerosis and association of protein-disulfide isomerase with superoxide dismutase 1. J Biol Chem 281(40):30152–30165
Balch WE, Morimoto RI, Dillin A et al (2008) Adapting proteostasis for disease intervention. Science 319(5865):916–919
Baleriola J, Walker CA, Jean YY et al (2014) Axonally synthesized ATF4 transmits a neurodegenerative signal across brain regions. Cell 158(5):1159–1172
Ballard C, Gauthier S, Corbett A et al (2011) Alzheimer’s disease. Lancet 377(9770):1019–1031
Belal C, Ameli NJ, El Kommos A et al (2012) The homocysteine-inducible endoplasmic reticulum (ER) stress protein Herp counteracts mutant α-synuclein-induced ER stress via the homeostatic regulation of ER-resident calcium release channel proteins. Hum Mol Genet 21(5):963–977
Bellucci A, Navarria L, Zaltieri M et al (2011) Induction of the unfolded protein response by α-synuclein in experimental models of Parkinson’s disease. J Neurochem 116(4):588–605
Bence NF, Sampat RM, Kopito RR (2001) Impairment of the ubiquitin-proteasome system by protein aggregation. Science 292(5521):1552–1555
Bennett EJ, Shaler TA, Woodman B et al (2007) Global changes to the ubiquitin system in Huntington’s disease. Nature 448(7154):704–708
Berezovska O, Lleo A, Herl LD, Frosch MP et al (2005) Familial Alzheimer’s disease presenilin 1 mutations cause alterations in the conformation of presenilin and interactions with amyloid precursor protein. J Neurosci Off J Soc Neurosci 25(11):3009–3017
Bernard-Marissal N, Sunyach C, Marissal T, Raoul C et al (2015) Calreticulin levels determine onset of early muscle denervation by fast motoneurons of ALS model mice. Neurobiol Dis 73:130–136
Betarbet R, Sherer TB, MacKenzie G et al (2000) Chronic systemic pesticide exposure reproduces features of Parkinson’s disease. Nat Neurosci 3(12):1301–1306
Bouman L, Schlierf A, Lutz AK et al (2011) Parkin is transcriptionally regulated by ATF4: evidence for an interconnection between mitochondrial stress and ER stress. Cell Death Differ 18(5):769–782
Boyce M et al (2005) A selective inhibitor of eIF2α dephosphorylation protects cells from ER stress. Sci 307(5711):935–939
Brandt R, Hundelt M, Shahani N (2005) Tau alteration and neuronal degeneration in tauopathies: mechanisms and models. Biochem Biophys Acta 1739(2–3):331–354
Brown AR, Rebus S, McKimmie CS et al (2005) Gene expression profiling of the preclinical scrapie-infected hippocampus. Biochem Biophys Res Commun 334(1):86–95
Brown RH (1998) SOD1 aggregates in ALS: cause, correlate or consequence? Nat Med 4(12):1362–1364
Carnemolla A, Fossale E, Agostoni E et al (2009) Rrs1 is involved in endoplasmic reticulum stress response in Huntington disease. J Biol Chem 284(27):18167–18173
Casas-Tinto S, Zhang Y, Sanchez-Garcia J et al (2011) The ER stress factor XBP1s prevents amyloid-beta neurotoxicity. Hum Mol Genet 20(11):2144–2160
Castillo-Carranza DL, Zhang Y, Guerrero-Munoz MJ et al (2012) Differential activation of the ER stress factor XBP1 by oligomeric assemblies. Neurochem Res 37(8):1707–1717
Caughey B, Lansbury PT (2003) Protofibrils, pores, fibrils, and neurodegeneration: separating the responsible protein aggregates from the innocent bystanders. Annu Rev Neurosci 26:267–298
Cavedo E, Lista S, Khachaturian Z et al (2014) The road ahead to cure Alzheimer’s disease: development of biological markers and neuroimaging methods for prevention trials across all stages and target populations. J Prev Alzheimer’s Dis 1(3):181–202
Cho KJ, Lee BI, Cheon SY et al (2009) Inhibition of apoptosis signal-regulating kinase 1 reduces endoplasmic reticulum stress and nuclear huntingtin fragments in a mouse model of Huntington disease. Neuroscience 163(4):1128–1134
Chung CY, Khurana V, Auluck PK et al (2013) Identification and rescue of α-synuclein toxicity in Parkinson patient–derived neurons. Sci 342(6161):983–987
Cisse M, Duplan E, Lorivel T et al (2016) The transcription factor XBP1s restores hippocampal synaptic plasticity and memory by control of the Kalirin-7 pathway in Alzheimer model. Mol Psychiatry
Colla E, Jensen PH, Pletnikova O, Troncoso JC et al (2012a) Accumulation of toxic α-synuclein oligomer within endoplasmic reticulum occurs in α-synucleinopathy in vivo. J Neurosci: Off J Soc Neurosci 32(10):3301–3305
Colla E, Coune P, Liu Y et al (2012b) Endoplasmic reticulum stress is important for the manifestations of α-synucleinopathy in vivo. J Neurosci: Off J Soc Neurosci 32(10):3306–3320
Conn KJ, Gao W, McKee A et al (2004) Identification of the protein disulfide isomerase family member PDIp in experimental Parkinson’s disease and Lewy body pathology. Brain Res 1022(1–2):164–172
Cooper AA, Gitler AD, Cashikar A et al (2006) Alpha-synuclein blocks ER-Golgi traffic and Rab1 rescues neuron loss in Parkinson’s models. Sci (New York, N.Y.) 313(5785):324–328
Cornejo VH, Hetz C (2013) Seminars in immunopathology. In: The unfolded protein response in Alzheimers disease, pp 277–292
Cornejo VH, Pihán P, Vidal RL et al (2013) Role of the unfolded protein response in organ physiology: lessons from mouse models. IUBMB Life 65(12):962–975
Costa-Mattioli M, Sossin WS, Klann E et al (2009) Translational control of long-lasting synaptic plasticity and memory. Neuron 61(1):10–26
Coune PG, Bensadoun J-C, Aebischer P et al (2011) Rab1A over-expression prevents Golgi apparatus fragmentation and partially corrects motor deficits in an alpha-synuclein based rat model of Parkinson’s disease. J Parkinson’s Dis 1(4):373–387
Credle JJ, Forcelli PA, Delannoy M et al (2015) α-Synuclein-mediated inhibition of ATF6 processing into COPII vesicles disrupts UPR signaling in Parkinson’s disease. Neurobiol Dis 76:112–125
Crunkhorn S (2015) Neurodegenerative disease: phosphatase inhibitor prevents protein-misfolding diseases. Nat Rev Drug Discov 14(6):386
Culmsee C, Landshamer S (2006) Molecular insights into mechanisms of the cell death program: role in the progression of neurodegenerative disorders. Curr Alzheimer Res 3(4):269–283
Das I, Krzyzosiak A, Schneider K, Wrabetz L et al (2015) Preventing proteostasis diseases by selective inhibition of a phosphatase regulatory subunit. Science (New York, N.Y.) 348(6231):239–242
Deitch JS, Alexander GM, Bensinger A et al (2014) Phenotype of transgenic mice carrying a very low copy number of the mutant human G93A superoxide dismutase-1 gene associated with amyotrophic lateral sclerosis. PLoS ONE 9(6):e99879
Di Domenico F, Head E, Butterfield DA et al (2014) Oxidative stress and proteostasis network: culprit and casualty of Alzheimers-like neurodegeneration. Adv Geriatrics
Dion PA, Daoud H, Rouleau GA (2009) Genetics of motor neuron disorders: new insights into pathogenic mechanisms. Nat Rev Genet 10(11):769–782
Dovey HF, John V, Anderson JP et al (2001) Functional gamma-secretase inhibitors reduce beta-amyloid peptide levels in brain. J Neurochem 76(1):173–181
Duennwald ML, Lindquist S (2008) Impaired ERAD and ER stress are early and specific events in polyglutamine toxicity. Genes Dev 22(23):3308–3319
Dufey E, Sepúlveda D, Rojas-Rivera D et al (2014) Cellular mechanisms of endoplasmic reticulum stress signaling in health and disease. 1. An overview. Am J Physiol Cell Physiol 307(7):C582–C594
Duran-Aniotz C, Martínez G, Hetz C (2014) Memory loss in Alzheimer’s disease: are the alterations in the UPR network involved in the cognitive impairment? Front Aging Neurosci 6:8
Duran-Aniotz C et al (2017) IRE1 signaling exacerbates Alzheimer’s disease pathogenesis. Acta Neuropathol 1–18
Duvoisin RC (1995) Recent advances in the genetics of Parkinson’s disease. Adv Neurol 69:33–40
Egawa N, Yamamoto K, Inoue H et al (2011) The endoplasmic reticulum stress sensor, ATF6α, protects against neurotoxin-induced dopaminergic neuronal death. J Biol Chem 286(10):7947–7957
Endres K, Reinhardt S (2013) ER-stress in Alzheimer’s disease: turning the scale? Am J Neurodegener Dis 2(4):247–265
Fawcett EM, Hoyt JM, Johnson JK et al (2015) Hypoxia disrupts proteostasis in Caenorhabditis elegans. Aging Cell 14(1):92–101
Ferreiro E, Oliveira CR, Pereira CM (2008) The release of calcium from the endoplasmic reticulum induced by amyloid-beta and prion peptides activates the mitochondrial apoptotic pathway. Neurobiol Dis 30(3):331–342
Filézac de L’Etang A, Maharjan N, Braña C et al (2015) Marinesco-Sjögren syndrome protein SIL1 regulates motor neuron subtype-selective ER stress in ALS. Nat Neurosci 18(2):227–238
Fouillet A, Levet C, Virgone A, Robin M et al (2012) ER stress inhibits neuronal death by promoting autophagy. Autophagy 8(6):915–926
Freeman OJ, Mallucci GR (2016) The UPR and synaptic dysfunction in neurodegeneration. Brain Res
Gkogkas C, Middleton S, Kremer AM et al (2008) VAPB interacts with and modulates the activity of ATF6. Hum Mol Genet 17(11):1517–1526
Glenner GG, Wong CW (2012) Alzheimer’s disease: initial report of the purification and characterization of a novel cerebrovascular amyloid protein 1984. Biochem Biophys Res Commun 425(3):534–539
Gonzalez-Perez P, Woehlbier U, Chian RJ et al (2015) Identification of rare protein disulfide isomerase gene variants in amyotrophic lateral sclerosis patients. Gene 566(2):158–165
Gorbatyuk MS, Shabashvili A, Chen W et al (2012) Glucose regulated protein 78 diminishes α-synuclein neurotoxicity in a rat model of Parkinson disease. Mol Ther 20(7):1327–1337
Gunawardena S, Goldstein LS (2005) Polyglutamine diseases and transport problems: deadly traffic jams on neuronal highways. Arch Neurol 62(1):46–51
Haass C (2004) Take five–BACE and the gamma-secretase quartet conduct Alzheimer’s amyloid beta-peptide generation. EMBO J 23(3):483–488
Halliday M, Radford H, Sekine Y et al (2015) Partial restoration of protein synthesis rates by the small molecule ISRIB prevents neurodegeneration without pancreatic toxicity. Cell Death Dis 6:e1672
Hamos JE, Oblas B, Pulaski-Salo D et al (1991) Expression of heat shock proteins in Alzheimer’s disease. Neurol 41(3):345
Hashida K, Kitao Y, Sudo H et al (2012) ATF6alpha promotes astroglial activation and neuronal survival in a chronic mouse model of Parkinsons disease. PLoS ONE 7(10):e47950
Henstridge CM, Pickett E, Spires-Jones TL (2016) Synaptic pathology: a shared mechanism in neurological disease. Ageing Res Rev 28:72–84
Herms JW, Korte S, Gall S et al (2000) Altered intracellular calcium homeostasis in cerebellar granule cells of prion protein-deficient mice. J Neurochem 75(4):1487–1492
Hetz C (2012) The unfolded protein response: controlling cell fate decisions under ER stress and beyond. Nat Rev Mol Cell Biol 13(2):89–102
Hetz CA, Soto C (2006) Stressing out the ER: a role of the unfolded protein response in prion-related disorders. Curr Mol Med 6(1):37–43
Hetz Flores C, Mollereau B (2014) Disturbance of endoplasmic reticulum proteostasis in neurodegenerative diseases. Nat Rev Neurosci 15(4):233
Hetz C, Russelakis-Carneiro M, Maundrell K, Castilla J et al (2003) Caspase-12 and endoplasmic reticulum stress mediate neurotoxicity of pathological prion protein. EMBO J 22(20):5435–5445
Hetz C, Russelakis-Carneiro M, Wälchli S et al (2005a) The disulfide isomerase Grp58 is a protective factor against prion neurotoxicity. J Neurosci 25(11):2793–2802
Hetz C, Lee A-H, Gonzalez-Romero D, Thielen P et al (2008) Unfolded protein response transcription factor XBP-1 does not influence prion replication or pathogenesis. Proc Natl Acad Sci 105(2):757–762
Hetz C, Thielen P, Matus S et al (2009) XBP-1 deficiency in the nervous system protects against amyotrophic lateral sclerosis by increasing autophagy. Genes Dev 23(19):2294–2306
Hetz C, Chevet E, Harding HP (2013) Targeting the unfolded protein response in disease. Nat Rev Drug Discov 12(9):703–719
Hetz C, Russelakis-Carneiro M, Walchli S et al (2005) The disulfide isomerase Grp58 is a protective factor against prion neurotoxicity. J Neurosci 25:2793–2802
Hippius H, Neundörfer G (2003) The discovery of Alzheimer’s disease. Dialogues Clin Neurosci 5(1):101–108
Ho YS, Yang X, Lau JC et al (2012) Endoplasmic reticulum stress induces tau pathology and forms a vicious cycle: implication in Alzheimer’s disease pathogenesis. J Alzheimer’s Dis: JAD 28(4):839–854
Honjo Y, Ito H, Horibe T, Takahashi R et al (2010) Protein disulfide isomerase-immunopositive inclusions in patients with Alzheimer disease. Brain Res 1349:90–96
Honjo Y, Kaneko S, Ito H et al (2011) Protein disulfide isomerase-immunopositive inclusions in patients with amyotrophic lateral sclerosis. Amyotroph Later Scler Off Publ World Fed Neurol Res Gr Mot Neuron Dis 12(6):444–450
Hoozemans JJM, Veerhuis R, Van Haastert ES et al (2005) The unfolded protein response is activated in Alzheimers disease. Acta Neuropathol 110(2):165–172
Hoozemans JJM, Van Haastert ES, Eikelenboom P et al (2007) Activation of the unfolded protein response in Parkinsons disease. Biochem Biophys Res Commun 354(3):707–711
Hoozemans JJ, van Haastert ES, Nijholt DA et al (2009) The unfolded protein response is activated in pretangle neurons in Alzheimer’s disease hippocampus. Am J Pathol 174(4):1241–1251
Hoozemans JJ, van Haastert ES, Nijholt DA et al (2012) Activation of the unfolded protein response is an early event in Alzheimer’s and Parkinson’s disease. Neuro-Degener Dis 10(1–4):212–215
Hu B-R, Janelidze S, Ginsberg MD et al (2001) Protein aggregation after focal brain ischemia and reperfusion. J Cereb Blood Flow Metab 21(7):865–875
Ito Y, Yamada M, Tanaka H et al (2009) Involvement of CHOP, an ER-stress apoptotic mediator, in both human sporadic ALS and ALS model mice. Neurobiol Dis 36(3):470–476
Jankovic J, Aguilar LG (2008) Current approaches to the treatment of Parkinsons disease. Neuropsychiatry Dis Treat 4(4):743–757
Jiang Y, Chadwick SR, Lajoie P (2016) Endoplasmic reticulum stress: the cause and solution to Huntington’s disease? Brain Res
Jiang HQ, Ren M, Jiang HZ et al (2014) Guanabenz delays the onset of disease symptoms, extends lifespan, improves motor performance and attenuates motor neuron loss in the SOD1 G93A mouse model of amyotrophic lateral sclerosis. Neuroscience 277:132–138
Jiang P, Gan M, Ebrahim AS et al (2010) ER stress response plays an important role in aggregation of α-synuclein. Mol Neurodegener 5:56
Kalathur RK, Giner-Lamia J, Machado S et al (2015) The unfolded protein response and its potential role in Huntington’s disease elucidated by a systems biology approach. F1000Res 4:103
Katayama T, Imaizumi K, Manabe T et al (2004) Induction of neuronal death by ER stress in Alzheimer’s disease. J Chem Neuroanat 28(1–2):67–78
Katayama T, Imaizumi K, Sato N et al (1999) Presenilin-1 mutations downregulate the signalling pathway of the unfolded-protein response. Nat Cell Biol 1(8):479–485
Kaushik S, Cuervo AM (2015) Proteostasis and aging. Nat Med 21(12):1406–1415
Kern A, Behl C (2009) The unsolved relationship of brain aging and late-onset Alzheimer disease. Biochem Biophys Acta 1790(10):1124–1132
Kikuchi H, Almer G, Yamashita S et al (2006) Spinal cord endoplasmic reticulum stress associated with a microsomal accumulation of mutant superoxide dismutase-1 in an ALS model. Proc Natl Acad Sci 103(15):6025–6030
Kim HJ, Raphael AR, LaDow ES et al (2014) Therapeutic modulation of eIF2α phosphorylation rescues TDP-43 toxicity in amyotrophic lateral sclerosis disease models. Nat Genet 46(2):152–160
Kitao Y, Imai Y, Ozawa K et al (2007) Pael receptor induces death of dopaminergic neurons in the substantia nigra via endoplasmic reticulum stress and dopamine toxicity, which is enhanced under condition of parkin inactivation. Hum Mol Genet 16(1):50–60
Kouroku Y, Fujita E, Jimbo A et al (2002) Polyglutamine aggregates stimulate ER stress signals and caspase-12 activation. Hum Mol Genet 11(13):1505–1515
Kouroku Y, Fujita E, Tanida I et al (2007) ER stress (PERK/eIF2alpha phosphorylation) mediates the polyglutamine-induced LC3 conversion, an essential step for autophagy formation. Cell Death Differ 14(2):230–239
Kwok CT, Morris AG, Frampton J et al (2013) Association studies indicate that protein disulfide isomerase is a risk factor in amyotrophic lateral sclerosis. Free Radic Biol Med 58:81–86
Labbadia J, Morimoto RI (2014) Proteostasis and longevity: when does aging really begin? F1000Prime Rep 6:7
De Lau LM, Breteler MM (2006) Epidemiology of Parkinson’s disease. Lancet Neurol 5(6):525–535
Lautenschlaeger J, Prell T, Grosskreutz J (2012) Endoplasmic reticulum stress and the ER mitochondria calcium cycle in amyotrophic lateral sclerosis. Amyotroph Later Scler 13(2):166–177
Ledesma MD, Galvan C, Hellias B et al (2002) Astrocytic but not neuronal increased expression and redistribution of parkin during unfolded protein stress. J Neurochem 83(6):1431–1440
Lee H-J, Patel S, Lee S-J (2005) Intravesicular localization and exocytosis of α-synuclein and its aggregates. J Neurosci 25(25):6016–6024
Lee DY, Lee KS, Lee HJ et al (2010a) Activation of PERK signaling attenuates Abeta-mediated ER stress. PLoS ONE 5(5):e10489
Lee JH, Won SM et al (2010b) Induction of the unfolded protein response and cell death pathway in Alzheimer’s disease, but not in aged Tg2576 mice. Exp Mol Med 42(5):386–394
Lee H, Noh JY, Oh Y et al (2012) IRE1 plays an essential role in ER stress-mediated aggregation of mutant huntingtin via the inhibition of autophagy flux. Hum Mol Genet 21(1):101–114
Liu SY, Wang W, Cai ZY et al (2013) Polymorphism -116C/G of human X-box-binding protein 1 promoter is associated with risk of Alzheimer’s disease. CNS Neurosci Ther 19(4):229–234
Loewen CA, Feany MB (2010) The unfolded protein response protects from tau neurotoxicity in vivo. PloS One 5(9)
Lourenco MV, Ferreira ST, De Felice FG (2015) Neuronal stress signaling and eIF2α phosphorylation as molecular links between Alzheimer’s disease and diabetes. Prog Neurobiol 129:37–57
Luo Y, Bolon B, Kahn S et al (2001) Mice deficient in BACE1, the Alzheimer’s beta-secretase, have normal phenotype and abolished beta-amyloid generation. Nat Neurosci 4(3):231–232
Ma T, Trinh MA, Wexler AJ et al (2013) Suppression of eIF2α kinases alleviates Alzheimer’s disease-related plasticity and memory deficits. Nat Neurosci 16(9):1299–1305
Maekawa S, Leigh PN, King A et al (2009) TDP-43 is consistently co-localized with ubiquitinated inclusions in sporadic and Guam amyotrophic lateral sclerosis but not in familial amyotrophic lateral sclerosis with and without SOD1 mutations. Neuropathology 29(6):672–683
Maharjan N, Saxena S (2016) ER strikes again: proteostasis dysfunction in ALS. EMBO J 35(8):798–800
Maly DJ, Papa FR (2014) Druggable sensors of the unfolded protein response. Nat Chem Biol 10(11):892–901
Martínez G, Vidal RL, Mardones P et al (2016) Regulation of memory formation by the transcription factor XBP1. Cell Rep 14(6):1382–1394
Martinez-Vicente M, Talloczy Z, Wong E et al (2010) Cargo recognition failure is responsible for inefficient autophagy in Huntington’s disease. Nat Neurosci 13(5):567–576
Masters CL, Multhaup G, Simms G et al (1985) Neuronal origin of a cerebral amyloid: neurofibrillary tangles of Alzheimer’s disease contain the same protein as the amyloid of plaque cores and blood vessels. EMBO J 4(11):2757
Matus S, Castillo K, Hetz C (2012) Hormesis: protecting neurons against cellular stress in Parkinson disease. Autophagy 8(6):997–1001
Matus S, Lopez E, Valenzuela V, Nassif M et al (2013) Functional contribution of the transcription factor ATF4 to the pathogenesis of amyotrophic lateral sclerosis. PLoS ONE 8(7):e66672
Mays CE, Soto C (2016) The stress of prion disease. Brain Res
McGeer PL, McGeer EG (2013) The amyloid cascade-inflammatory hypothesis of Alzheimer disease: implications for therapy. Acta Neuropathol 126(4):479–497
Mercado G, Valdés P, Hetz C (2013) An ERcentric view of Parkinson’s disease. Trends Mol Med 19(3):165–175
Mercado G, Castillo V, Soto P et al (2016) ER stress and Parkinson’s disease: pathological inputs that converge into the secretory pathway. Brain Res 1648(Pt B):626–632
Mitsuda T, Hayakawa Y, Itoh M et al (2007) ATF4 regulates γ-secretase activity during amino acid imbalance. Biochem Biophys Res Commun 352(3):722–727
Moreno JA, Radford H, Peretti D et al (2012) Sustained translational repression by eIF2α-P mediates prion neurodegeneration. Nat 485(7399):507–511
Moreno JA, Halliday M, Molloy C et al (2013) Oral treatment targeting the unfolded protein response prevents neurodegeneration and clinical disease in prion-infected mice. Sci Transl Med 5(206):206ra138
Mukherjee A, Morales-Scheihing D, Gonzalez-Romero D et al (2010) Calcineurin inhibition at the clinical phase of prion disease reduces neurodegeneration, improves behavioral alterations and increases animal survival. PLoS Pathog 6(10):e1001138
Nagata T, Ilieva H, Murakami T et al (2007) Increased ER stress during motor neuron degeneration in a transgenic mouse model of amyotrophic lateral sclerosis. Neurol Res 29(8):767–771
Nishimura AL, Mitne-Neto M, Silva HC, Richieri-Costa A et al (2004) A mutation in the vesicle-trafficking protein VAPB causes late-onset spinal muscular atrophy and amyotrophic lateral sclerosis. Am J Hum Genet 75(5):822–831
Nishitoh H, Matsuzawa A, Tobiume K et al (2002) ASK1 is essential for endoplasmic reticulum stress-induced neuronal cell death triggered by expanded polyglutamine repeats. Genes Dev 16(11):1345–1355
Nishitoh H, Kadowaki H, Nagai A et al (2008) ALS-linked mutant SOD1 induces ER stress-and ASK1-dependent motor neuron death by targeting Derlin-1. Genes Dev 22(11):1451–1464
Oakley H, Cole SL, Logan S, Maus E et al (2006) Intraneuronal beta-amyloid aggregates, neurodegeneration, and neuron loss in transgenic mice with five familial Alzheimer’s disease mutations: potential factors in amyloid plaque formation. J Neurosci Off J Soc Neurosci 26(40):10129–10140
Oakes SA, Papa FR (2015) The role of endoplasmic reticulum stress in human pathology. Annu Rev Pathol 10:173–194
Orr HT, Zoghbi HY (2007) Trinucleotide repeat disorders. Annu Rev Neurosci 30:575–621
Page G, Rioux Bilan A, Ingrand S et al (2006) Activated double-stranded RNA-dependent protein kinase and neuronal death in models of Alzheimer’s disease. Neurosci 139(4):1343–1354
Pasini S, Corona C, Liu J et al (2015) Specific downregulation of hippocampal ATF4 reveals a necessary role in synaptic plasticity and memory. Cell Rep 11(2):183–191
Powers ET, Balch WE (2013) Diversity in the origins of proteostasis networks–a driver for protein function in evolution. Nat Rev Mol Cell Biol 14(4):237–248
Prudencio M, Belzil VV, Batra R, Ross CA et al (2015) Distinct brain transcriptome profiles in C9orf72-associated and sporadic ALS. Nat Neurosci 18(8):1175–1182
Prusiner SB (1998) Prions. Proc Natl Acad Sci 95(23):13363–13383
Prusiner SB, Scott MR (1997) Genetics of prions. Annu Rev Genet 31:139–175
Reitz C (2012) Alzheimer’s disease and the amyloid cascade hypothesis: a critical review. Int J Alzheimers Dis 2012
Rozas P, Bargsted L, Martínez F et al (2016) The ER proteostasis network in ALS: determining the differential motoneuron vulnerability. Neurosci Lett
Ross CA, Poirier MA (2005) What is the role of protein aggregation in neurodegeneration? Nat Rev Mol Cell Biol 6(11):891–898
Ryu EJ, Harding HP, Angelastro JM et al (2002) Endoplasmic reticulum stress and the unfolded protein response in cellular models of Parkinson’s disease. J Neurosci 22(24):10690–10698
Sado M, Yamasaki Y, Iwanaga T et al (2009) Protective effect against Parkinson’s disease-related insults through the activation of XBP1. Brain Res 1257:16–24
Salminen A, Kauppinen A, Suuronen T et al (2009) ER stress in Alzheimer’s disease: a novel neuronal trigger for inflammation and Alzheimer’s pathology. J Neuroinflamm 6:41
Sato N, Urano F, Yoon Leem J et al (2000) Upregulation of BiP and CHOP by the unfolded-protein response is independent of presenilin expression. Nat Cell Biol 2(12):863–870
Saxena S, Caroni P (2011) Selective neuronal vulnerability in neurodegenerative diseases: from stressor thresholds to degeneration. Neuron 71(1):35–48
Saxena S, Cabuy E, Caroni P (2009) A role for motoneuron subtype-selective ER stress in disease manifestations of FALS mice. Nat Neurosci 12(5):627–636
Sämann J, Hegermann J, von Gromoff E et al (2009) Caenorhabditits elegans LRK-1 and PINK-1 act antagonistically in stress response and neurite outgrowth. J Biol Chem 284(24):16482–16491
Scheff SW, Ansari MA, Mufson EJ (2016) Oxidative stress and hippocampal synaptic protein levels in elderly cognitively intact individuals with Alzheimer’s disease pathology. Neurobiol Aging 42:1–12
Scheper W, Hoozemans JJ (2015) The unfolded protein response in neurodegenerative diseases: a neuropathological perspective. Acta Neuropathol 130(3):315–331
Si L, Xu T, Wang F, Liu Q (2012) X-box-binding protein 1-modified neural stem cells for treatment of Parkinson’s disease. Neural Regener Res 7(10):736–740
Silva RM, Ries V, Oo TF, Yarygina O et al (2005) CHOP/GADD153 is a mediator of apoptotic death in substantia nigra dopamine neurons in an in vivo neurotoxin model of parkinsonism. J Neurochem 95(4):974–986
Slodzinski H, Moran LB, Michael GJ et al (2009) Homocysteine-induced endoplasmic reticulum protein (herp) is up-regulated in parkinsonian substantia nigra and present in the core of Lewy bodies. Clin Neuropathol 28(5):333–343
Smith HL, Mallucci GR (2016) The unfolded protein response: mechanisms and therapy of neurodegeneration. Brain J Neurol 139(Pt 8):2113–2121
Soto C (2003) Unfolding the role of protein misfolding in neurodegenerative diseases. Nat Rev Neurosci 4(1):49–60
Sreedharan J, Brown RH (2013) Amyotrophic lateral sclerosis: problems and prospects. Ann Neurol 74(3):309–316
Steele AD, Hetz C, Yi CH et al (2007) Prion pathogenesis is independent of caspase-12. Prion 1(4):243–247
De Strooper B, Karran E (2016) The cellular phase of Alzheimers disease. Cell 164(4):603–615
Sun S, Sun Y, Ling SC et al (2015) Translational profiling identifies a cascade of damage initiated in motor neurons and spreading to glia in mutant SOD1-mediated ALS. Proc Natl Acad Sci USA 112(50):E6993–E7002
Suzuki H, Matsuoka M (2012) TDP-43 toxicity is mediated by the unfolded protein response-unrelated induction of C/EBP homologous protein expression. J Neurosci Res 90(3):641–647
Suzuki H, Kanekura K, Levine TP et al (2009) ALS-linked P56S-VAPB, an aggregated loss-of-function mutant of VAPB, predisposes motor neurons to ER stress-related death by inducing aggregation of co-expressed wild-type VAPB. J Neurochem 108(4):973–985
Tobisawa S, Hozumi Y, Arawaka S et al (2003) Mutant SOD1 linked to familial amyotrophic lateral sclerosis, but not wild-type SOD1, induces ER stress in COS7 cells and transgenic mice. Biochem Biophys Res Commun 303(2):496–503
Tompkins MM, Hill WD (1997) Contribution of somal Lewy bodies to neuronal death. Brain Res 775(1–2):24–29
Torres M, Castillo K, Armisén R et al (2010) Prion protein misfolding affects calcium homeostasis and sensitizes cells to endoplasmic reticulum stress. PLoS ONE 5(12):e15658
Torres M, Medinas DB, Matamala JM et al (2015) The protein-disulfide isomerase ERp57 regulates the steady-state levels of the prion protein. J Biol Chem 290(39):23631–23645
Tsaytler P et al (2011) Selective inhibition of a regulatory subunit of protein phosphatase 1 restores proteostasis. Sci 332(6025):91–94
Unterberger U, Höftberger R, Gelpi E et al (2006) Endoplasmic reticulum stress features are prominent in Alzheimer disease but not in prion diseases in vivo. J Neuropathol Exp Neurol 65(4):348–357
Urano F, Wang X, Bertolotti A et al (2000) Coupling of stress in the ER to activation of JNK protein kinases by transmembrane protein kinase IRE1. Sci 287(5453):664–666
Valdés P, Mercado G, Vidal RL et al (2014) Control of dopaminergic neuron survival by the unfolded protein response transcription factor XBP1. Proc Natl Acad Sci USA 111(18):6804–6809
Valenzuela V, Martínez G, Duran-Aniotz C et al (2016) Gene therapy to target ER stress in brain diseases. Brain Res
van der Harg JM, Nölle A, Zwart R et al (2014) The unfolded protein response mediates reversible tau phosphorylation induced by metabolic stress. Cell Death Dis 5:e1393
Varma D, Sen D (2015) Role of the unfolded protein response in the pathogenesis of Parkinson’s disease. Acta Neurobiol Exp 75(1):1–26
Vassar R (2009) Phosphorylation of the translation initiation factor eIF2alpha increases BACE1 levels and promotes amyloidogenesis. Alzheimer’s Dementia 5(4):P81–P82
Vidal R, Caballero B, Couve A, Hetz C (2011) Converging pathways in the occurrence of endoplasmic reticulum (ER) stress in Huntington’s disease. Curr Mol Med 11(1):1–12
Vidal RL, Figueroa A, Court FA et al (2012) Targeting the UPR transcription factor XBP1 protects against Huntington’s disease through the regulation of FoxO1 and autophagy. Hum Mol Genet 21(10):2245–2262
Vieira FG, Ping Q, Moreno AJ et al (2015) Guanabenz treatment accelerates disease in a mutant SOD1 mouse model of ALS. PLoS ONE 10(8):e0135570
Vitte J, Traver S, De Paula M et al (2010) Leucine-rich repeat kinase 2 is associated with the endoplasmic reticulum in dopaminergic neurons and accumulates in the core of Lewy bodies in Parkinson disease. J Neuropathol Exp Neurol 69(9):959–972
Vossel KA, Zhang K, Brodbeck J et al (2010) Tau reduction prevents Abeta-induced defects in axonal transport. Sci (New York, N.Y.) 330(6001):198
Walker FO (2007) Huntington’s disease. Lancet 369(9557):218–228
Walker L, McAleese KE, Thomas AJ et al (2015) Neuropathologically mixed Alzheimer’s and Lewy body disease: burden of pathological protein aggregates differs between clinical phenotypes. Acta Neuropathol 129(5):729–748
Walter P, Ron D (2011) The unfolded protein response: from stress pathway to homeostatic regulation. Sci 334(6059):1081–1086
Wang M, Kaufman RJ (2016) Protein misfolding in the endoplasmic reticulum as a conduit to human disease. Nat 529(7586):326–335
Wang L, Popko B, Roos RP (2011) The unfolded protein response in familial amyotrophic lateral sclerosis. Hum Mol Genet 20(5):1008–1015
Wang M, Ye R, Barron E et al (2010) Essential role of the unfolded protein response regulator GRP78/BiP in protection from neuronal apoptosis. Cell Death Differ 17(3):488–498
Wang L, Popko B, Tixier E et al (2014) Guanabenz, which enhances the unfolded protein response, ameliorates mutant SOD1-induced amyotrophic lateral sclerosis. Neurobiol Dis 71:317–324
Wate R, Ito H, Zhang JH et al (2005) Expression of an endoplasmic reticulum-resident chaperone, glucose-regulated stress protein 78, in the spinal cord of a mouse model of amyotrophic lateral sclerosis. Acta Neuropathol 110(6):557–562
Woehlbier U, Colombo A, Saaranen MJ et al (2016) ALS-linked protein disulfide isomerase variants cause motor dysfunction. EMBO J 35(8):845–865
Yoo BC, Krapfenbauer K, Cairns N et al (2002) Overexpressed protein disulfide isomerase in brains of patients with sporadic Creutzfeldt-Jakob disease. Neurosci Lett 334(3):196–200
Yoon SO, Park DJ, Ryu JC et al (2012) JNK3 perpetuates metabolic stress induced by Aβ peptides. Neuron 75(5):824–837
Yuan Y, Cao P, Smith MA et al (2011) Dysregulated LRRK2 signaling in response to endoplasmic reticulum stress leads to dopaminergic neuron degeneration in C. elegans. PLoS ONE 6(8):e22354
Zhang YJ, Jansen-West K, Xu YF et al (2014) Aggregation-prone c9FTD/ALS poly(GA) RAN-translated proteins cause neurotoxicity by inducing ER stress. Acta Neuropathol 128(4):505
Zuleta A, Vidal RL, Armentano D et al (2012) AAV-mediated delivery of the transcription factor XBP1s into the striatum reduces mutant Huntingtin aggregation in a mouse model of Huntingtons disease. Biochem Biophys Res Commun 420(3):558–563
Acknowledgements
Supported by Millennium Institute No. P09-015-F, FONDAP program 15150012, CONICYT-Brazil 441921/2016-7, ALS Therapy Alliance 2014-F-059, Muscular Dystrophy Association 382453, Michael J. Fox Foundation for Parkinson’s Research—Target Validation grant No 9277, FONDECYT No. 1140549, Office of Naval Research-Global (ONR-G) N62909-16-1-2003, FONDEF D11E1007, U.S. Air Force Office of Scientific Research FA9550-16-1-0384, FONDEF ID16I10223, and ALSRP Therapeutic Idea Award AL150111 (CH). FC is a postdoctoral fellow funded by FONDAP program 15150012.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2017 Springer International Publishing AG
About this chapter
Cite this chapter
Cabral-Miranda, F., Hetz, C. (2017). ER Stress and Neurodegenerative Disease: A Cause or Effect Relationship?. In: Wiseman, R., Haynes, C. (eds) Coordinating Organismal Physiology Through the Unfolded Protein Response. Current Topics in Microbiology and Immunology, vol 414. Springer, Cham. https://doi.org/10.1007/82_2017_52
Download citation
DOI: https://doi.org/10.1007/82_2017_52
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-78529-5
Online ISBN: 978-3-319-78530-1
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)