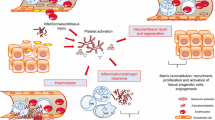
Abstract
The pathophysiologic course of sepsis involves the release of cyto- and chemokines in addition to the activation of endothelial and neutrophil cells, initiating a cascade of cell-surface interactions. Activation of the coagulation system has been characterized by widespread intravascular fibrin deposition and platelet aggregation (disseminated intravascular coagulation, DIC) with subsequent microvascular and tissue injury, ultimately leading to multiple organ failure (MOF) and death. The contributing role of platelets in the pathophysiology of sepsis and related organ dysfunction is not entirely clear, although the degree and duration of thrombocytopenia, as well as the net change in the platelet count, are important determinants for survival [1, 2]. Of note, the involvement of platelets in sepsis-associated coagulopathy was already studied by many groups more than 25 years ago, focusing on the interaction of platelets with endotoxin, and the role of thrombin generation and DIC on platelet function [3, 4]. However, regarding the various pathogenic mechanisms that have recently been implicated in the activation of coagulation in sepsis, a reassessment of the role of platelets is needed.
Chapter PDF
Similar content being viewed by others
Keywords
- Severe Sepsis
- Fresh Freeze Plasma
- Thrombotic Thrombocytopenic Purpura
- Granulocyte Elastase
- Congenital Thrombotic Thrombocytopenic Purpura
These keywords were added by machine and not by the authors. This process is experimental and the keywords may be updated as the learning algorithm improves.
References
Vanderschueren S, De Weerdt A, Malbrain M, et al (2000) Thrombocytopenia and prognosis in intensive care. Crit Care Med 28:1871–1876
Martinez MA, Pena JM, Fernandez A, et al (1999) Time course and prognostic significance of hemostatic changes in sepsis: relation to tumor necrosis factor-alpha. Crit Care Med 27:1303–1308
Levin J, Poore TE, Young NS, et al (1972) Gram-negative sepsis: detection of endotoxemia with the limulus test. With studies of associated changes in blood coagulation, serum lipids, and complement. Ann Intern Med 76:1–7
Evans G, Lewis AF, Mustard JF (1969) The role of platelet aggregation in the development of endotoxin shock. Br J Surg 56:624
Neame PB, Kelton JG, Walker IR, Stewart IO, Nossel HL, Hirsh J (1980) Thrombocytopenia in septicemia: the role of disseminated intravascular coagulation. Blood 56:88–92
Taylor FB Jr, Chang AC, Mather T, et al (1991) DEGR-Factor Xa blocks disseminated intravascular coagulation initiated by E. coli without preventing shock or organ damage. Blood 78:364–371
Taylor FB Jr, Kinasewitz GT (2004) Activated protein C in sepsis. J Thromb Haemost 2:708–717
Levi M (2004) Platelets at a crossroad of pathogenic pathways in sepsis. J Thromb Haemost 2:2094–2095
Wagner DD, Burger PC (2003) Platelets in inflammation and thrombosis. Arterioscler Thromb Vasc Biol 23:2131–2137
Yaguchi A, Lobo FLM, Vincent, J-L, et al (2004) Platelet function in sepsis. J Thromb Haemost 2:2096–2102
Mannucci PM (2004) Treatment of von Willebrand’s Disease. N Engl J Med 351:683–694
Kayal S, Jais JP, Aguini N, Chaudiere J, Labrousse J (1998) Elevated circulating E-selectin, intercellular adhesion molecule 1, and von Willebrand factor in patients with severe infection. Am J Respir Crit Care Med 157:776–784
McGill SN, Ahmed NA, Christou NV (1998) Increased plasma von Willebrand factor in the systemic inflammatory response syndrome is derived from generalized endothelial cell activation. Crit Care Med 26:296–300
Moake JL (2002) Thrombotic microangiopathies. N Engl J Med 347:589–600
Furlan M, Robles R, Galbusera M, et al (1998) Von Willebrand factor-cleaving protease in thrombotic thrombocytopenic purpura and the hemolytic-uremic syndrome. N Engl J Med 339:1578–1584
Nguyen T, Hall M, Han Y, et al (2001) Microvascular thrombosis in pediatric multiple organ failure: Is it a therapeutic target? Pediatr Crit Care Med 2:187–196
Nguyen TC, Hall MW, Han YY, Seidberg N, Carcillo JA (2001) Randomized controlled trial of plasma exchange therapy for thrombocytopenia associated multiple organ failure in children. Pediatr Res 49:42A (abst)
Bianchi V, Robles R, Alberio L, Furlan M, Lämmle B (2002) Von Willebrand factor-cleaving protease (ADAMTS-13) in thrombocytopenic disorders: a severely deficient activity is specific for thrombotic thrombocytopenic purpura. Blood 100:710–713
Loof AH, van Vliet HH, Kappers-Klunne MC (2001) Low activity of von Willebrand factor-cleaving protease is not restricted to patients suffering from thrombotic thrombocytopenic purpura. Br J Haematol 112:1087–1088
Mannucci PM, Canciani MT, Forza I, Lussana F, Lattuada A, Rossi E (2001) Changes in health and disease of the metalloprotease that cleaves von Willebrand factor. Blood 98:2730–2735
Arya M, Anvari B, Romo GM, et al (2002) Ultralarge multimers of von Willebrand factor form spontaneous high-strength bonds with the platelet glycoprotein Ib-IX complex: studies using optical tweezers. Blood 99:3971–3977
Claus RA, Bockmeyer CL, Kentouche K, Reinhart K, Lösche W, Brunkhorst FM (2004) Decreased proteolytic activity of von Willebrand Factor cleaving protease ADAMTS-13 in survivors and non-survivors with septic shock. Platelets 15:486
Ono T, Mimuro J, Madoiwa S, et al (2005) Severe secondary deficiency of von Willebrand Factor-Cleaving Protease (ADAMTS-13) in patients with sepsis-induced disseminated intra-vascular coagulation: its correlation to development of renal failure. Blood Sep 27 (Epub ahead of print)
Borchiellini A, Fijnvandraat K, ten Cate JW, et al (1996) Quantitative analysis of von Willebrand factor propeptide release in vivo: effect of experimental endotoxemia and administration of 1-deamino-8-D-arginine vasopressin in humans. Blood 88:2951–2958
Sadler JE (1998) Biochemistry and genetics of von Willebrand factor. Annu Rev Biochem 67:395–424
Ruggeri ZM (2003) Von Willebrand factor, platelets and endothelial cell interactions. J Thromb Haemost 1:1335–1342
Furlan M, Robles R, Affolter D, Meyer D, Baillod P, Lammle B (1993) Triplet structure of von Willebrand factor reflects proteolytic degradation of high molecular weight multimers. Proc Natl Acad Sci USA 90:7503–7507
Xie L, Chesterman CN, Hogg PJ (2001) Control of von Willebrand factor multimer size by thrombospondin-1. J Exp Med 193:1341–1349
Soejima K, Nakagaki T (2005) Interplay between ADAMTS-13 and von Willebrand factor in inherited and acquired thrombotic microangiopathies. Semin Hematol 42:56–62
Ware LB, Eisner MD, Thompson BT, Parsons PE, Matthay MA (2004) Significance of von Willebrand factor in septic and nonseptic patients with acute lung injury. Am J Respir Crit Care Med 170:766–772
Ware LB, Conner ER, Matthay MA (2001) von Willebrand factor antigen is an independent marker of poor outcome in patients with early acute lung injury. Crit Care Med 29:2325–2331
van Mourik JA, Romani de Wit T (2001) Von Willebrand factor propeptide in vascular disorders. Thromb Haemost 86:164–171
Romani De Wit T, Fijnheer R, Brinkman HJ, Kersting S, Hene RJ, van Mourik JA (2003) Endothelial cell activation in thrombotic thrombocytopenic purpura (TTP): a prospective analysis. Br J Haematol 123:522–527
de Wit TR, van Mourik JA (2001) Biosynthesis, processing and secretion of von Willebrand factor: biological implications. Best Pract Res Clin Haematol 14:241–250
Levy GG, Nichols WC, Lian EC, et al (2001) Mutations in a member of the ADAMTS gene family cause thrombotic thrombocytopenic purpura. Nature 413:488–494
Kokame K, Matsumoto M, Soejima K, et al (2002) Mutations and common polymorphisms in ADAMTS-13 gene responsible for von Willebrand factor-cleaving protease activity. Proc Natl Acad Sci USA 99:11902–11907
Schneppenheim R, Budde U, Oyen F, et al (2003) von Willebrand factor cleaving protease and ADAMTS-13 mutations in childhood TTP. Blood 101:1845–1850
Plaimauer B, Zimmermann K, Volkel D, et al (2002) Cloning, expression, and functional characterization of the von Willebrand factor-cleaving protease (ADAMTS-13). Blood 100: 3626–3632
Zheng X, Chung D, Takayama TK, Majerus EM, Sadler JE, Fujikawa K (2001) Structure of von Willebrand factor-cleaving protease (ADAMTS-13), a metalloprotease involved in thrombotic thrombocytopenic purpura. J Biol Chem 276:41059–41063
Majerus EM, Zheng X, Tuley EA, Sadler JE (2003) Cleavage of the ADAMTS-13 propeptide is not required for protease activity. J Biol Chem 278:46643–46648
Soejima K, Matsumoto M, Kokame K, et al (2003) ADAMTS-13 cysteine-rich/spacer domains are functionally essential for von Willebrand factor cleavage. Blood 102:3232–3237
Tao Z, Wang Y, Choi H, et al (2005) Cleavage of ultralarge multimers of von Willebrand factor by C-terminal-truncated mutants of ADAMTS-13 under flow. Blood 106:141–143
Uemura M, Tatsumi K, Matsumoto M, et al (2005) Localization of ADAMTS-13 to the stellate cells of human liver. Blood 106:922–924
Zhou W, Inada M, Lee TP, et al (2005) ADAMTS-13 is expressed in hepatic stellate cells. Lab Invest 85:780–788
Claus RA, Bockmeyer CL, Kentouche K, et al (2005) Transcriptional regulation of ADAMTS-13. Thromb Haemost 94:41–45
Suzuki M, Murata M, Matsubara Y, et al (2004) Detection of von Willebrand factor-cleaving protease (ADAMTS-13) in human platelets. Biochem Biophys Res Commun 313:212–216
Liu L, Choi H, Bernardo A, et al (2005) Platelet-derived VWF-cleaving metalloprotease ADAMTS-13. J Thromb Haemost 3:2536–2544
Gerritsen HE, Robles R, Lammle B, Furlan M (2001) Partial amino acid sequence of purified von Willebrand factor-cleaving protease. Blood 98:1654–1661
Zheng X, Nishio K, Majerus EM, Sadler JE (2003) Cleavage of von Willebrand factor requires the spacer domain of the metalloprotease ADAMTS-13. J Biol Chem 278:30136–30141
Furlan M, Robles R, Morselli B, Sandoz P, Lammle B (1999) Recovery and half-life of von Willebrand factor-cleaving protease after plasma therapy in patients with thrombotic thrombocytopenic purpura. Thromb Haemost 81:8–13
Furlan M, Robles R, Lamie B (1996) Partial purification and characterization of a protease from human plasma cleaving von Willebrand factor to fragments produced by in vivo proteolysis. Blood 87:4223–4234
Tsai HM (1996) Physiologic cleavage of von Willebrand factor by a plasma protease is dependent on its conformation and requires calcium ion. Blood 87:4235–4244
Klaus C, Plaimauer B, Studt JD, et al (2004) Epitope mapping of ADAMTS-13 autoantibodies in acquired thrombotic thrombocytopenic purpura. Blood 103:4514–4519
Scheiflinger F, Knobl P, Trattner B, et al (2003) Nonneutralizing IgM and IgG antibodies to von Willebrand factor-cleaving protease (ADAMTS-13) in a patient with thrombotic thrombocytopenic purpura. Blood 102:3241–3243
Tsai HM, Lian EC (1998) Antibodies to von Willebrand factor-cleaving protease in acute thrombotic thrombocytopenic purpura. N Engl J Med 339:1585–1594
Motto DG, Chauhan AK, Zhu G, et al (2005) Shigatoxin triggers thrombotic thrombocytopenic purpura in genetically susceptible ADAMTS-13-deficient mice. J Clin Invest 115:2752–2761
Bernardo A, Ball C, Nolasco L, Moake JF, Dong JF (2004) Effects of inflammatory cytokines on the release and cleavage of the endothelial cell-derived ultralarge von Willebrand factor multimers under flow. Blood 104:100–106
Crawley JT, Lam JK, Rance JB, Mollica LR, O’Donnell JS, Lane DA (2005) Proteolytic inactivation of ADAMTS-13 by thrombin and plasmin. Blood 105:1085–1093
Reiter RA, Varadi K, Turecek PL, Jilma B, Knobl P (2005) Changes in ADAMTS-13 (von-Willebrand-factor-cleaving protease) activity after induced release of von Willebrand factor during acute systemic inflammation. Thromb Haemost 93:554–558
Nolasco LH, Turner NA, Bernardo A, et al (2005) Hemolytic uremic syndrome-associated Shiga toxins promote endothelial cell secretion and impair ADAMTS-13 cleavage of unusually large von Willebrand factor multimers. Blood 106:4199–4209
Sadler JE, Moake JL, Miyata T, George JN (2004) Recent advances in thrombotic thrombocytopenic purpura. Hematology (Am Soc Hematol Educ Program) 2004:407–423
Veyradier A, Girma JP (2004) Assays of ADAMTS-13 activity. Semin Hematol 41:41–47
Obert B, Tout H, Veyradier A, Fressinaud E, Meyer D, Girma JP (1999) Estimation of the von Willebrand factor-cleaving protease in plasma using monoclonal antibodies to vWF. Thromb Haemost 82:1382–1385
Kokame K, Matsumoto M, Fujimura Y, Miyata T (2004) VWF73, a region from D1596 to R1668 of von Willebrand factor, provides a minimal substrate for ADAMTS-13. Blood 103:607–612
Kokame K, Nobe Y, Kokubo Y, Okayama A, Miyata T (2005) FRETS-VWF73, a first fluorogenic substrate for ADAMTS-13 assay. Br J Haematol 129:93–100
Gerritsen HE, Turecek PL, Schwarz HP, Lammle B, Furlan M (1999) Assay of von Willebrand factor (vWF)-cleaving protease based on decreased collagen binding affinity of degraded vWF: a tool for the diagnosis of thrombotic thrombocytopenic purpura (TTP). Thromb Haemost 82:1386–1389
Bohm M, Vigh T, Scharrer I (2002) Evaluation and clinical application of a new method for measuring activity of von Willebrand factor-cleaving metalloprotease (ADAMTS-13). Ann Hematol 81:430–435
Studt JD, Bohm M, Budde U, Girma JP, Varadi K, Lammle B (2003) Measurement of von Willebrand factor-cleaving protease (ADAMTS-13) activity in plasma: a multicenter comparison of different assay methods. J Thromb Haemost 1:1882–1887
Tripodi A, Chantarangkul V, Bohm M, et al (2004) Measurement of von Willebrand factor cleaving protease (ADAMTS-13): results of an international collaborative study involving 11 methods testing the same set of coded plasmas. J Thromb Haemost 2:1601–1609
Zhou W, Tsai HM (2004) An enzyme immunoassay of ADAMTS-13 distinguishes patients with thrombotic thrombocytopenic purpura from normal individuals and carriers of ADAMTS-13 mutations. Thromb Haemost 91:806–811
Whitelock JL, Nolasco L, Bernardo A, Moake J, Dong JF, Cruz MA (2004) ADAMTS-13 activity in plasma is rapidly measured by a new ELISA method that uses recombinant VWF-A2 domain as substrate. J Thromb Haemost 2:485–491
Studt JD, Hovinga JA, Antoine G, et al (2005) Fatal congenital thrombotic thrombocytopenic purpura with apparent ADAMTS-13 inhibitor: in vitro inhibition of ADAMTS-13 activity by hemoglobin. Blood 105:542–544
Mannucci PM, Capoferri C, Canciani MT (2004) Plasma levels of von Willebrand factor regulate ADAMTS-13, its major cleaving protease. Br J Haematol 126:213–218
Haslan RJ, Rosson GM (1972) Aggregation of human blood platelets by vasopressin. Am J Physiol 223:958–967
Leone M, Albanèse J, Delmas A, Chaabane W, Gamier F, Martin C (2004) Terlipressin in catecholamine resistant septic shock patients. Shock 22:314–319
Dunser MW, Fries DR, Schobersberger W, et al (2004) Does arginine vasopressin influence the coagulation system in advanced vasodilatory shock with severe multiorgan dysfunction syndrome? Anesth Analg 99:201–206
Reiter RA, Knobl P, Varadi K, Turecek PL (2003) Changes in von Willebrand factor-cleaving protease (ADAMTS-13) activity after infusion of desmopressin. Blood 101:946–948
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2006 Springer Science + Business Media Inc.
About this paper
Cite this paper
Claus, R.A., Reinhart, K., Brunkhorst, F.M. (2006). Altered Functionality of von Willebrand Factor in Sepsis and Thrombocytopenia — Potential Role of the vWF Cleaving Protease ADAMTS-13. In: Vincent, JL. (eds) Intensive Care Medicine. Springer, New York, NY. https://doi.org/10.1007/0-387-35096-9_21
Download citation
DOI: https://doi.org/10.1007/0-387-35096-9_21
Publisher Name: Springer, New York, NY
Print ISBN: 978-0-387-30156-3
Online ISBN: 978-0-387-35096-7
eBook Packages: MedicineMedicine (R0)