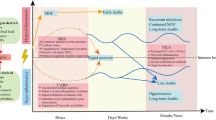
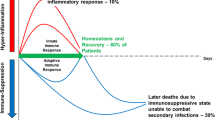
Abstract
Recognition molecules, inflammatory cells, and the cytokines they produce are the principle means for host tissues to recognize invading microbes, and to initiate intercellular communication between the innate and acquired immune systems. However, activation of host innate immunity may also occur in the absence of microbial recognition, through expression of internal “danger signals” produced by tissue ischemia and necrosis, or through the release of free radicals. When activation of the innate immune system is severe enough, the host response itself can propel the patient into a systemic inflammatory response syndrome (SIRS), or even multi-system organ failure (MSOF) and shock. Although the majority of patients survive the initial SIRS insult, these patients remain at increased risk of developing secondary or opportunistic infections due to the frequent onset of a compensatory anti-inflammatory response syndrome (CARS). The initial activation of the innate immune response often leads to macrophage deactivation, T-cell anergy, and the rapid apoptotic loss of lymphoid tissues, which all contribute to the development of this CARS syndrome and its associated morbidity and mortality. Initial efforts to treat the septic and critically ill patient with anti-cytokine therapies directed at the SIRS response have been disappointing, and therapeutic efforts to modify the immune response during sepsis syndromes will require a more thorough understanding of the innate and acquired immune responses.
Chapter PDF
Similar content being viewed by others
Keywords
- Natural Killer Cell
- Septic Shock
- Innate Immune Response
- Systemic Inflammatory Response Syndrome
- Innate Immune System
These keywords were added by machine and not by the authors. This process is experimental and the keywords may be updated as the learning algorithm improves.
References
Fearon, D.T. and R.M. Locksley. 1996. The instructive role of innate immunity in the acquired immune response. Science 272:50–53.
Janeway, C.A.J. 1992. The immune system evolved to discriminate infectious nonself from noninfectious self. Immunol.Today 13:11–16.
Matzinger, P. 1994. Tolerance, danger, and the extended family. Annu.Rev.Immunol. 12:991–1045:991–1045.
Bendelac, A. and D.T. Fearon. 1997. Innate pathways that control acquired immunity [editorial]. Curr.Opin.Immunol. 9:1–3.
Brown, E., J.P. Atkinson, and D.T. Fearon. 1994. Innate immunity: 50 ways to kill a microbe. Curr.Opin.Immunol. 6:73–74.
Ezekowitz, R.B. and J.A. Hoffmann. 1996. Innate immunity. Curr.Opin.Immunol. 8:1–2.
Oberholzer, A., C. Oberholzer, and L.L. Moldawer. 2000. Cytokine signaling--regulation of the immune response in normal and critically ill states. Crit.Care Med. 28:N3–12.
Janeway, C.A.J. 2001. How the immune system works to protect the host from infection: a personal view. Proc.Natl.Acad.Sci.U.S.A. 98:7461–7468.
Beutler, B. 2000. Endotoxin, toll-like receptor 4, and the afferent limb of innate immunity. Curr.Opin.Microbiol. 3:23–28.
O’Neill, L.A. and C.A. Dinarello. 2000. The IL-1 receptor/toll-like receptor superfamily: crucial receptors for inflammation and host defense [In Process Citation]. Immunol.Today 21:206–209.
Brightbill, H.D., D.H. Libraty, S.R. Krutzik, R.B. Yang, J.T. Belisle, J.R. Bleharski, M. Maitland, M.V. Norgard, S.E. Plevy, S.T. Smale, P.J. Brennan, B.R. Bloom, P.J. Godowski, and R.L. Modlin. 1999. Host defense mechanisms triggered by microbial lipoproteins through toll-like receptors. Science 285:732–736.
Schwandner, R., R. Dziarski, H. Wesche, M. Rothe, and C.J. Kirschning. 1999. Peptidoglycan- and lipoteichoic acid-induced cell activation is mediated by toll-like receptor 2. J.Biol.Chem. 274:17406–17409.
Yoshimura, A., E. Lien, R.R. Ingalls, E. Tuomanen, R. Dziarski, and D. Golenbock. 1999. Cutting edge: recognition of Gram-positive bacterial cell wall components by the innate immune system occurs via Toll-like receptor 2. J.Immunol. 163:1–5.
Takeshita, F., C.A. Leifer, I. Gursel, K.J. Ishii, S. Takeshita, M. Gursel, and D.M. Klinman. Cutting edge: role of toll-like receptor 9 in cpg dna-induced activation of human cells. J.Immunol.2001.Oct.l.;167.(7.):3555.-8. 167:3555–3558.
Klinman, D.M., A.K. Yi, S.L. Beaucage, J. Conover, and A.M. Krieg. 1996. CpG motifs present in bacteria DNA rapidly induce lymphocytes to secrete interleukin 6, interleukin 12, and interferon gamma. Proc.Natl.Acad.Sci.U.S.A. 93:2879–2883.
Schnare, M., G.M. Barton, A.C. Holt, K. Takeda, S. Akira, and R. Medzhitov. 2001. Toll-like receptors control activation of adaptive immune responses. Nat.Immunol. 2:947–950.
Gabay, C. and I. Kushner. 1999. Acute-phase proteins and other systemic responses to inflammation. N.Engl.J.Med. 340:448–454.
Qureshi, S.T., L. Lariviere, G. Leveque, S. Clermont, K.J. Moore, P. Gros, and D. Malo. 1999. Endotoxin-tolerant mice have mutations in Toll-like receptor 4 (Tlr4) [see comments] [published erratum appears in J Exp Med 1999 May 3;189(9):following 1518]. J.Exp.Med. 189:615–625.
Poltorak, A., X. He, I. Smirnova, M.Y. Liu, C.V. Huffel, X. Du, D. Birdwell, E. Alejos, M. Silva, C. Galanos, M. Freudenberg, P. Ricciardi-Castagnoli, B. Layton, and B. Beutler. 1998. Defective LPS signaling in C3H/HeJ and C57BL/10ScCr mice: mutations in Tlr4 gene. Science 282:2085–2088.
Akira, S., K. Takeda, and T. Kaisho. 2001. Toll-like receptors: critical proteins linking innate and acquired immunity. Nat.Immunol. 2:675–680.
Kaisho, T. and S. Akira. 2001. Dendritic-cell function in Toll-like receptor- and MyD88-knockout mice. Trends.Immunol. 22:78–83.
Huang, Q., D. Liu, P. Majewski, L.C. Schulte, J.M. Korn, R.A. Young, E. Lander, and N. Hacohen. 2001. The plasticity of dendritic cell responses to pathogens and their components. Science 294:870–875.
Tobias, P.S., K. Soldau, and R.J. Ulevitch. 1986. Isolation of a lipopolysaccharide-binding acute phase reactant from rabbit serum. J.Exp.Med. 164:777–793.
Ulevitch, R.J. and P.S. Tobias. 1995. Receptor-dependent mechanisms of cell stimulation by bacterial endotoxin. Annu.Rev.Immunol. 13:437–57:437–457.
Oberholzer, A., C. Oberholzer, and L.L. Moldawer. 2001. Sepsis syndromes: understanding the role of innate and acquired immunity. Shock 16:83–96.
Tomlinson, S. 1993. Complement defense mechanisms. Curr.Opin.Immunol. 5:83–89.
Liszewski, M.K., T.W. Post, and J.P. Atkinson. 1991. Membrane cofactor protein (MCP or CD46): newest member of the regulators of complement activation gene cluster. Annu.Rev.Immunol. 9:431–55.:431–455.
Frank, M.M. and L.F. Fries. 1991. The role of complement in inflammation and phagocytosis. Immunol.Today 12:322–326.
Bhakdi, S. and J. Tranum-Jensen. 1991. Complement lysis: a hole is a hole. Immunol.Today 12:318–320.
Kreutz, M., U. Ackermann, S. Hauschildt, S.W. Krause, D. Riedel, W. Bessler, and R. Andreesen. 1997. A comparative analysis of cytokine production and tolerance induction by bacterial lipopeptides, lipopolysaccharides and Staphyloccous aureus in human monocytes. Immunology 92:396–401.
Munoz, C., J. Carlet, C. Fitting, B. Misset, J.P. Bleriot, and J.M. Cavaillon. 1991. Dysregulation of in vitro cytokine production by monocytes during sepsis. J.Clin.Invest. 88:1747–1754.
Rosenbach, T. and B.M. Czarnetzki. 1987. Comparison of the generation in vitro of chemotactically active LTB4 and its omega-metabolites by human neutrophils and lymphocytes/monocytes. Clin.Exp.Immunol. 69:221–228.
Miyamoto, K., M. Lange, G. McKinley, C. Stavropoulos, S. Moriya, H. Matsumoto, and Y. Inada. 1996. Effects of sho-saiko-to on production of prostaglandin E2 (PGE2), leukotriene B4 (LTB4) and superoxide from peripheral monocytes and polymorphonuclear cells isolated from HIV infected individuals. Am.J.Chin.Med. 24:1–10.
Simoni, J., G. Simoni, C.D. Lox, D.E. McGunegle, and M. Feola. 1994. Cytokines and PAF release from human monocytes and macrophages: effect of hemoglobin and contaminants. Artif.Cells Blood Substit.Immobil.Biotechnol. 22:525–534.
Camussi, G., C. Tetta, R. Coda, and J. Benveniste. 1981. Release of platelet-activating factor in human pathology. I. Evidence for the occurrence of basophil degranulation and release of platelet-activating factor in systemic lupus erythematosus. Lab.Invest. 44:241–251.
Dugas, B., M.D. Mossalayi, C. Damais, and J.P. Kolb. 1995. Nitric oxide production by human monocytes: evidence for a role of CD23. Immunol.Today 16:574–580.
Campbell, J.J., J. Hedrick, A. Zlotnik, M.A. Siani, D.A. Thompson, and E.C. Butcher. 1998. Chemokines and the arrest of lymphocytes rolling under flow conditions. Science 279:381–384.
Foreman, K.E., A.A. Vaporciyan, B.K. Bonish, M.L. Jones, K.J. Johnson, M.M. Glovsky, S.M. Eddy, and P.A. Ward. 1994. C5a-induced expression of P-selectin in endothelial cells. J.Clin.Invest. 94:1147–1155.
Noble, K.E., P. Panayiotidis, P.W. Collins, A.V. Hoffbrand, and K.L. Yong. 1996. Monocytes induce E-selectin gene expression in endothelial cells: role of CD11/CD18 and extracellular matrix proteins. Eur.J.Immunol. 26:2944–2951.
Melder, R.J., L.L. Munn, S. Yamada, C. Ohkubo, and R.K. Jain. 1995. Selectin- and integrin-mediated T-lymphocyte rolling and arrest on TNF-alpha-activated endothelium: augmentation by erythrocytes. Biophys.J. 69:2131–2138.
Lim, Y.C., K. Snapp, G.S. Kansas, R. Camphausen, H. Ding, and F.W. Luscinskas. 1998. Important contributions of P-selectin glycoprotein ligand-1-mediated secondary capture to human monocyte adhesion to P-selectin, E-selectin, and TNF-alpha-activated endothelium under flow in vitro. J.Immunol. 161:2501–2508.
Kuypers, T.W. and D. Roos. 1989. Leukocyte membrane adhesion proteins LFA-1, CR3 and p150,95: a review of functional and regulatory aspects. Res.Immunol. 140:461–486.
Taub, D.D. 1996. Chemokine-leukocyte interactions. The voodoo that they do so well. Cytokine.Growth Factor.Rev. 7:355–376.
Ward, S.G. and J. Westwick. 1998. Chemokines: understanding their role in T-lymphocyte biology. Biochem.J. 333:457–470.
Nelson, P.J. and A.M. Krensky. 1998. Chemokines, lymphocytes and viruses: what goes around, comes around. Curr.Opin.Immunol. 10:265–270.
Leonard, E.J., A. Skeel, T. Yoshimura, and J. Rankin. 1993. Secretion of monocyte chemoattractant protein-1 (MCP-1) by human mononuclear phagocytes. Adv.Exp.Med.Biol. 351:55–64.:55–64.
Engelhardt, E., A. Toksoy, M. Goebeler, S. Debus, E.B. Brocker, and R. Gillitzer. 1998. Chemokines IL-8, GROalpha, MCP-1, IP-10, and Mig are sequentially and differentially expressed during phase-specific infiltration of leukocyte subsets in human wound healing. Am.J.Pathol. 153:1849–1860.
Antony, V.B., S.W. Godbey, S.L. Kunkel, J.W. Hott, D.L. Hartman, M.D. Burdick, and R.M. Strieter. 1993. Recruitment of inflammatory cells to the pleural space. Chemotactic cytokines, IL-8, and monocyte chemotactic peptide-1 in human pleural fluids. J.Immunol. 151:7216–7223.
Uguccioni, M., P. Gionchetti, D.F. Robbiani, F. Rizzello, S. Peruzzo, M. Campieri, and M. Baggiolini. 1999. Increased expression of IP-10, IL-8, MCP-1, and MCP-3 in ulcerative colitis. Am.J.Pathol. 155:331–336.
Tekstra, J., H. Beekhuizen, Van De Gevel JS, B.I. Van, C.W. Tuk, and R.H. Beelen. 1999. Infection of human endothelial cells with Staphylococcus aureus induces the production of monocyte chemotactic protein-1 (MCP-1) and monocyte Chemotaxis. Clin.Exp.Immunol. 117:489–495.
Capelli, A., S.A. Di, I. Gnemmi, P. Balbo, C.G. Cerutti, B. Balbi, M. Lusuardi, and C.F. Donner. 1999. Increased MCP-1 and MIP-1beta in bronchoalveolar lavage fluid of chronic bronchitics. Eur.Respir.J. 14:160–165.
Hoch, R.C., R. Rodriguez, T. Manning, M. Bishop, P. Mead, W.C. Shoemaker, and E. Abraham. 1993. Effects of accidental trauma on cytokine and endotoxin production. Crit.Care Med. 21:839–845.
Zhao, Y.X., H. Zhang, B. Chiu, U. Payne, and R.D. Inman. 1999. Tumor necrosis factor receptor p55 controls the severity of arthritis in experimental Yersinia enterocolitica infection. Arthritis Rheum. 42:1662–1672.
Birdsall, H.H., D.M. Green, J. Trial, K.A. Youker, A.R. Burns, C.R. MacKay, G.J. LaRosa, H.K. Hawkins, C.W. Smith, L.H. Michael, M.L. Entman, and R.D. Rossen. 1997. Complement C5a, TGF-beta 1, and MCP-1, in sequence, induce migration of monocytes into ischemic canine myocardium within the first one to five hours after reperfusion. Circulation 95:684–692.
Ribeiro, R.A., M.V. Souza-Filho, M.H. Souza, S.H. Oliveira, C.H. Costa, F.Q. Cunha, and H.S. Ferreira. 1997. Role of resident mast cells and macrophages in the neutrophil migration induced by LTB4, fMLP and C5a des arg. Int.Arch.Allergy Immunol. 112:27–35.
Vassalli, P. 1992. The pathophysiology of tumor necrosis factors. Ann.Rev.Immunol. 10:411–452.
Beutler, B. and A. Cerami. 1986. Cachectin and tumour necrosis factor as two sides of the same biological coin. Nature 320:584–588.
Ksontini, R., S.L. MacKay, and L.L. Moldawer. 1998. Revisiting the role of tumor necrosis factor alpha and the response to surgical injury and inflammation. Arch Surg 133:558–567.
Tracey, K.J., B. Beutler, S.F. Lowry, J. Merryweather, S. Wolpe, I.W. Milsark, R.J. Hariri, T.J. Fahey, A. Zentella, J.D. Albert, and et al. 1986. Shock and tissue injury induced by recombinant human cachectin. Science 234:470–474.
Tracey, K.J., S.F. Lowry, and A. Cerami. 1988. Cachetin/TNF-alpha in septic shock and septic adult respiratory distress syndrome. Am.Rev.Respir.Dis. 138:1377–1379.
Tracey, K.J., Y. Fong, D.G. Hesse, K.R. Manogue, A.T. Lee, G.C. Kuo, S.F. Lowry, and A. Cerami. 1987. Anti-cachectin/TNF monoclonal antibodies prevent septic shock during lethal bacteraemia. Nature 330:662–664.
Baumann, H., V. Onorato, J. Gauldie, and G.P. Jahreis. 1987. Distinct sets of acute phase plasma proteins are stimulated by separate human hepatocyte-stimulating factors and monokines in rat hepatoma cells. J.Biol.Chem. 262:9756–9768.
Castell, J.V., M.J. Gomez-Lechon, M. David, T. Andus, T. Geiger, R. Trullenque, R. Fabra, and P.C. Heinrich. 1989. Interleukin-6 is the major regulator of acute phase protein synthesis in adult human hepatocytes. FEBS Lett. 242:237–239.
Yamada, Y., K. Kimball, S. Okusawa, G. Vachino, N. Margolis, J.W. Sohn, J.J. Li, G. Wakabayashi, K. McAdam, and J.F. Burke. 1990. Cytokines, acute phase proteins, and tissue injury. C-reactive protein opsonizes dead cells for debridement and stimulates cytokine production. Ann.N.Y.Acad.Sci. 587:351–61.:351–361.
Thiel, S., U. Holmskov, L. Hviid, S.B. Laursen, and J.C. Jensenius. 1992. The concentration of the C-type lectin, mannan-binding protein, in human plasma increases during an acute phase response. Clin.Exp.Immunol. 90:31–35.
Thiel, S., S.V. Petersen, T. Vorup-Jensen, M. Matsushita, T. Fujita, C.M. Stover, W.J. Schwaeble, and J.C. Jensenius. 2000. Interaction of C1q and mannan-binding lectin (MBL) with C1r, C1s, MBL-associated serine proteases 1 and 2, and the MBL-associated protein MAp19. J.Immunol. 165:878–887.
Fraser, I.P., H. Koziel, and R.A. Ezekowitz. 1998. The serum mannose-binding protein and the macrophage mannose receptor are pattern recognition molecules that link innate and adaptive immunity. Semin.Immunol. 10:363–372.
Movat, H.Z., M.I. Cybulsky, I.G. Colditz, M.K. Chan, and C.A. Dinarello. 1987. Acute inflammation in gram-negative infection: endotoxin, interleukin 1, tumor necrosis factor, and neutrophils. Fed.Proc. 46:97–104.
Bone, R.C. 1992. Inhibitors of complement and neutrophils: a critical evaluation of their role in the treatment of sepsis. Crit.Care Med. 20:891–898.
Welbourn, C.R. and Y. Young. 1992. Endotoxin, septic shock and acute lung injury: neutrophils, macrophages and inflammatory mediators. Br.J.Surg. 79:998–1003.
Harris, M.C., J. Stroobant, C.S. Cody, S.D. Douglas, and R.A. Polin. 1983. Phagocytosis of group B Streptococcus by neutrophils from newborn infants. Pediatr.Res. 17:358–361.
Schorlemmer, H.U., T. Hofstaetter, and F.R. Seiler. 1984. Phagocytosis of immune complexes by human neutrophils and monocytes: relative importance of Fc and C3b receptors. Behring.Inst.Mitt. 88–97.
Borrego, F., M. Ulbrecht, E.H. Weiss, J.E. Coligan, and A.G. Brooks. 1998. Recognition of human histocompatibility leukocyte antigen (HLA)-E complexed with HLA class I signal sequence-derived peptides by CD94/NKG2 confers protection from natural killer cell-mediated lysis. J.Exp.Med. 187:813–818.
Moretta, A., C. Bottino, M. Vitale, D. Pende, R. Biassoni, M.C. Mingari, and L. Moretta. 1996. Receptors for HLA class-I molecules in human natural killer cells. Annu.Rev.Immunol. 14:619–48.:619–648.
Biron, C.A., K.B. Nguyen, G.C. Pien, L.P. Cousens, and T.P. Salazar-Mather. 1999. Natural killer cells in antiviral defense: function and regulation by innate cytokines. Annu.Rev.Immunol. 17:189–220.:189–220.
Herberman, R.B., C.W. Reynolds, and J.R. Ortaldo. 1986. Mechanism of cytotoxicity by natural killer (NK) cells. Annu.Rev.Immunol. 4:651–80.:651–680.
Calandra, T., J.D. Baumgartner, G.E. Grau, M.M. Wu, P.H. Lambert, J. Schellekens, J. Verhoef, and M.P. Glauser. 1990. Prognostic values of tumor necrosis factor/cachectin, interleukin-1, interferon-alpha, and interferon-gamma in the serum of patients with septic shock. Swiss-Dutch J5 Immunoglobulin Study Group. J.Infect.Dis. 161:982–987.
Drost, A.C., D.G. Burleson, W.G. Cioffi, Jr., B.S. Jordan, A.D. Mason, Jr., and B.A.J. Pruitt. 1993. Plasma cytokines following thermal injury and their relationship with patient mortality, burn size, and time postburn. J.Trauma. 35:335–339.
Casey, L.C., R.A. Balk, and R.C. Bone. 1993. Plasma cytokine and endotoxin levels correlate with survival in patients with the sepsis syndrome [see comments]. Ann Intern.Med. 119:771–778.
Sherry, R.M., J.I. Cue, J.K. Goddard, J.B. Parramore, and J.T. DiPiro. 1996. Interleukin-10 is associated with the development of sepsis in trauma patients. J.Trauma. 40:613–616.
Neidhardt, R., M. Keel, U. Steckholzer, A. Safret, U. Ungethuem, O. Trentz, and W. Ertel. 1997. Relationship of interleukin-10 plasma levels to severity of injury and clinical outcome in injured patients. J.Trauma. 42:863–870.
Suter, P.M., S. Suter, E. Girardin, P. Roux Lombard, G.E. Grau, and J.M. Dayer. 1992. High bronchoalveolar levels of tumor necrosis factor and its inhibitors, interleukin-1, interferon, and elastase, in patients with adult respiratory distress syndrome after trauma, shock, or sepsis. Am.Rev.Respir.Dis. 145:1016–1022.
Holzheimer, R.G., M. Schein, and D.H. Wittmann. 1995. Inflammatory response in peritoneal exudate and plasma of patients undergoing planned relaparotomy for severe secondary peritonitis..Arch.Surg. 130:1314–1319.
American College of Chest Physicians/Society of Critical Care Medicine Consensus Conference. 1992. Definitions for sepsis and the response to surgical injury and inflammation. Crit.Care Med. 20:864–871.
Bone, R.C. 1996. Toward a theory regarding the pathogenesis of the systemic inflammatory response syndrome: what we do and do not know about cytokine regulation. Crit.Care Med. 24:163–172.
Bone, R.C. 1996. Sir Isaac Newton, sepsis, SIRS, and CARS. Crit.Care Med. 24:1125–1128.
Wessels, M.R., P. Butko, M. Ma, H.B. Warren, A.L. Lage, and M.C. Carroll. 1995. Studies of group B streptococcal infection in mice deficient in complement component C3 or C4 demonstrate an essential role for complement in both innate and acquired immunity. Proc.Natl.Acad.Sci.U.S.A. 92:11490–11494.
Czermak, B.J., V. Sarma, C.L. Pierson, R.L. Warner, M. Huber-Lang, N.M. Bless, H. Schmal, H.P. Friedl, and P.A. Ward. 1999. Protective effects of C5a blockade in sepsis. Nat.Med. 5:788–792.
Bengtsson, A., H. Redl, E. Paul, G. Schlag, T.E. Mollnes, and J. Davies. 1993. Complement and leukocyte activation in septic baboons. Circ.Shock 39:83–88.
Friedman, G., S. Jankowski, M. Shahla, M. Goldman, R.M. Rose, R.J. Kahn, and J.L. Vincent. 1996. Administration of an antibody to E-selectin in patients with septic shock. Crit.Care Med. 24:229–233.
Cummings, C.J., C.N. Sessler, L.D. Beall, B.J. Fisher, A.M. Best, and A.A. Fowler. 1997. Soluble E-selectin levels in sepsis and critical illness. Correlation with infection and hemodynamic dysfunction. Am.J.Respir.Crit.Care Med. 156:431–437.
Schlag, G., H.R. Redi, G.O. Till, J. Davies, U. Martin, and L. Dumont. 1999. Anti-L-selectin antibody treatment of hemorrhagic-traumatic shock in baboons. Crit.Care Med. 27:1900–1907.
Essani, N.A., M.A. Fisher, C.A. Simmons, J.L. Hoover, A. Farhood, and H. Jaeschke. 1998. Increased P-selectin gene expression in the liver vasculature and its role in the pathophysiology of neutrophil-induced liver injury in murine endotoxin shock. J.Leukoc.Biol. 63:288–296.
Ohlsson, K., P. Bjork, M. Bergenfeldt, R. Hageman, and R.C. Thompson. 1990. Interleukin-1 receptor antagonist reduces mortality from endotoxin shock. Nature 348:550–552.
Fischer, E., M.A. Marano, Z.K. Van, C.S. Rock, A.S. Hawes, W.A. Thompson, L. DeForge, J.S. Kenney, D.G. Remick, and D.C. Bloedow. 1992. Interleukin-1 receptor blockade improves survival and hemodynamic performance in Escherichia coli septic shock, but fails to alter host responses to sublethal endotoxemia. J.Clin.Invest. 89:1551–1557.
Zeni, F., B. Freeman, and C. Natanson. 1997. Anti-inflammatory therapies to treat sepsis and septic shock: a reassessment [editorial; comment]. Crit.Care Med. 25:1095–1100.
Baue, A.E. 1997. Multiple organ failure, multiple organ dysfunction syndrome, and systemic inflammatory response syndrome. Why no magic bullets? Arch Surg 132:703–707.
Ertel, W., J.P. Kremer, J. Kenney, U. Steckholzer, D. Jarrar, O. Trentz, and F.W. Schildberg. 1995. Downregulation of proinflammatory cytokine release in whole blood from septic patients. Blood 85:1341–1347.
Zheng, L., G. Fisher, R.E. Miller, J. Peschon, D.H. Lynch, and M.J. Lenardo. 1995. Induction of apoptosis in mature T cells by tumour necrosis factor. Nature 377:348–351.
Dhein, J., H. Walczak, C. Baumler, K.M. Debatin, and P.H. Krammer. 1995. Autocrine T-cell suicide mediated by APO-l/(Fas/CD95) [see comments]..Nature 373:438–441.
Jimenez, M.F., R.W. Watson, J. Parodo, D. Evans, D. Foster, M. Steinberg, O.D. Rotstein, and J.C. Marshall. 1997. Dysregulated expression of neutrophil apoptosis in the systemic inflammatory response syndrome [In Process Citation]. Arch Surg 132:1263–1269.
Keel, M., U. Ungethum, U. Steckholzer, E. Niederer, T. Hartung, O. Trentz, and W. Ertel. 1997. Interleukin-10 counterregulates proinflammatory cytokine-induced inhibition of neutrophil apoptosis during severe sepsis. Blood 90:3356–3363.
Matute-Bello, G., W.C. Liles, F. Radella, K.P. Steinberg, J.T. Ruzinski, M. Jonas, E.Y. Chi, L.D. Hudson, and T.R. Martin. 1997. Neutrophil apoptosis in the acute respiratory distress syndrome. Am.J.Respir.Crit.Care Med. 156:1969–1977.
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2003 Springer Science+Business Media New York
About this chapter
Cite this chapter
Oberholzer, A., Oberholzer, C., Moldawer, L.L. (2003). Activation of the Innate Immune Response in Critical Illness. In: Doughty, L.A., Linden, P. (eds) Immunology and Infectious Disease. Molecular and Cellular Biology of Critical Care Medicine, vol 3. Springer, Boston, MA. https://doi.org/10.1007/978-1-4615-0245-6_2
Download citation
DOI: https://doi.org/10.1007/978-1-4615-0245-6_2
Publisher Name: Springer, Boston, MA
Print ISBN: 978-1-4613-4984-6
Online ISBN: 978-1-4615-0245-6
eBook Packages: Springer Book Archive