Abstract
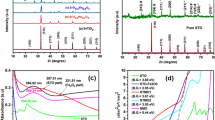
A simple solid-state route has been utilized to prepare Sr3FeNb2O9 particles with the tetragonal (SG P4/mmm), double-perovskite structure. The structural investigation and a calcination temperature optimization in the range of 700–1300 °C revealed that a pure phase Sr3FeNb2O9 could be achieved at a temperature of 1200 °C. The crystallites with the sizes in the range of ~5–10 μm, exhibited a bandgap of 2.25 eV as deduced from diffuse reflectance studies. The structural and the optical properties demonstrated its suitability for visible-light active photocatalyst applications. The electrochemical characterization indicated a comparatively negative flat-band potential value for Sr3FeNb2O9 particles with respect to the reduction potential of water, indicating its capability to reduce water molecule. A very low Pt loading (< 0.2 wt%) over Sr3FeNb2O9 yielded a high photocatalytic hydrogen evolution efficiency under visible light (λ ≥ 420 nm). Its photoreduction activity for water was found to be 1.4 times higher than that of the reference Sr2FeNbO6 photocatalyst prepared separately. The overall physico-chemical analysis in this study revealed that the suitable band-edge position of Sr3FeNb2O9 could be attributed to its superior performance as compared to Sr2FeNbO6. A mechanism is proposed to explain the photocatalytic behavior of the Sr3FeNb2O9 photocatalyst.
Similar content being viewed by others
References
D. E. Scaife, Solar Energy 25, 41 (1980).
Y. Matsumoto, J. Solid State Chem. 126, 227 (1996).
J. S. Lee, Catal. Surv. Asia 9, 217 (2005).
A. Kudo and Y. Miseki, Chem. Soc. Rev. 38, 253 (2009).
F. E. Osterloh, Chem. Mater. 20, 35 (2008).
H. G. Kim, D. W. Hwang and J. S. Lee, J. Am. Chem. Soc. 126, 8913 (2004).
K. Maeda, T. Takata, M. Hara, N. Saito, Y. Inoue, H. Kobayashi and K. Domen, J. Am. Chem. Soc. 127, 8286 (2005).
H. G. Kim, P. H. Borse, W. Choi and J. S. Lee, Angew. Chem. Int. Ed. 44, 4585 (2005).
J. S. Jang, D. W. Hwang and J. S. Lee, Catal. Today, 120, 174 (2007).
Y. Matsumoto, M. Omae, K. Sugiyama and E.-I. Sato, J. Phys. Chem. 91, 577 (1987).
T. Sreethawong, Y. Suzuki and S. Yoshikawa, J. Solid State Chem. 178, 329 (2005).
J. S. Jang, H. G. Kim, S. M. Ji, S. W. Bae, J. H. Jung, B. H. Shon and J. S. Lee, J. Solid State Chem. 179, 1064 (2006).
E. D. Jeong, S. M. Yu, J. Y. Yoon, J. S. Bae, C. R. Cho, K. T. Lim, R. Dom, P. H. Borse and H. G. Kim, J. Ceram. Proc. Res. 13, 305 (2012).
R. Dom, P. H. Borse, C. R. Cho, J. S. Lee, S. M. Yu, J. H. Yoon, T. E. Hong, E. D. Jeong and H. G. Kim, J. Ceram. Proc. Res. 13, 451 (2012).
R. Asahi, T. Morikawa, T. Ohwaki, K. Aoki and Y. Taga, Science 293, 269 (2001).
P. H. Borse, C. R. Cho, K. T. Lim, Hong, E. D. Jeong, J. H. Yoon, S. M. Yu and H. G. Kim, J. Ceram. Proc. Res. 13, 42 (2012).
H. G. Kim, P. H. Borse, J. S. Jang, E. D. Jeong and J. S. Lee, Mater. Lett. 62, 1427 (2008).
N. Hatanaka, T. Kobayashi, H. Yoneyama and H. Tamura, Electrochim. Acta 27, 1129 (1982).
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Cha, Y.J., Bae, J.S., Hong, T.E. et al. Structural, optical and visible-light photocatalytic properties of Sr3FeNb2O9 oxide. Journal of the Korean Physical Society 65, 520–525 (2014). https://doi.org/10.3938/jkps.65.520
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.3938/jkps.65.520