Abstract
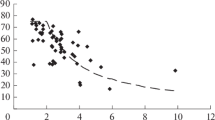
The emission of HCl from calcium chloride during the air–steam gasification of solid fuel in the filtration combustion mode was studied. The limiting amounts of HCl released into the gas phase under real conditions of a shaft kiln gasifier were estimated. It was shown that the most important factors responsible for the stability of CaCl2 are the humidity of an oxidant gas and the process temperature.
Similar content being viewed by others
References
Gould, O.E., US Patent 4732091, 1988.
Addink, K., Bakker, W.C.M., and Olie, K., Environ. Sci. Technol., 1995, vol. 29, p. 2055.
Glazov, S.V. and Polianczyk, E.V., Combust. Explos. Shock Waves, 2015, vol. 51, no. 5, p. 540.
Kolesnikova, Yu.Yu., Kislov, V.M., and Salgansky, E.A., Russ. J. Phys. Chem. B, 2016, vol. 10, no. 5, p. 791.
Salgansky, E.A., Zaichenko, A.Yu., Podlesniy, D.N., Salganskaya, M.V., and Tsvetkov, M.V., Fuel, 2017, vol. 210, p. 491.
Balabaeva, E.M., Korshunova, L.A., Manelis, G.B., Polianchik, E.V., and Tsvetkov, M.V., Int. J. Sci. Alter. Energy Ecol., 2009, vol. 76, no. 8, p. 190.
Tsvetkov, M.V., Zaichenko, A.Yu., and Zhirnov, A.A., Theor. Found. Chem. Eng., 2013, vol. 47, no. 5, p. 608.
Tsvetkov, M.V., Cand. Sci. (Chem.) Dissertation, Chernogolovka: Inst. Probl. Chem. Phys., Russ. Acad. Sci., 2010.
Weinell, C.E., Jensen, P.I., Dam-Johansen, K., and Livbjerg, H., Ind. Eng. Chem. Res., 1992, vol. 31, p. 164.
Matsukata, M., Takeda, K., Miyatani, T., and Ueyama, K., Chem. Eng. Sci., 1996, vol. 51, p. 2529.
Partanen, J., Backman, P., Backman, R., and Hupa, M., Fuel, 2005, vol. 84, p. 1664.
Wang, W., Zhicheng, Y., and Bjerle, I., Fuel, 1996, vol. 75, p. 207.
Trusov, B.G., Abstracts of Papers, XIV Int. Symp. on Chemical Thermodynamics, St. Petersburg, 2002, p. 483.
Zaichenko, A.Yu., Zhirnov, A.A., Manelis, G.B., Polianchik, E.V., and Zholudev, A.F., Theor. Found. Chem. Eng., 2010, vol. 44, no. 1, p. 30.
Salgansky, E.A., Kislov, V.M., Glazov, S.V., and Salganskaya, M.V., J. Combustion, 2016. vol. 3, no. 5.
Kislov, V.M., Salganskii, E.A., Tsvetkov, M.V., and Tsvetkova, Yu.Yu., Russ. J. Appl. Chem., 2017, vol. 90, no. 5, p. 716.
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © M.V. Tsvetkov, E.V. Polianczyk, A.Yu. Zaichenko, Yu.Yu. Tsvetkova, D.N. Podlesniy, 2018, published in Khimiya Tverdogo Topliva, 2018, No. 2, pp. 31–35.
About this article
Cite this article
Tsvetkov, M.V., Polianczyk, E.V., Zaichenko, A.Y. et al. Stability of Calcium Chloride in the Air–Steam Gasification of Solid Fuel in Filtration Mode. Solid Fuel Chem. 52, 86–90 (2018). https://doi.org/10.3103/S036152191802009X
Received:
Published:
Issue Date:
DOI: https://doi.org/10.3103/S036152191802009X