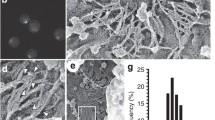
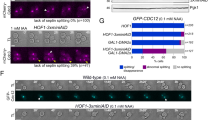
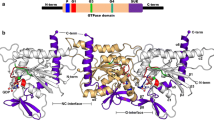
Abstract
Septins are a family of eukaryotic guanosine phosphate-binding proteins that form linear heterooligomeric complexes, which, in turn, polymerize end-on-end into filaments. These filaments further assemble into higher-order structures at distinct subcellular locations. Dynamic changes in the organization of septin cortex structures appear during cell cycle progression. A variety of regulatory proteins and posttranslational modifications are involved in changes to the structure of septin assemblies during the entire cell cycle. In particular, septin-associated protein kinases mediate changes to septin higher order structures or interconnect cellular morphogenesis with the cell cycle. Yeast cyclin-dependent kinase, a master cell cycle regulator, is required for the initiation of a new septin ring. Here, using epifluoresence and electron microscopy, we show that upon phosphorylation by the Cdc28 kinase, septin filaments disassemble into hetero-octameric building blocks, and that filament depolymerization is specifically G1 cyclin-dependent.



Similar content being viewed by others
Abbreviations
- CDK:
-
Cyclin-dependent kinase
- DTT:
-
Dithiothreitol
- EGFP:
-
Enhanced green fluorescent protein
- IPTG:
-
Isopropyl-β-D-thiogalactopyranosid
- NTA:
-
Nitrilotriacetic acid
- PMSF:
-
Phenylmethylsulfonylfluorid
- SD:
-
Standard deviation
References
Albuquerque CP, Smolka MB, Payne SH, Bafna V, Eng J, Zhou H (2008) A multidimensional chromatography technology for in-depth phosphoproteome analysis. Mol Cell Proteomics 7:1389–1396. https://doi.org/10.1074/mcp.M700468-MCP200
Ausubel FM, Brent R, Kingston RE, Moore DD, Seidman JG, Smith JA, Struhl K (1995) Current protocols in molecular biology. John Wiley and Sons, New York
Bertin A, McMurray MA, Grob P, Park SS, Garcia G 3rd, Patanwala I, Ng HL, Alber T, Thorner J, Nogales E (2008) Saccharomyces cerevisiae septins: supramolecular organization of heterooligomers and the mechanism of filament assembly. Proc Natl Acad Sci U S A 105:8274–8279. https://doi.org/10.1073/pnas.0803330105
Bröcker C, Kuhlee A, Gatsogiannis C, Balderhaar HJ, Hönscher C, Engelbrecht-Vandré S, Ungermann C, Raunser S (2012) Molecular architecture of the multisubunit homotypic fusion and vacuole protein sorting (HOPS) tethering complex. Proc Natl Acad Sci U S A 109:1991–1996. https://doi.org/10.1073/pnas.1117797109
Cvrčková F, De Virgilio C, Manser E, Pringle JR, Nasmyth K (1995) Ste20-like protein kinases are required for normal localization of cell growth and for cytokinesis in budding yeast. Genes Dev 9:1817–1830
Egelhofer TA, Villén J, McCusker D, Gygi SP, Kellogg DR, Hardwick KG (2008) The Septins Function in G1 Pathways that Influence the Pattern of Cell Growth in Budding Yeast. PLoS ONE 3:e2022. https://doi.org/10.1371/journal.pone.0002022
Farkasovsky M, Herter P, Voss B, Wittinghofer A (2005) Nucleotide binding and filament assembly of recombinant yeast septin complexes. Biol Chem 386:643–656. https://doi.org/10.1515/BC.2005.075
Garcia G III, Bertin A, Li Z, Song Y, McMurray MA, Thorner J, Nogales E (2011) Subunit-dependent modulation of septin assembly: budding yeast septin Shs1 promotes ring and gauze formation. J Cell Biol 195:993–1004. https://doi.org/10.1083/jcb.201107123
Garcia G III, Finnigan GC, Heasley LR, Sterling SM, Aggarwal A, Pearson CG, Nogales E, McMurray MA, Thorner J (2016) Assembly, molecular organization, and membrane-binding properties of development specific septins. J Cell Biol 212:515–529. https://doi.org/10.1083/jcb.201511029
Haan C, Behrmann I (2007) A cost effective non-commercial ECL-solution for western blot detections yielding strong signals and low background. J Immunol Methods 318:11–19. https://doi.org/10.1016/j.jim.2006.07.027
Hartwell LH, Smith D (1985) Altered fidelity of mitotic chromosome transmission in cell cycle mutants of S. cerevisiae. Genetics 110:381–395
Holt LJ, Tuch BB, Villén J, Johnson AD, Gygi SP, Morgan DO (2009) Global analysis of Cdk1 substrate phosphorylation sites provides insights into evolution. Science 325:1682–1686. https://doi.org/10.1126/science.1172867
Huang D, Friesen H, Andrews B (2007) Pho85, a multifunctional cyclin dependent protein kinase in budding yeast. Mol Microbiol 66:303–314. https://doi.org/10.1111/j.1365-2958.2007.05914.x
Johnson ES, Blobel G (1999) Cell cycle-regulated attachment of the ubiquitin-related protein SUMO to the yeast septins. J Cell Biol 147(5):981–994
Lee JM, Greenleaf AL (1991) CTD kinase large subunit is encoded by CTK1, a gene required for normal growth of Saccharomyces cerevisiae. Gene Expr 1:149–167
Liao SM, Zhang J, Jeffery DA, Koleske AJ, Thompson CM, Chao DM, Viljoen M, van Vuuren HJ, Young RA (1995) A kinase-cyclin pair in the RNA polymerase II holoenzyme. Nature 374:193–196
Lorincz AT, Reed SI (1984) Primary structure homology between the product of yeast cell division control gene CDC28 and vertebrate oncogenes. Nature 307:183–185
McQuilken M, Jentzsch MS, Verma A, Mehta SB, Oldenbourg R, Gladfelter AS (2017) Analysis of Septin reorganization at cytokinesis using polarized fluorescence microscopy. Front Cell Dev Biol 5:42. https://doi.org/10.3389/fcell.2017.00042
Meseroll RA, Howard L, Gladfelter AS (2012) Septin ring size scaling and dynamics require the coiled-coil region of Shs1p. Mol Biol Cell 23:3391–3406. https://doi.org/10.1091/mbc.E12-03-0207
Nigg EA (1993) Cellular substrates of p34(cdc2) and its companion cyclin-dependent kinases. Trends Cell Biol 3:296–301
Patasi C, Godočíková J, Michlíková S, Nie Y, Káčeriková R, Kválová K, Raunser S, Farkašovský M (2015) The role of Bni5 in the regulation of septin higher-order structure formation. Biol Chem 396:1325–1337. https://doi.org/10.1515/hsz-2015-0165
Perez AM, Finnigan GC, Roelants FM, Thorner J (2016) Septin-associated protein kinases in the yeast Saccharomyces cerevisiae. Front Cell Dev Biol 4:119. https://doi.org/10.3389/fcell.2016.00119
Qiu W, Neo SP, Yu X, Cai M (2008) A novel septin-associated protein, Syp1p,is required for normal cell cycle-dependent septin cytoskeleton dynamics in yeast. Genetics 180:1445–1457. https://doi.org/10.1534/genetics.108.091900
Rueden CT, Schindelin J, Hiner MC, DeZonia BE, Walter AE, Arena ET, Eliceiri KW (2017) ImageJ2: ImageJ for the next generation of scientific image data. BMC Bioinf 18:529. https://doi.org/10.1186/s12859-017-1934-z
Sadian Y, Gatsogiannis C, Patasi C, Hofnagel O, Goody RS, Farkasovsky M, Raunser S (2013) The role of Cdc42 and Gic1 in the regulation of septin filament formation and dissociation. eLife 2:e01085. https://doi.org/10.7554/eLife.01085
Simon M, Seraphin B, Faye G (1986) KIN28, yeast split gene coding for a putative protein kinase homologous to CDC28. EMBO J 5:2697–2701
Sinha I, Wang YM, Philp R, Li CR, Yap WH, Wang Y (2007) Cyclin-dependent kinases control septin phosphorylation in Candida albicans hyphal development. Dev Cell 13:421–432. https://doi.org/10.1016/j.devcel.2007.06.011
Smith MB, Li H, Shen T, Huang X, Yusuf E, Vavylonis D (2010) Segmentation and tracking of cytoskeletal filaments using open active contours. Cytoskeleton 67:693–705. https://doi.org/10.1002/cm.20481
Swaney DL, Beltrao P, Starita L, Guo A, Rush J, Fields S, Krogan NJ, Villén J (2013) Global analysis of phosphorylation and ubiquitylation cross-talk in protein degradation. Nat Methods 10:676–682. https://doi.org/10.1038/nmeth.2519
Tang CS, Reed SI (2002) Phosphorylation of the septin cdc3 in G1 by the cdc28 kinase is essential for efficient septin ring disassembly. Cell Cycle 1:42–49
Versele M, Thorner J (2004) Septin collar formation in budding yeast requires GTP binding and direct phosphorylation by the PAK, Cla4. J Cell Biol 164:701–715. https://doi.org/10.1083/jcb.200312070
Yao S, Neiman A, Prelich G (2000) BUR1 and BUR2 encode a divergent cyclin-dependent kinase-cyclin complex important for transcription in vivo. Mol Cell Biol 20:7080–7087
Acknowledgments
We gratefully acknowledge financial support from the Humboldt Foundation 3.4 - 1006989 - SVK - IP (MF and SR) and VEGA (Vedecká Grantová Agentúra, Ministerstvo Školstva Slovenskej Republiky) Grant 2/0002/15 (MF). We thank Jacob Bauer for helpful discussions and critical reading of the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethical approval
All of the experiments complied with the current Slovak law.
Conflict of interest
The authors declare no conflict of interest.
Electronic supplementary material
ESM 1
(PDF 1282 kb)
Rights and permissions
About this article
Cite this article
Káčeriková, R., Godočíková, J., Wang, Z. et al. Modulation of septin higher-order structure by the Cdc28 protein kinase. Biologia 73, 1025–1033 (2018). https://doi.org/10.2478/s11756-018-0116-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.2478/s11756-018-0116-4