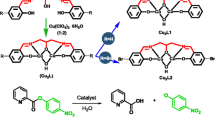
Abstract
A new metallomicellar system containing cerium(III), a macrocylic polyamine ligand, and the nonionic surfactant Brij35(polyoxyethylene(23) lauryl ether) was prepared and used as a catalyst in the hydrolysis of bis(4-nitrophenyl) phosphate (BNPP). Catalytic rate of the BNPP hydrolysis was measured kinetically using the UV-VIS spectrophotometric method. The results indicate that the metallomicellar system has relatively high stability and excellent catalytic function in the BNPP hydrolysis; also, the reaction rate of the BNPP catalytic hydrolysis increased by a factor of ca. 1 × 1010 compared to the BNPP spontaneous hydrolysis due to the catalytic effect of the active species and the local concentration effect of the micelles in the metallomicellar system. Experimental results also showed that the mono-hydroxy complex containing the macrocyclic polyamine ligand and cerium(III) is the real active species in the BNPP catalytic hydrolysis, and that the micelles provide a useful catalytic environment for the reaction. On basis of the research results, the reaction mechanism of BNPP catalytic hydrolysis has been proposed.
Similar content being viewed by others
References
Anbu, S., Kandaswamy, M., & Varghese, B. (2010). Structural, electrochemical, phosphate-hydrolysis, DNA binding and cleavage studies of new macrocyclic binuclear nickel(II) complexes. Dalton Transactions, 39, 3823–3832. DOI: 10.1039/b923078e.
Daniel, R. M. (1996). The upper limits of enzyme thermal stability. Enzyme and Microbial Technology, 19, 74–79. DOI: 10.1016/0141-0229(95)00174-3.
Douglas, B. E. (1983). Inorganic synthesis (pp. 16). Beijing, China: Sceince Publishing Company.
Ferreira, D. E. C., De Almeida, W. B., Neves, A., & Rocha, W. R. (2008). Theoretical investigation of the reaction mechanism for the phosphate diester hydrolysis using an asymmetric dinuclear metal complex as a biomimetic model of the purple acid phosphatase enzyme. Physical Chemistry Chemical Physics, 10, 7039–7046. DOI: 10.1039/b809189g.
Franklin, S. J. (2001). Lanthanide-mediated DNA hydrolysis. Current Opinion in Chemical Biology, 5, 201–208. DOI: 10.1016/s1367-5931(00)00191-5.
Gunnlaugsson, T., O’Brien, J. E., & Mulready, S. (2002). Glycine-alanine conjugated macrocyclic lanthanide ion complexes as artificial ribonucleases. Tetrahedron Letters, 43, 8493–8497. DOI:10.1016/s0040-4039 (02)02071-3.
Iranzo, O., Kovalevsky, A. Y., Morow, J. R., & Richard, J. P. (2003). Physical and kinetic analysis of the cooperative role of metal ions in catalysis of phosphodiester cleavage by a dinuclear Zn(II) complex. Journal of the American Chemical Society, 125, 1988–1993. DOI: 10.1021/ja027728v.
Jiang, F. B., Jiang, B. Y., Chen, Y., Yu, X. Q., & Zeng, X. C. (2004). Metallomicellar catalysis: effects of bridge-connecting ligands on the hydrolysis of PNPP catalyzed by Cu(II) complexes of ethoxyl-diamine ligands in micellar solution. Journal of Molecular Catalysis A: Chemical, 210, 9–16. DOI:10.1016/j.molcata.2003.07.019.
Jiang, W. D., Xu, B., Lin, Q., Li, J. Z., Liu, F. A., Zeng, X. C., & Chen, H. (2008). Metal-promoted hydrolysis of bis(p-nitrophenyl)phosphate by trivalent manganese complexes with Schiff base ligands in Gemini micellar solution. Colloids and Surfaces A: Physicochemical and Engineering Aspects, 315, 103–109. DOI:10.1016/j.colsurfa.2007.07.018.
Jones, D. R., Lindoy, L. F., & Sargeson, A. M. (1983). Hydrolysis of phosphate esters bound to cobalt(III). Kinetics and mechanism of intramolecular attack of hydroxide on coordinated 4-nitrophenyl phosphate. Journal of the American Chemical Society, 105, 7327–7336. DOI: 10.1021/ja00363a021.
Jurek, P. E., Jurek, A. M., & Martell, A. E. (2000). Phosphate diester hydrolysis by mono- and dinuclear lanthanum complexes with an unusual third-order dependence. Inorganic Chemistry, 39, 1016–1020. DOI: 10.1021/ic9906961.
Katada, H., Seino, H., Mizobe, Y., Sumaoka, J., & Komiyama, M. (2008). Crystal structure of Ce((IV)/dipicolinate complex as catalyst for DNA hydrolysis. Journal of Biological Inorganic Chemistry, 13, 249–255. DOI: 10.1007/s00775-007-0315-x.
Komiyama, M., Takeda, N., & Shigekawa, H. (1999). Hydrolysis of DNA and RNA by lanthanide ions: mechanistic studies leading to new applications. Chemical Communications, 1999, 1443–1451. DOI: 10.1039/a901621j.
Kuchma, M. H., Komanski, C. B., Colon, J., Teblum, A., Masunov, A. E., Alvarado, B., Babu, S., Seal, S., Summy, J., & Baker, C. H. (2010). Phosphate ester hydrolysis of biologically relevant molecules by cerium oxide nanoparticles. Nanomedicine: Nanotechnology, Biology and Medicine, 6, 738–744. DOI:10.1016/j.nano.2010.05.004.
Li, J. Z., Xie, J. Q., Li, S. X., Zeng, W., Zeng, X. C., & Qin, S. Y (2005). Study of the BNPP hydrolysis catalyzed by the Schiff base magnesium(III) complexes bearing polyether side chains in the micelle solution. Acta Chimica Sinica, 63, 114–120.
Lombardo, V., Bonomi, R., Sissi, C., & Mancin, F. (2010). Phosphate diesters and DNA hydrolysis by dinuclear Zn(II) complexes featuring a disulfide bridge and H-bond donors. Tetrahedron, 66, 2189–2195. DOI:10.1016/j.tet.2010.01.050.
Luedtke, N. W., & Schepartz, A. (2005). Lanthanide-mediated phosphoester hydrolysis and phosphate elimination from phosphopeptides. Chemical Communications, 2005, 5426–5428. DOI: 10.1039/b510123a.
Maldonado, A. L., & Yatsimirsky, A. K. (2005). Kinetics of phosphodiester cleavage by differently generated cerium(IV) hydroxo species in neutral solutions. Organic & Biomolecular Chemistry, 3, 2859–2867. DOI: 10.1039/b506170a.
Mancin, F., & Tecilla, P. N. (2007). Zinc(II) complexes as hydrolytic catalysts of phosphate diester cleavage: from model substrates to nucleic acids. New Journal of Chemistry, 31, 800–817. DOI: 10.1039/b703556j.
Patel, M. N., Patel, S. H., & Pansuriya, P. B. (2011). DNA binding and cleavage by dinuclear nickel(II) complexes with neutral bidentate ligands and ciprofloxacin. Medicinal Chemistry Research, 20, 1371–1384. DOI: 10.1007/s00044-010-9486-z.
Rammo, J., Hettich, R., Roigk, A., & Schneider, H. J. (1996). Catalysis of DNA cleavage by lanthanide complexes with nucleophilic or intercalating ligands and their kinetic characterization. Chemical Communications, 1996, 105–107. DOI: 10.1039/cc9960000105.
Rossi, L. M., Neves, A., Hörner, R., Terenzi, H., Szpoganicz, B., & Sugai, J. (2002). Hydrolytic activity of a dinuclear copper(II,II) complex in phosphate diester and DNA cleavage. Inorganica Chimica Acta, 337, 366–370. DOI: 10.1016/s0020-1693(02)01111-8.
Taşcioğlu, S. (1996). Micellar solutions as reaction media. Tetrahedron, 52, 11113–11152. DOI: 10.1016/0040-4020(96)00669-2.
Tjioe, L., Joshi, T., Forsyth, C. M., Moubaraki, B., Murray, K. S., Brugger, J., Graham, B., & Spiccia, L. (2012). Phosphodiester cleavage properties of copper(II) complexes of 1,4,7-triazacyclononane ligands bearing single alkyl guanidine pendants. Inorganic Chemistry, 51, 939–953. DOI:10.1021/ic2019814.
Tonde, S. S., Kumbhar, A. S., Padhye, S. B., & Butcher, R. J. (2006). Self-activating nuclease activity of copper (II) complexes of hydroxyl-rich ligands. Journal of Inorganic Biochemistry, 100, 51–57. DOI:10.1016/j.jinorgbio.2005.09.014.
Wang, H. T., Hu, T. T., Zhang, Q. L., Liu, J. H., Ren, X. Z., Li, C. H., Wang, F., & Zhang, P. X. (2008). Synthesis, DNA-binding and luminescent properties of polypyridyl ruthenium(II) complex. Acta Chimica Sinica, 66, 1565–1571.
Westheimer, F. H. (1987). Why nature chose phosphates. Science, 235, 1173–1178. DOI: 10.1126/science.2434996.
Xiang, Y., Jiang, B. Y., Zeng, X. C., & Xie, J. Q. (2002). Metallomicellar catalysis: Catalytic cleavage of p-nitrophenyl picolinate by Cu2+ complex of 4-chloride-2,6-bis(N-hydroxyethylaminomethyl)-benzophenol in micellar solution. Journal of Colloid and Interface Science, 247, 366–371. DOI:10.1006/jcis.2001.8069.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Xie, JQ., Li, C., Wang, M. et al. Preparation of a new metallomicelle catalyst for the hydrolysis of bis(4-nitrophenyl) phosphate. Chem. Pap. 67, 365–371 (2013). https://doi.org/10.2478/s11696-012-0281-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.2478/s11696-012-0281-9