Abstract
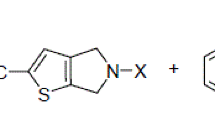
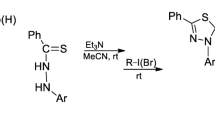
The reaction of propanedioic acid, 2-diazo-1,3-bis(1,1-dimethylethyl) ester (di-tert-butyl diazomalonate) with a series of cyclopenta[b]thiophenes in the presence of catalytic rhodium acetate was studied. The resulting S—C ylides underwent a rearrangement to form a heterocycle with different topology; thialene, in very low yields. Experimental and spectral data for all compounds are provided.
Similar content being viewed by others
References
Bonini, B. F., Capito, E., Comes-Franchini, M., Ricci, A., Bottoni, A., Bernardi, F., Miscione, G. P., Giordano, L., & Cowley, A. R. (2004). Diastereoselective synthesis of thieno[3′,2′:4,5]cyclopenta[1,2-d][1,3]oxazolines t new ligands fot the copper-catalyzed asymmetric conjugate addition of diethylzinc to enones. European Journal of Organic Chemistry, 2004, 4442–4451. DOI: 10.1002/ejoc.200400351.
Bowles, T., Jones, R., Porter, A. E. A., Rechka, J. A., Rzepa, H. S., & Williams, D. J. (1985). 2H-thiopyrans as intermediates in the rearrangement of thiophenium bisalkoycarbonylmethylides to thiophene-2-malonates; the crystal and molecular structure of 2,2-bis-t-butoxycarbonyl-5-bromo-2Hthiopyran and 2,2-bis-t-butoxycarbonyl-6-bromo-2H-thiopyran. Journal of the Chemical Society, Chemical Communications, 1985, 1590–1592. DOI: 10.1039/c39850001590.
Bowles, T., Gillespie, R. J., Porter, A. E. A., Rechka, J. A., & Rzepa, H. S. (1988a). Thermally induced rearrangement of thiopheniobis(alkoxycarbonyl)methanides. Journal of the Chemical Society, Perkin Transactions 1, 1988, 803–807. DOI: 10.1039/p19880000803.
Bowles, T., Jones, R., Porter, A. E. A., Rechka, J. A., Rzepa, H. S., & Williams, D. J. (1988b). Synthesis of some thiophenium bis(t-butoxycarbonyl)methylides. Journal of the Chemical Society, Perkin Transactions 1, 1988, 1023–1027. DOI: 10.1039/p19880001023.
Ito, Y., Borecka, B., Olovsson, G., Trotter, J., & Scheffer, J. R. (1995). Control of the solid-state photodimerization of some derivatives and analogs of trans-cinnamic acid by ethylenediamine. Tetrahedron Letters, 36, 6087–6090. DOI: 10.1016/0040-4039(95)01210-9.
Klein, R. F. X., & Horák, V. (1986). New synthesis and spectroscopic studies of thialene (cyclopenta[b]thiapyran). Journal of Organic Chemistry, 51, 4644–4651. DOI: 10.1021/jo00374a027.
Mayer, R., Franke, J., Horák, V., Hanker, I., & ZahradnÍk, V. (1961). Synthese und Eigenschaften des Thialens (Cyclopenta-Thiapyran). Tetrahedron Letters, 2, 289–294. DOI: 10.1016/s0040-4039(01)84064-8.
Mayer, R., & Franke, J. (1965). Schwefel-Helerocyclen. XLII. Eine weitere Synthese von Thialen (Cyclopenta[b]thiopyran) und die erste Darstellung des Benzothialens Indeno[2,1-b] thiopyran. Journal für Praktische Chemie, 30, 262–272. DOI: 10.1002/prac.19650300505.
Machara, A., Kurfürst, M., KozmÍk, V., Petříčková H., Dvořáková, H., & Svoboda, J. (2004). A nonconcerted cycloaddition of fused 2-vinylthiophenes with dimethyl acetylenedicarboxylate. Tetrahedron Letters, 45, 2189–2192 DOI: 10.1016/j.tetlet.2004.01.030.
Machara, A., KozmÍk, V., Pojarová, M., Dvořáková, H., & Svoboda, J. (2009). Preparation and rearrangement study of novel thiophenium- and selenophenium-ylides. Collection of Czechoslovak Chemical Communications, 74, 785–798. DOI: 10.1135/cccc2009001.
MacKenzie, N. E., & Thomson, R. H. (1982). Ring contractions of thiochroman-4-ones and thiochromen-4-ones. Journal of the Chemical Society, Perkin Transactions 1, 1982, 395–402. DOI: 10.1039/p19820000395.
Mohamadi, F., Spees, M. M., & Grindey, G. B. (1992). Sulfonylureas: a new class of cancer chemotherapeutic agents. Journal of Medicinal Chemistry, 35, 3012–3016. DOI: 10.1021/jm00094a013.
Poirier, Y., & Lozach, N. (1966). Heterocyclic sulfur-compounds. XXII. Sulfuration of 2-alkylidene-1-indanones and 2-arylmethylene-1-indanones. Bulletin de la Societe Chimique de France, 1966, 1062–1068.
Porter, A. E. A. (1989). The chemistry of thiophenium salts and thiophenium ylids. Advances in Heterocyclic Chemistry, 45, 151–184. DOI: 10.1016/s0065-2725(08)60330-x.
Sam, J., & Thompson, A. C. (1963). Thiaindanones. Thiophene isosteres of indanone. Journal of Pharmaceutical Sciences, 52, 898–901. DOI: 10.1002/jps.2600520918.
Smith, D. G. (1990). 4-Amido-3,4-dihydro-2H-1-benzothiopyran-3-ols and their sulphoxide and sulphone derivatives — cromakalim analogues. Journal of the Chemical Society, Perkin Transactions 1, 1990, 3187–3191. DOI: 10.1039/p19900003187.
Stuckwisch, C. G., & Bailey, J. V. (1963). Reaction of organometallic compounds with propiolactone. The Journal of Organic Chemistry, 28, 2362–2363. DOI: 10.1021/jo01044a050.
Vuorinen, E., Chalmers, A. A., Dillen, J. L. M., & Modro, T. A. (1991). Benzo[b]thiophenium S,C-ylides: Preparation, structure and comparison with thiophenium analogues. Tetrahedron, 47, 8611–8620. DOI: 10.1016/s0040-4020(01)82404-2.
Yuen, P.W. (2004). U.S. Patent No. 6710190 Washington, D.C., USA: U.S. Patent and Trademark Office.
Author information
Authors and Affiliations
Corresponding author
Additional information
Dedicated to Professor Štefan Toma on the occasion of his 75th birthday
Rights and permissions
About this article
Cite this article
Machara, A., Svoboda, J. Thiophenium-ylides: Synthesis and reactivity. Chem. Pap. 67, 59–65 (2013). https://doi.org/10.2478/s11696-012-0222-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.2478/s11696-012-0222-7