Abstract
New 2-substituted diazaphospholane-2-oxides (I-III, V-VIII) and diazaphosphorinane-2-oxide (IV) were synthesised and characterised by 1H, 13C, and 31P NMR, IR spectroscopy, and elemental analysis. The presence of chiral diamino groups in compounds II and V–VIII gives rise to various diastereomers so that the 31P{1H} NMR spectra demonstrated three and two peaks with different ratios, respectively. Also, the 1H and 13C{1H} NMR spectra of compounds II and V–VIII revealed three and two sets of signals for the related conformers (diastereomers). Interestingly, the 31P NMR spectrum of V in D2O indicated a great upfield shift (Δδ = 19.0) for 31P relative to the value obtained in DMSO-d6 (solvent effect). The two signals in V split further to three signals in the presence of β-cyclodextrin. Moreover, conformational analysis of diazaphospholane V was studied by ab initio calculations at the HF and B3LYP levels of theory using the Gaussian 98 program. Results indicated that among four suggested diastereomers (C1–C4) of V, C1 and C3 containing methyl group in the equatorial position are the most stable forms.
Similar content being viewed by others
References
Amirkhanov, V. M., Ovchynnikov, V. A., Glowiak, T., & Kozlowski, H. (1997). Crystal and molecular structures of N,N′-diphenyl-N″-trichloroacetyl-phosphorictriamide and N,N′-tetraethyl-N″-benzoylphosphorictriamide. The effect of various substituents on the structural parameters of the [C(O)N(H)P(O)] moiety. Zeitschrift für Naturforschung B - A Journal of Chemical Sciences, 52, 1331–1336.
Berlicki, Ł., Rudzińska, E., & Kafarski, P. (2003). Enantiodifferentiation of aminophosphonic and aminophosphinic acids with α- and β-cyclodextrins. Tetrahedron: Asymmetry, 14, 1535–1539. DOI: 10.1016/S0957-4166(03)00273-8.
Corbridge, D. E. C. (1995). Phosphorus: An outline of its chemistry, biochemistry, and technology (5th ed., pp. 55, 57). Amsterdam, The Netherlands: Elsevier.
De la Cruz, A., Koeller, K. J., Rath, N. P., Spilling, C. D., & Vasconcelos, I. C. F. (1998). The synthesis, structure and properties of diazaphospholes: Reagents and ligands for asymmetric synthesis. Tetrahedron, 54, 10513–10524. DOI: 10.1016/S0040-4020(98)00502-X.
Denmark, S. E., Miller, P. C., & Wilson, S. R. (1991). Configuration, conformation, and colligative properties of a phosphorus-stabilized anion. Journal of the American Chemical Society, 113, 1468–1470. DOI: 10.1021/ja00004a089.
Devillers, J., Navech, J., & Albrand, J.-P. (1971). Heterocycles contenant du phosphore—V. Analyse des spectres de resonance magnetique protonique des oxo-2 methoxy ou phénoxy-2, methyl-3, oxazaphospholanes-1,3,2: Exemples de spectres du type ABXY. Organic Magnetic Resonance, 3, 177–186. DOI: 10.1002/mrc.1270030203.
Du, H., Zhao, B., & Shi, Y. (2008). Catalytic asymmetric allylic and homoallylic diamination of terminal olefins via formal C-H activation. Journal of the American Chemical Society, 130, 8590–8591. DOI: 10.1021/ja8027394.
Dutasta, J. P., Esteban-Calderon, C., Tinant, B., & Declercq, J.-P. (1990). Structure of bis(1,3-di-o-anisyl-2-thioxo-1,3,2λ 5-diazaphosphorinan-2-yl) oxide. Acta Crystallographica Section C: Crystal Structure Communications, C46, 68–71. DOI: 10.1107/S0108270189005172.
Dutasta, J. P., Grand, A., Guimares, A. C., & Robert, J. B. (1979). Dioxaphospholanes-1,3,2, dimerisation, obtention de composes heterocycliques phosphores et oxygenes a dix chainons: tetra-oxa-1,3,6,8 diphosphecanes-2. Tetrahedron, 35, 197–207. DOI: 10.1016/S0040-4020(01)99482-7.
Gholivand, K., Oroujzadeh, N., Erben, M. F., & Della Védova, C. O. (2009a). Synthesis, spectroscopy, computational study and prospective biological activity of two novel 1,3,2-diazaphospholidine-2,4,5-triones. Polyhedron, 28, 541–547. DOI: 10.1016/j.poly.2008.11.024.
Gholivand, K., Pourayoubi, M., Farshadian, S., Molani, S., & Shariatinia, Z. (2005a). Synthesis and crystal structure of 5,5-dimethyl-2-(p-methylanilino)-2-oxo-1,3,2-diazaphosphorinane. Analytical Sciences: X-ray Structure Analysis Online, 21, x55–x56.
Gholivand, K., Pourayoubi, M., & Shariatinia, Z. (2007a). 2,3J(P,X) [X = H, C] coupling constants dependency to the ring size, hybridization and substituents in new diazaphospholes and diazaphosphorinanes, NMR and X-ray crystallography studies. Polyhedron, 26, 837–844. DOI: 10.1016/j.poly.2006.09.092.
Gholivand, K., Shariatinia, Z., Afshar, F., Faramarzpour, H., & Yaghmaian, F. (2007b). New 1,3,2-diazaphosphorinanes; syntheses, spectroscopic characterization, X-ray crystallography and ab initio calculations. Main Group Chemistry, 6, 231–248. DOI: 10.1080/10241220801994742.
Gholivand, K., Shariatinia, Z., Ansar, S., Mashhadi, S. M., & Daeepour, F. (2009b). The first naphthodiazaphosphorinane in the solid phase; syntheses, spectroscopic studies and X-ray crystallography of some new 1,3,2-diheterophosphorus compounds. Structural Chemistry, 20, 481–488. DOI: 10.1007/s11224-009-9445-9.
Gholivand, K., Shariatinia, Z., Mahzouni, H. R., & Amiri, S. (2007c). Phosphorus heterocycles: synthesis, spectroscopic study and X-ray crystallography of some new diazaphosphorinanes. Structural Chemistry, 18, 653–660. DOI: 10.1007/s11224-007-9197-3.
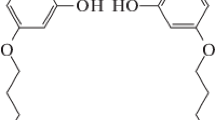
Gholivand, K., Shariatinia, Z., Pourayoubi, M., & Farshadian, S. (2005b). Syntheses and spectroscopic study of some new diazaphospholes and diazaphosphorinanes. Crystal structure of
 . Zeitschrift für Naturforschung B - A Journal of Chemical Sciences, 60, 1021–1026.
. Zeitschrift für Naturforschung B - A Journal of Chemical Sciences, 60, 1021–1026.Gholivand, K., Shariatinia, Z., Yaghmaian, F., & Faramarzpour, H. (2006). Substituent effects on the spectroscopic and structural parameters of several new 1,3,2-diazaphosphorinanes. Syntheses, spectroscopic characterization, and X-ray crystallography. Bulletin of the Chemical Society of Japan, 79, 1604–1606.
Hall, C. R., Inch, T. D., Pottage, C., Williams, N. E., Campbell, M. M., & Kerr, P. F. (1983). Use of carbohydrate derivatives for studies of phosphorus stereochemistry. Part 8. Preparation and some reactions of 1,3,2-oxazaphospholidine-2-ones and -2-thiones derived from 2-deoxy-3,4,6-tri-O-methyl-2-methylamino-D-glucopyranose. Journal of the Chemical Society, Perkin Transactions 1, 1983, 1967–1975. DOI: 10.1039/P19830001967.
Khaikin, L. S., Grikina, O. E., Vilkov, L. V., & Boggs, J.E. (1988). Structure of 1,2,3-diazaphosphole and several of its derivatives. Use of the results of nonempirical quantum chemical calculations in the electron diffraction investigation of 2-acetyl-5-methyl- and 5-methyl-2-phenyl-1,2,3-diazaphospholes. Journal of Structural Chemistry, 28, 607–611. DOI: 10.1007/BF00749603.
Kim, H., Nguyen, Y., Yen, C. P.-H., Chagal, L., Lough, A.J., Kim, B. M., & Chin, J. (2008). Stereospecific synthesis of C 2 symmetric diamines from the mother diamine by resonance-assisted hydrogen-bond directed diaza-Cope rearrangement. Journal of the American Chemical Society, 130, 12184–12191. DOI: 10.1021/ja803951u.
Kranz, M., Denmark, S. E., Swiss, K. A., & Wilson, S. R. (1996). An ab initio study of the P-C bond rotation in phosphoryl- and thiophosphoryl-stabilized carbanions: Fiveand six-membered heterocycles. The Journal of Organic Chemistry, 61, 8551–8563. DOI: 10.1021/jo9602783.
Lucet, D., Le Gall, T., & Mioskowski, C. (1998). The chemistry of vicinal diamines. Angewandte Chemie International Edition, 37, 2580–2627. DOI: 10.1002/(SICI)1521-3773(19981016)37:19<2580::AID-ANIE2580>3.0.CO;2-L.
Nielsen, J., & Dahl, O. (1984). Stereochemistry of substitution at trico-ordinate phosphorus. Journal of the Chemical Society, Perkin Transactions 2, 1984, 553–558. DOI: 10.1039/P29840000553.
Nifantiev, E. E., Sorokina, S. F., Borisenko, A. A., Zavalishina, A. I., & Vorobjeva, L. A. (1981). Cyclic organic derivatives of hypophosphorous acid. Tetrahedron, 37, 3183–3194. DOI: 10.1016/S0040-4020(01)98852-0.
Olson, D. E., & Du Bois, J. (2008). Catalytic C-H amination for the preparation of substituted 1,2-diamines. Journal of the American Chemical Society, 130, 11248–11249. DOI: 10.1021/ja803344v.
Parker, D. (1991). NMR determination of enantiomeric purity. Chemical Reviews, 91, 1441–1457. DOI: 10.1021/cr00007a009.
Peyronel, J. F., Samuel, O., & Fiaud, J. C. (1987). New chiral bicyclic phosphoramides derived from (L)-glutamic acid. The Journal of Organic Chemistry, 52, 5320–5325. DOI: 10.1021/jo00233a004.
Saibabu Kotti, S. R. S., Timmons, C., & Li, G. (2006). Vicinal diamino functionalities as privileged structural elements in biologically active compounds and exploitation of their synthetic chemistry. Chemical Biology & Drug Design, 67, 101–114. DOI: 10.1111/j.1747-0285.2006.00347.x.
Setzer, W. N., Black, B. G., Hovanes, B. A., & Hubbard, J. L. (1989). Conformational analysis of 1,3,2-oxazaphospholanes derived from ephedrine and pseudoephedrine. The Journal of Organic Chemistry, 54, 1709–1713. DOI: 10.1021/jo00268a038.
Topacli, C., & Topacli, A. (2003). Ab initio calculations and vibrational structure of sulfanilamide. Journal of Molecular Structure, 644, 145–150. DOI: 10.1016/S0022-2860(02)00473-8.
Varghese, H. T., Panicker, C. Y., & Philip, D. (2006). Vibrational spectroscopic studies and ab initio calculations of sulfanilamide. Spectrochimica Acta Part A, 65, 155–158. DOI: 10.1016/j.saa.2005.09.040.
Viso, A., Fernández de la Pradilla, R., García, A., & Flores, A. (2005). α,β-Diamino acids: Biological significance and synthetic approaches. Chemical Reviews, 105, 3167–3196. DOI: 10.1021/cr0406561.
Wang, B., Du, H. F., & Shi, Y. (2008). A palladiumcatalyzed dehydrogenative diamination of terminal olefins. Angewandte Chemie International Edition, 47, 8224–8227. DOI: 10.1002/anie.200803184.
Warnat, K. (1941). Sulphanilamide phosphoric acid derivative and process for the manufacture thereof. U.S. Patent No.2245539. Washington, D.C., USA: U.S. Patent and Trademark Office.
Zalán, Z., Martinek, T. A., Lázár, L., & Fülöp, F. (2003). Synthesis and conformational analysis of 1,3,2-diazaphosphorino [6,1-a]isoquinolines, a new ring system. Tetrahedron, 59, 9117–9125. DOI: 10.1016/j.tet.2003.09.062.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Gholivand, K., Ghaziani, F. Synthesis, spectroscopic and configurational study, and ab initio calculations of new diazaphospholanes. Chem. Pap. 65, 691–699 (2011). https://doi.org/10.2478/s11696-011-0047-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.2478/s11696-011-0047-9




 . Zeitschrift für Naturforschung B - A Journal of Chemical Sciences, 60, 1021–1026.
. Zeitschrift für Naturforschung B - A Journal of Chemical Sciences, 60, 1021–1026.