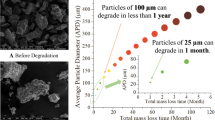
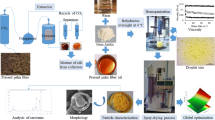
Abstract
Melt dispersion technique was investigated for carnauba wax microparticles production. Microbeads with spherical shape and narrow size distribution were produced. The main objective of this study was to investigate the effect of significant process variables (initial wax concentration, stirring speed, stirring time, and surfactants) on sphericity, size distribution, and morphological properties of wax microparticles. Optimal conditions were evaluated on the basis of particle size distribution and visual analysis. Surface morphology of microparticles was characterized by scanning electron microscopy (SEM). Effects of process conditions on the size distribution of particles were evaluated by sieve analysis. Main purpose of these investigations was to apply optimized parameters to aroma encapsulation for their use in food and feed industry.
Similar content being viewed by others
References
Albertini, B., Passerini, N., Pattarino, F., & Rodriguez, L. (2008). New spray congealing atomizer for the microencapsulation of highly concentrated solid and liquid substances. European Journal of Pharmaceutics and Biopharmaceutics, 69, 348–357. DOI: 10.1016/j.ejpb.2007.09.011.
Barakat, N. S., & Yassin, A. B. E. (2006). In vitro characterization of carbamazepine-loaded precifac lipospheres. Drug Delivery, 13, 95–104. DOI: 10.1080/10717540500313661.
Card, Q. P., Deerfield, N. K., & Palatine, S. (1960). Flavor premixes for animal feeds. U. S. Patent No. 2,921,853. U.S. Patent Office.
Cheboyina, S., & Wyandt, C. M. (2008). Wax-based sustained release matrix pellets prepared by a novel freeze pelletization technique: II. In vitro drug release studies and release mechanisms. International Journal of Pharmaceutics, 359, 167–173. DOI: 10.1016/j.ijpharm.2008.04.001.
Curle, N., & Davies, H. J. (1968). Modern fluid dynamics (Vol. 1). London, UK: Van Nostrand.
Donhowe, I. G., & Fennema, O. (1993). Water vapor and oxygen permeability of wax films. Journal of the American Oil Chemists’ Society, 70, 867–873. DOI: 10.1007/BF02545345.
Dunker, M. F. W. (1960). Colloids, emulsions and suspensions. In J. B. Sprowls (Ed.), American pharmacy: Textbook of pharmaceutical principles, processes and preparations (5th ed., pp. 107–148). Philadelphia, PA, USA: J. B. Lippincott Company.
Fuchs, M., Turchiuli, C., Bohin, M., Cuvelier, M. E., Ordonnaud, C., Peyrat-Maillard, M. N., & Dumoulin, E. (2006). Encapsulation of oil in powder using spray drying and fluidized bed agglomeration. Journal of Food Engineering, 75, 27–35. DOI: 10.1016/j.jfoodeng.2005.03.047.
Gowda, D. V., & Shivakumar, H. G. (2007). Preparation and evaluation of waxes/fat microspheres loaded with lithium carbonate for controlled release. Indian Journal of Pharmaceutical Sciences, 69, 251–256. DOI: 10.4103/0250-474X.33152.
Hinze, J. O. (1955). Fundamentals of the hydrodynamic mechanism of splitting in dispersion process. AIChE Journal, 1, 289–295. DOI: 10.1002/aic.690010303.
Jenning, V., & Gohla, S. (2000). Comparison of wax and glyceride solid lipid nanoparticles (SLN®). International Journal of Pharmaceutics, 196, 219–222. DOI: 10.1016/S0378-5173(99)00426-3.
Holmberg, K., Jönsson, B., Kronberg, B., & Lindman, B. (2003). Surfactants and polymers in aqueous solution (2nd ed., p. 463). Etobicoke, ON, Canada: Wiley. DOI: 10.1002/0470856424.ch21.
Kamble, R., Maheshwari, M., Paradkar, A., & Kadam, S. (2004). Melt solidification technique: Incorporation of higher wax content in ibuprofen beads. AAPS PharmSciTech, 5(4), 75–83. DOI: 10.1208/pt050461.
Katona, J. M., Sovilj, V. J., Petrović, L. B., & Mucić, N. Z. (2010). Tensiometric investigation of the interaction and phase separation in a polymer mixture-ionic surfactant ternary system. Journal of the Serbian Chemical Society, 75, 823–831. DOI: 10.2298/JSC100112056K.
Kolmogorov, A. N. (1949). On the disintegration of drops in turbulent flow. Doklady Akademii Nauk SSSR, 66, 825–828.
McClements, D. J. (1999). Food emulsions: Principles, practice, and techniques (2nd ed.). Boca Raton, FL, USA: CRC Press.
Media Cybernetics, Inc. (2007). Image Pro plus v. 6.2. Bethesda, MD, USA: Media Cybernetcs, Inc.
Mellema, M., Van Benthum, W. A. J., Boer, B., Von Harras, J., & Visser, A. (2006). Wax encapsulation of water-soluble compounds for application in foods. Journal of Microencapsulation, 23, 729–740. DOI: 10.1080/02652040600787900.
Milanovic, J., Manojlovic, V., Levic, S., Rajic, N., Nedovic, V., & Bugarski, B. (2010). Microencapsulation of flavors in Carnauba wax. Sensors, 10, 901–912. DOI: 10.3390/s100100901.
Miyagawa, Y., Okabe, T., Yamaguchi, Y., Miyajima, M., Sato, H., & Sunada, H. (1996). Controlled-release of diclofenac sodium from wax matrix granule. International Journal of Pharmaceutics, 138, 215–224. DOI: 10.1016/0378-5173(96)04547-4.
Özyazıcı, M., Gökçe, E. H., & Ertan, G. (2006). Release and diffusional modeling of metronidazole lipid matrices. European Journal of Pharmaceutics and Biopharmaceutics, 63, 331–339. DOI: 10.1016/j.ejpb.2006.02.005.
Passerini, N., Perissutti, B., Albertini, B., Voinovich, D., Moneghini, M., & Rodriguez, L. (2003). Controlled release of verapamil hydrochloride from waxy microparticles prepared by spray congealing. Journal of Controlled Release, 88, 263–275. DOI: 10.1016/S0168-3659(03)00009-9.
Popplewell, M. L., & Porzio, A. M. (2001). Fat-coated encapsulation composition and method for preparing the same. U.S. Patent No. 6,245,366. U.S. Patent Office.
Reithmeier, H., Herrmann, J., & Göpferich, A. (2001). Development and characterization of lipid microparticles as a drug carrier for somatostatin. International Journal of Pharmaceutics, 218, 133–143. DOI: 10.1016/S0378-5173(01)00620-2.
Rodriguez, L., Passerini, N., Cavallari, C., Cini, M., Sancin, P., & Fini, A. (1999). Description and preliminary evaluation of a new ultrasonic atomizer for spray-congealing processes. International Journal of Pharmaceutics, 183, 133–143. DOI: 10.1016/S0378-5173(99)00076-9.
Singh, R., Poddar, S. S., & Chivate, A. (2007). Sintering of wax for controlling release from pellets. AAPS PharmSciTech, 8, E175–E183. DOI: 10.1208/pt0803074.
Villalobos-Hernández, J. R., & Müller-Goymann, C. C. (2007). In vitro erythemal UV-A protection factors of inorganic sunscreens distributed in aqueous media using carnauba wax-decyl oleate nanoparticles. European Journal of Pharmaceutics and Biopharmaceutics, 65, 122–125. DOI: 10.1016/j.ejpb.2006.07.013.
Villalobos-Hernández, J. R., & Müller-Goymann, C. C. (2006a). Sun protection enhancement of titanium dioxide crystals by the use of carnauba wax nanoparticles: The synergistic interaction between organic and inorganic sunscreens at nanoscale. International Journal of Pharmaceutics, 322, 161–170. DOI: 10.1016/j.ijpharm.2006.05.037.
Villalobos-Hernández, J. R., & Müller-Goymann, C. C. (2006b). Physical stability, centrifugation tests, and entrapment eficency studies of carnauba wax-decyl oleate nanoparticles used for the dispersion of inorganic sunscreens in aqueous media. European Journal of Pharmaceutics and Biopharmaceutics, 63, 115–127. DOI: 10.1016/j.ejpb.2006.01.005.
Villalobos-Hernández, J. R., & Müller-Goymann, C. C. (2005). Novel nanoparticulate carrier system based on carnauba wax and decyl oleate for the dispersion of inorganic sunscreens in aqueous media. European Journal of Pharmaceutics and Biopharmaceutics, 60, 113–122. DOI: 10.1016/j.ejpb.2004.11.002.
Wang, L., Ando, S., Ishida, Y., Ohtani, H., Tsuge, S., & Nakayama, T. (2001). Quantitative and discriminative analysis of carnauba waxes by reactive pyrolysis-GC in the presence of organic alkali using a vertical microfurnace pyrolyzer. Journal of Analytical and Applied Pyrolysis, 58-59, 525–537. DOI: 10.1016/S0165-2370(00)00155-8.
Walstra, P., & Smudlers, I. (1997). Making emulsion and foams: an overview. In E. Dickinson, & B. Bergenstähl (Eds.), Food colloids, proteins, lipids and polysaccharides (pp. 367–381). Cambridge, UK: Royal Society of Chemistry.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Milanovic, J., Levic, S., Manojlovic, V. et al. Carnauba wax microparticles produced by melt dispersion technique. Chem. Pap. 65, 213–220 (2011). https://doi.org/10.2478/s11696-011-0001-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.2478/s11696-011-0001-x