Abstract
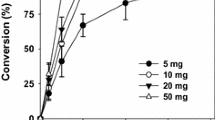
Terpene esters of fatty acids have potential applications in food, cosmetic, and pharmaceutical industries. The present study focuses on the synthesis of terpene esters of long chain fatty acids catalyzed by Candida antarctica lipase B. Different parameters like temperature, solvent, and enzyme concentration for the esterification of terpene alcohols (geraniol and citronellol) with oleic acid were studied. Maximum conversion (98 %) was found for both terpene esters at 60°C in 2,2,4-trimethylpentane as well as in dry hexane and around 95–97 % in other tested solvents. The reaction was also carried out using stearic and linoleic acid in hexane to study the effects of unsaturation in the substrate in which stearic acid showed the maximum conversion. The reaction was monitored by 1H nuclear magnetic resonance spectroscopy. Using the peak integration values of methylene protons of terpene and terpene ester of δ = 3.6 and 4.0 for citronellol and δ = 4.2 and 4.6 for geraniol, respectively, percentage conversions of each of the esters were calculated.
Similar content being viewed by others
References
Chatterjee, T., & Bhattacharyya, D. K. (1998). Synthesis of terpene esters by an immobilized lipase in a solvent-free system. Biotechnology Letters, 20, 865–868. DOI: 10.1023/A:1005315609952.
Costa Neto, P. R., Balparda Caro, M. S., Mazzuco, L. M., & Nascimento, M. G. (2004). Quantification of soybean oil ethanolysis with 1H NMR. Journal of the American Oil Chemists’ Society, 81, 1111–1114. DOI: 10.1007/s11746-004-1026-0.
Croteau, R., (1980). The biosynthesis of terpene compounds. In R. Croteau (Ed.), Fragrance and flavor substances (pp. 13–14). Pattensen, Germany: D&PS Verlag.
Fernandez-Lafuente, R. (2010). Lipase from Thermomyces lanuginosus: Uses and prospects as an industrial biocatalyst. Journal of Molecular Catalysis B: Enzymatic, 62, 197–212. DOI: 10.1016/j.molcatb.2009.11.010.
Fernandez-Lorente, G., Palomo, J. M., Cabrera, Z., Fernandez-Lafuente, R., & Guisan, J. M. (2006). Improved catalytic properties of immobilized lipases by the presence of very low concentrations of detergents in the reaction medium. Biotechnology and Bioengineering, 97, 242–250. DOI: 10.1002/bit.21230.
Gandhi, N. N. (1997). Applications of lipase. Journal of the American Oil Chemists’ Society, 74, 621–634. DOI: 10.1007/s11746-997-0194-x.
Gelbard, G., Brès, O., Vargas, R. M., Vielfaure, F., & Schuchardt, U. F. (1995). 1H Nuclear Magnetic Resonance determination of the yield of the transesterification of rapeseed oil with methanol. Journal of the American Oil Chemists’ Society, 72, 1239–1241. DOI: 10.1007/BF02540998.
Gonzalez-Navarro, H., & Braco, L. (1998). Lipase-enhanced activity in flavour ester reactions by trapping enzyme conformers in the presence of interfaces. Biotechnology and Bioengineering, 59, 122–127. DOI: 10.1002/(SICI)1097-0290(19980705)59:1〈122::AID-BIT16〉3.0.CO;2-K.
Gotor-Fernández, V., Brieva, R., & Gotor, V. (2006). Lipases: Useful biocatalysts for the preparation of pharmaceuticals. Journal of Molecular Catalysis B: Enzymatic, 40, 111–120. DOI: 10.1016/j.molcatb.2006.02.010.
Krings, U., & Berger, R. G. (1998). Biotechnological production of flavours and fragrances. Applied Microbiology and Biotechnology, 49, 1–8. DOI: 10.1007/s002530051129.
Langrand, G., Triantaphylides, C., & Baratti, J. (1988). Lipase catalyzed formation of flavor esters. Biotechnology Letters, 10, 549–554. DOI: 10.1007/BF01027127.
Mahapatra, P., Kumari, A., Garlapati, V. K., Banerjee, R., & Nag, A. (2009a). Enzymatic synthesis of fruit flavor esters by immobilized lipase from Rhizopus oligosporus optimized with response surface methodology. Journal of Molecular Catalysis B: Enzymatic, 60, 57–63. DOI: 10.1016/j.molcatb.2009.03.010.
Mahapatra, P., Kumari, A., Kumar, G. V., Banerjee, R., & Nag, A. (2009b). Kinetics of solvent-free geranyl acetate synthesis by Rhizopus oligosporus NRRL 5905 lipase immobilized on to cross-linked silica. Biocatalysis and Biotransformation, 27, 124–130. DOI: 10.1080/10242420802583366.
Mattson, F. H., & Grundy, S. M. (1985). Comparison of effects of dietary saturated, monounsaturated, and polyunsaturated fatty acids on plasma lipids and lipoproteins in man. Journal of Lipid Research, 26, 194–202.
Morgenstern, M., Cline, J., Meyer, S., & Cataldo, S. (2006). Determination of the kinetics of biodiesel production using proton Nuclear magnetic resonance spectroscopy (1H NMR). Energy & Fuels, 20, 1350–1353. DOI: 10.1021/ef0503764.
Mukherjee, K. D., & Kiewitt, I. (1998). Substrate specificity of lipases in protease preparations. Journal of Agricultural and Food Chemistry, 46, 2427–2432. DOI: 10.1021/jf971010v.
Palomo, J. M., Fernández-Lorente, G., Mateo, C., Fuentes, M., Fernández-Lafuente, R., & Guisan, J. M. (2002). Modulation of the enantioselectivity of Candida antarctica B lipase via conformational engineering: kinetic resolution of (±)-α-hydroxy-phenylacetic acid derivatives. Tetrahedron: Asymmetry, 13, 1337–1345. DOI: 10.1016/S0957-4166(02)00325-7.
Patil, D., De Leonardis, A., & Nag, A. (2010). Synthesis of biosurfactants from natural resources. Journal of Food Biochemistry. DOI: 10.1111/j.1745-4514.2010.00414.x. (In press)
Patil, D., Nag, S., Nag, A., & Basak, A. (2008). Comparison of catalytic activities between esterase and lipase in the synthesis of drugs and flavor and amide compounds. International Journal of Chemical Sciences, 6, 11–16.
Schindler, J., & Schmid, R. D. (1982). Fragrance or aroma chemicals. Microbial synthesis and enzymatic transformation - a review. Process Biochemistry, 17, 2–8.
Seo, C. W., Yamada, Y., & Okada, H. (1982). Synthesis of fatty acid esters by Corynebacterium sp. S-401. Agricultural and Biological Chemistry, 46, 405–409.
Sheldon, R. A. (1996). Large-scale enzymatic conversions in nonaqueous media. In A. M. P. Koskinen & A. M. Klibanov (Eds.), Enzymatic reactions in organic media (pp. 266–307). Glasgow, UK: Blackie Academic and Professional.
Uppenberg, J., Hansen, M. T., Patkar, S., & Jones, T. A. (1994). The sequence, crystal structure determination and refinement of two crystal forms of lipase B from Candida antarctica. Structure, 2, 293–308. DOI: 10.1016/S0969-2126(00)00031-9.
Yadav, G. D., & Lathi, P. S. (2003). Synthesis of citronellol laurate in organic media catalyzed by immobilized lipases: kinetic studies. Journal of Molecular Catalysis B: Enzymatic, 27, 113–119. DOI: 10.1016/j.molcatb.2003.10.004.
Zaks, A., & Klibanov, A. M. (1985). Enzyme-catalyzed processes in organic solvents. Proceedings of the National Academy of Sciences of the United States of America, 82, 3192–3196.
Zeng, J. Q., Zhang, Y. M., Liu, L. M., & Zhang, J. C. (2000). The continuous reaction-separation process for the lipasecatalyzed synthesis of citronellyl oleate in supercritical CO2. Chinese Journal of Organic Chemistry, 20, 195–197.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Patil, D., Das, D. & Nag, A. Enzymatic synthesis and analytical monitoring of terpene ester by 1H NMR spectroscopy. Chem. Pap. 65, 9–15 (2011). https://doi.org/10.2478/s11696-010-0077-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.2478/s11696-010-0077-8