Abstract
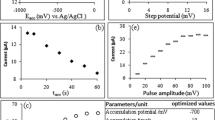
Electrochemical behavior of resazurin on HMDE in Britton-Robinson (B-R) buffers (pH 2.0–10.0) was studied using the square-wave voltammetry (SWV), square-wave adsorptive stripping voltammetry (SWAdSV), and cyclic voltammetry (CV) techniques. The voltammogram of resazurin in B-R buffer at pH < 4.0 exhibited two cathodic reduction peaks. The voltammetric peaks were obtained at −0.144 V (reversible) and −1.250 V (irreversible) at pH 3.2, and correspond to the reduction of resorufin to dihydroresorufin and to the catalytic hydrogen wave, respectively. At pH > 4.0, a new irreversible cathodic reduction peak, assigned to the protonation of N-oxide on the phenoxazin ring, was observed. Electrochemical parameters (I p/E p, I p/v, I p/pH, I p/t acc) of the compound were determined. From the voltammetric data, electrochemical reduction mechanisms for all peaks have been suggested. Maximum peak current for the reversible peak was obtained at pH 4.1. A linear relationship between the current and concentration was determined, and also the lowest detection limit was found as 3.25 × 10−8 mol L−1 and 1.98 × 10−10 mol L−1 for SWV and SWAdSV, respectively.
Similar content being viewed by others
References
Baizer, M. M., & Lund, H. (1983). Organic electrochemistry: An introduction and a guide (2nd ed., pp. 26–29). New York, NY, USA: Marcel Dekker.
Baker, C. N., Banerjee, S. N., & Tenover, F. C. (1994). Evaluation of Alamar colorimetric MIC method for antimicrobial susceptibility testing of gram-negative bacteria. Journal of Clinical Microbiology, 32, 1261–1267.
Bard, A. J., & Faulkner, L. R. (1986). Electrochemical methods: Fundamentals and applications (pp. 522–538). Beijing, China: Wiley, Chemical Industry Press.
Bueno, C., Willegas, M. L., Bertolotti, S. G., Previtali, C. M., Neummann, M. G., & Encinas, M. V. (2002). The excitedstate interaction of resazurin and resorufin with amines in aqueous solutions. Photophysics and photochemical reaction. Photochemistry and Photobiology, 76, 385–390. DOI: 10.1562/0031-8655(2002)0760385TESIOR2.0.CO2.
Crawford, P. W., Scamehorn, R. G., Hollestein, U., Ryan, M. D., & Kovacic, P. (1986). Cyclic voltammetry of phenazines and quinoxalines including mono- and di-N-oxides. Relation to structure and antimicrobial activity. Chemico-Biological Interactions, 60, 67–84. DOI: 10.1016/0009-2797(86)90018-9.
Emerson, T. R., & Rees, C. W. (1962). The deoxygenation of heterocyclic N-oxides. Part II. Polarographic reduction. Journal of the Chemical Society, 1962, 1923–1927. DOI: 10.1039/JR9620001923.
Gorton, L., Tortensson, A., Jaegfeldt, H., & Johansson, G. (1984). Electrocatalytic oxidation of reduced nicotinamide coenzymes by graphite electrodes modified with an adsorbed phenoxazinium salt, meldola blue. Journal of Electroanalytical Chemistry, 161, 103–120. DOI: 10.1016/S0022-0728(84)80253-3.
Hu, X.-Y., Xiao, Y., & Chen, H.-Y. (1999). Adsorption characteristics of Fe(CN) 3−6 /4− 6 on Au colloids as monolayer film on cysteamine-modified gold electrode. Journal of Electroanalytical Chemistry, 466, 26–30. DOI: 10.1016/S0022-0728(99)00113-8.
Jenkins, A. T. A., Dash, H.-A., Boundy, S., Halliwell, C. M., & ffrench-Constant, R. H. (2006). Methoxy-resorufin ether as an electrochemically active biological probe for cytochrome P450 o-demethylation. Bioelectrochemistry, 68, 67–71. DOI: 10.1016/j.bioelechem.2005.04.002.
Kubota, T., & Miyazaki, H. (1962). Polarography of pyridine N-oxide and its alkyl derivatives. Bulletin of the Chemical Society of Japan, 35, 1549–1551. DOI: 10.1246/bcsj.35.1549.
Laviron, E., Roullier, L., & Dergrand, C. (1980). A multiplayer model for the study of space distributed redox modified electrodes: Part II. Theory and application of linear potential sweep voltammetry for a simple reaction. Journal of Electroanalytical Chemistry, 112, 11–23. DOI: 10.1016/S0022-0728(80)80003-9.
Leach, S. C., Weaver, R. D., Kinoshita, K., & Lee, W.W. (1981). Polarographic analysis of heterocyclic nitrogen compounds. Journal of Electroanalytical Chemistry, 129, 213–227. DOI: 10.1016/S0022-0728(81)80015-0.
Maeda, H., Matsu-ura, S., Yamauchi, Y., & Ohmori, H. (2001). Resazurin as an electron acceptor in glucose oxidasecatalyzed oxidation of glucose. Chemical & Pharmaceutical Bulletin, 49, 622–625. DOI: 10.1248/cpb.49.622.
Montejano, H. A., Gervaldo, M., & Bertolotti, S. G. (2005). The excited-states quenching of resazurin and resorufin by p-benzoquinones in polar solvents. Dyes and Pigments, 64, 117–124. DOI: 10.1016/j.dyepig.2004.05.002.
Moyer, R. H., & Campbell, J. J. R. (1963). Mechanism of resazurin reduction in milk of low bacterial content. Journal of Dairy Science, 46, 897–906.
Nicholson, R. S., & Shain, I. (1964). Theory of stationary electrode polarography. Single scan and cyclic methods applied to reversible, irreversible, and kinetic systems. Analytical Chemistry, 36, 706–723. DOI: 10.1021/ac60210a007.
Nigović, B. (2006). Electrochemical properties and square-wave voltammetric determination of pravastatin. Analytical and Bioanalytical Chemistry, 384, 431–437. DOI: 10.1007/s00216-005-0188-1.
Olea-Azar, C., Rigol, C., Mendizábal, F., Briones, R., Cerecetto, H., Di Maio, R., Risso, M., González, M., & Porcal, W. (2003). Electrochemical and microsomal production of free radicals from 1,2,5-oxadiazole N-oxide as potential antiprotozoal drugs. Spectrochimica Acta Part A: Molecular and Biomolecular Spectroscopy, 59, 69–74. DOI: 10.1016/S1386-1425(02)00141-5.
Ottaway, J. M. (1972). In E. Bishop (Ed.), Indicators (pp. 469–529), Oxford, UK: Pergamon Press.
Pospíšil, L., Trsková, R., Fuoco, R., & Colombini, M. P. (1995). Electrochemistry of s-triazine herbicides: Reduction of atrazine and terbutylazine in aqueous solutions. Journal of Electroanalytical Chemistry, 395, 189–193. DOI: 10.1016/0022-0728(95)04096-7.
Prütz, W. A. (1994). Reduction of resazurin by glutathione activated by sulfanes and selenite. Journal of the Chemical Society, Chemical Communications, 1994, 1639–1640. DOI: 10.1039/C39940001639.
Prütz, W. A., Butler J., & Land, J. E. (1996). Photocatalytic and free radical interactions of the heterocyclic N-oxide resazurin with NADH, GSH, and DOPA. Archives of Biochemistry and Biophysics, 327, 239–248. DOI: 10.1006/abbi.1996.0116.
Ruddock, G. W., & Greenstock, C. L. (1977). Application of polarography to the study of chemical radiosensitization of DNA components. Biochimica et Biophysica Acta, 496, 197–202. DOI: 10.1016/0304-4165(77)90128-3.
Tiballi, R. N., He, X., Zarins, L. T., Revankar, S. G., & Kauffman, C. A. (1995). Use of a colorimetric system for yeast susceptibility testing. Journal of Clinical Microbiology, 33, 915–917.
Zabransky, R. J., Dinuzzo, A. R., & Woods, G. L. (1995). Detection of vancomycin resistance in enterococci by the Alamar MIC system. Journal of Clinical Microbiology, 33, 791–793.
Zón, M. A., Angulo, M., & Rodríguez Melladore, J. M. (1994). Study of the reduction mechanism of the N-oxides of pyridine monocarboxylic acids at mercury electrodes. Journal of Electroanalytical Chemistry, 365, 213–220. DOI: 10.1016/0022-0728(93)02990-Y.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Çakir, S., Arslan, E.Y. Voltammetry of resazurin at a mercury electrode. Chem. Pap. 64, 386–394 (2010). https://doi.org/10.2478/s11696-010-0007-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.2478/s11696-010-0007-9