Abstract
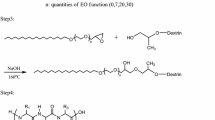
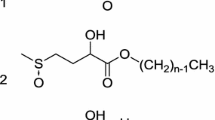
Water-soluble, partially hydrophobized polysaccharides have become attractive due to their potential to act as polymeric surfactants. From carboxymethyl starch (CMS), water-soluble polymeric surfactants were prepared by esterification using two unconvential methods based on (A) a reaction with mixed anhydrides and (B) with methyl laureate (MELA) and methyl esters of the fatty acid complex of rape seed oil (MERO) under ‘solvent-free’ conditions. The obtained CMS derivatives were characterized by structural, surface-active and surfactant performance properties. Esterification using method B was successful in contrast to method A which yielded derivatives with a very low extent of esterification. Although the derivatives reduced the surface tension moderately, they exhibited remarkable emulsifying properties. The performance tests indicated good washing power and antiredeposition efficiency. The results suggest that suitable polymeric surfactants can be prepared by the unconventional esterification method B using both acylation agents. The preparations might substitute the expensive and invasive industrially applied conventional methods.
Similar content being viewed by others
References
Aburto, J., Alric, I., & Borredon, E. (2005). Organic solvent-free transesterification of various starches with lauric acid methyl ester and triacyl glycerides. Starch/Stärke, 57, 145–152. DOI: 10.1002/star.200400380.
Akiyama, E., Kashimoto, A., Fukuda, K., Hotta, H., Suzuki, T., & Kitsuki, T. J. (2005). Thickening properties and emulsification mechanisms of new derivatives of polysaccharides in aqueous solution. Journal of Colloid and Interface Science, 282, 448–457. DOI: 10.1016/j.jcis.2004.08.178.
Alpert, N. L., Keiser, W. E., & Szymanski, H. A. (1997). IR, theory and practices of infrared spectroscopy. New York: Plenum Press.
Anastas, P. T., & Williams, T. C. (1998). Green chemistry. London: Oxford University Press.
Burczyk, B. (2002). Biodegradable and chemodegradable nonionic surfactants. In Encyclopedia of surface and colloid science (Vol. 1, pp. 724–752). New York: Marcel Dekker.
Fang, J. M., Fowler, P. A., Tomkinson, J., & Hill, C. A. S. (2002). The preparation and characterisation of a series of chemically modified potato starches. Carbohydrate Polymers, 47, 245–252. DOI: 10.1016/S0144-8617(01)00187-4.
Girardeau, S., Aburto, J., Vaca-Garcia, C., Alric, I., & Borredon, E. (1999). A performing method of transesterification of cellulose and amylose. In Proceedings of the IV. European Symposium on Industrial Crops and Products, 23–25 March 1999 (pp. 229). Münster, Germany: Landwirtschaftsverlag GmbH.
Heinze, T., Rensing, S., & Koschella, A. (2007). Starch derivatives of high degree of functionalization. 13. Novel amphiphilic starch products. Starch/Stärke, 59, 199–207. DOI: 10.1002/star.200600561.
Ihara, T., Nishioka, T., Kiyosaki, T., & Kamitani, H. (2004). Solution properties of a novel polysaccharide derivative. Chemistry Letters, 33, 1094–1095. DOI: 10.1246/cl.2004.1094.
Latetin, A. I., Gal’braikh, L. S., & Rogovin, Z. A. (1968). Use of the ester exchange reaction for the synthesis of cellulose stearates. Polymer Science USSR, 10, 761–766.
Lunkenheimer, K., & Miller, R. (1979). On the purity of aqueous surfactant solutions and the dynamic surface tension behaviour. Tenside, 16, 312–316.
Miníková, S. (2008). The synthesis and study of the structure and properties of novel hydroxyethylcellulose derivatives. Ph.D. thesis, Trenčín University of Alexander Dubček, Púchov.
Mocanu, G., Mihai, D., LeCerf, D., Picton, L., & Muller, G. (2004). Synthesis of new associative gel microspheres from carboxymethyl pullulan and their interactions with lysozyme. European Polymer Journal, 40, 283–289. DOI: 10.1016/j.eurpolymj.2003.09.019.
Paik, Y. H., & Swift, G. (1995). Polysaccharides as raw materials for the detergent industry. Chemistry & Industry, 2, 55–59.
Piasecki, A. (2002). Biodegradable and chemically degradable anionic surfactants. In Encyclopedia of surface and colloid science (Vol. 1, pp. 894–917). New York: Marcel Dekker.
Rooney, M. L. (1976). Interesterification of starch with methyl palmitate. Polymer, 17, 555–558. DOI: 10.1016/0032-3861(76)90267-6.
Rosilio, V., Albrecht, G., Baszkin, A., & Merle, L. (2000). Surface properties of hydrophobically modified carboxymethylcellulose derivatives. Effect of salt and proteins. Colloids and Surfaces B: Biointerfaces, 19, 163–172. DOI: 10.1016/SO927 7765(00)00151 X.
Rouzes, C., Durand, A., Leonard, M., & Dellacherie, E. (2002). Surface activity and emulsification properties of hydrophobically modified dextrans. Journal of Colloid and Interface Science, 253, 217–223. DOI: 10.1006/jcis.2002.8357.
Sarkar, N. (1984). Structural interpretation of the interfacial properties of aqueous solutions of methylcellulose and hydroxypropyl methylcellulose. Polymer, 25, 481–486. DOI: 10.1016/0032-3861(84)90206-4.
Shogren, R. L. (1996). Preparation, thermal properties, and extrusion of high-amylose starch acetates. Carbohydrate Polymers, 29, 57–62. DOI: 10.1016/0144-8617(95)00143-3.
Skalková, P., Sroková, I., Sasinková, V., & Ebringerová, A. (2006). Polymeric surfactants from beechwood glucuronoxylan. Tenside Surfactants Detergents, 43, 137–141.
Sroková, I., Ebringerová, A., & Heinze, T. (2001). Emulsifying agents based on O-(carboxymethyl)starch. Tenside Surfactants Detergents, 38, 277–279.
Sroková, I., Miníková, S., Ebringerová, A., Sasinková, V., & Heinze, T. (2003). Novel O-(2-hydroxyethyl) cellulose-based nonionic biosurfactants. Tenside Surfactants Detergents, 40, 73–76.
Sroková, I., Talába, P., Hodul, P., & Balážová, A. (1998). Emulsifying agents based on O-(carboxymethyl)celulose. Tenside Surfactants Detergents, 35, 342–344.
Sroková, I., Tomanová, V., Ebringerová, A., & Heinze, T. (2004). Water-soluble esterified O-(carboxymethyl)cellulose) derivatives-synthesis and properties. Macromolecular Materials and Engineering, 289, 63–69. DOI: 10.1002/mame. 200300124.
Stüpel, H. (1957). Synthetische Wasch und Reinigungsmittel (1st ed.). Stuttgart: Konradin Verlag.
Vaca-Garcia, C., & Borredon, M. E. (1999). Solvent-free fatty acylation of cellulose and lignocellulosic wastes. Part 2: Reactions with fatty acid. Bioresource Technology, 70, 135–142. DOI: 10.1016/S0960-8524(99)00034-6.
Vaca-Garcia, C., Thiebaud, S., Borredon, M. E., & Gozzelino, G. (1998). Cellulose esterification with fatty acids and acetic anhydride in lithium chloride/N,N-dimethylacetamide medium. Journal of the American Oil Chemists’ Society, 75, 315–319. DOI: 10.1007/s11746-998-0047-2.
Xing, G. X., Zhang, S. F., Ju, B. Z., & Yang, J. Z. (2006). Microwave-assisted synthesis of starch maleate by dry method. Starch/Stärke, 58, 464–467. DOI: 10.1002/star. 200600507.
Zhang, L. M. (2001). Cellulosic associative thickeners. Carbohydrate Polymers, 45, 1–10. DOI: 10.1016/SO144.8617(00) 00276-9.
Žoldáková, A., Sroková, I., Sasinková, V., Hirsch, J., & Ebringerová, A. (2005). Biosurfactants based on partially esterified O-(carboxymethyl)starch. Chemical Papers, 59, 362–367.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Mazíková, V., Sroková, I. & Ebringerová, A. Solvent-free synthesis and properties of carboxymethyl starch fatty acid ester derivatives. Chem. Pap. 63, 71–76 (2009). https://doi.org/10.2478/s11696-008-0088-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.2478/s11696-008-0088-x