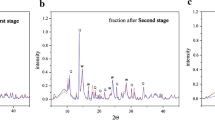
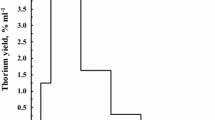
Abstract
Determination of thallium in polluted environmental samples and their extracts obtained by a modified BCR three-step sequential extraction procedure was used to study thallium distribution and mobility in the monitored polluted area affected by acidification (Šobov, Central Slovakia). The results of fractionation applied to 5 soil certified reference materials and 14 environmental samples show that the vast majority of thallium occurred in the residual fraction. This means that highly toxic thallium is strongly entrapped in the parent rock materials remains immobile and its environmental toxicity is therefore reduced. The limit of detection for thallium in the studied fractions was lower than 0.050 mg kg−1, the precision (RSD) of the ultratrace determination of thallium in the studied fractions was better than 17 % and the accuracy of the used method was verified by analyzing certified reference materials.
Similar content being viewed by others
References
Adriano, D. C. (2001). Trace elements in terrestrial environments — Biochemistry, bioavailability and risk of metals (2nd ed.), New York: Springer.
Agnihotri, P. K., Deb, M. K., Mishra, R. K., & Thakur, M. (1998). Fractionation and spectrophotometric determination of thallium with N,N′-diphenylbenzamidine, brilliant green and cetylpyridinium chloride. Chemical Speciation and Bioavailability, 10, 53–60.
Barrows, M. E., Petrocelli, S. R., & Macek, K. J. (1978). Bio-concentration and elimination of selected water pollutants by bluegill sunfish. In: R. Hague (Ed.), Dynamics, exposure and hazard assessment of toxic chemicals (pp. 379–392), Ann Arbor, MI: Ann Arbor Publishers, Inc.
Bowen, H. J. M. (1979). Environmental chemistry of the elements. London: Academic Press.
Boyarsky, I., & Crisan, A. D. (2006). Toxicity, Thallium. In: eMedecine. Retrieved May, 2006, from http://www.emedecine.com/EMERG/topic926.htm.
Cvetkovic, J., Arpadjan, S., Karadjova, I., & Stafilov, T. (2002). Determination of thallium in wine by electrothermal atomic absorption spectrometry after extraction preconcentration. Spectrochimica Acta Part B: Atomic Spectrometry 57, 1101–1106. DOI: 10.1016/S0584-8547(02)00032-0.
Ensafi, A. A., & Rezaei, B. (1998). Speciation of thallium by flow injection analysis with spectrofluorimetric detection. Mi-crochemical Journal, 60, 75–83. DOI: 10.1006/mchj.1998. 1625.
Emers, U. (1988). Environmental exposure to thallium. The Science of the Total Environment, 71, 285–292. DOI: 10.1016/0048-9697(88)90199-4.
Frantz, G. & Carlson, R. M. (1987). Effects of rubidium, cesium, and thallium on interlayer potassium release from transvaal vermiculite. Soil Science Society of America Journal, 51, 305–308.
Jakubowska, M., Pasieczna, A., Zembrzuski, W., Swit, Z., & Lukaszewski, Z. (2007). Thallium in fractions of soil formed on floodplain terraces. Chemosphere, 66, 611–618. DOI: 10.1016/j.chemosphere.2006.07.098.
Jakubowska, M., Zembrzuski, W., & Lukaszewski, Z. (2006). Oxidative extraction versus total decomposition of soil in the determination of thallium. Talanta, 68, 1736–1739. DOI: 10.1016/j.talanta.2005.08.035.
Karlsson, S., Bäckström, M., & Lifvergren, T. (2003). Distribution of thallium in a contaminated lake. ICP Information Newsletter, 28, 556–557.
Kemper, F. H., & Bertram, H. P. (1991). Thallium. In E. Merian (Ed.) Metals and their compounds in the environment: Occurrence, analysis and biological relevance, Chapter II. 29, (pp. 1227–1241). Weinheim: Wiley-VCH.
Lin, T. S., & Nriagu, J. O., (1999). Thallium speciation in river waters with Chelex-100 resin. Analytica Chimica Acta, 395, 301–307. DOI: 10.1016/S0003-2670(99)00358-X.
Lukaszewski, Z., Karbowska, B., & Zembrzuski, W. (2003). Determination of mobile thallium in soil by flow injection differential pulse anodic stripping voltammetry. Electroanalysis, 15, 480–483. DOI: 10.1002/elan.200390057.
Medveï, J., Streško. V., Kubová J., & Polakovičová J., (1998). Efficiency of decomposition procedures for the determination of some elements in soils by atomic spectroscopic methods. Fresenius Journal of Analytical Chemistry, 360, 219–224. DOI: 10.1007/s002160050678.
Mossop, K. F., & Davidson, C. M. (2003). Comparison of original and modified BCR sequential extraction procedures for the fractionation of copper, iron, lead, manganese and zinc in soils and sediments. Analytica Chimica Acta, 478, 111–118. DOI: 10.1016/S0003-2670(02)01485-X.
Rauret, G., López-Sánchez, J. F., Sahuquillo, A., Barahona, E., Lachica, M., Ure, A. M., Davidson, C. M., Gomez, A., Lück, D., Bacon, J., Yli-Halla, M., Muntau, B. H., & Quevauviller, P. (2000). Application of a modified BCR sequential extraction (three-step) procedure for the determination of ex-tractable trace metal contents in a sewage sludge amended soil reference material (CRM 483), complemented by a three-year stability study of acetic acid and EDTA extractable metal content. Journal of Environmental Monitoring, 2, 228–233. DOI: 10.1039/b001496f.
Sager, M. (1992). Speciation of thallium in river sediments by consecutive leaching techniques. Microchimica Acta, 106, 241–251. DOI: 10.1007/BF01242096.
Tsalev, D. L., & Slaveykova, V. I. (1992). Chemical modification in electrothermal atomic absorption spectrometry. Organization and classification of data by multivariate methods. Journal of Analytical Atomic Spectrometry, 7, 147–153. DOI: 10.1039/JA9920700147.
Van Reeuwijk, L. P. (Ed.) (1995). Procedures for soil analysis, 5th ed. Wageningen: International Soil Reference and Information Centre.
Vidal, M., & Rauret, M., (1993). Two approaches for sequential extraction of radionuclides in soils: Batch and column methods. International Journal of Environmental Analytical Chemistry, 51, 85–95. DOI: 10.1080/03067319308027613.
Villar, M., Alava, F., Lavilla, I., & Bendicho, C. (2001). Operational speciation of thallium in environmental solid samples by electrothermal atomic absorption spectrometry according to the BCR sequential extraction scheme. Journal of Analytical Atomic Spectrometry, 16, 1424–1428. DOI: 10.1039/b106546g.
Volynsky, A. B. (2000). Mechanisms of action of platinum group modifiers in electrothermal atomic absorption spectrometry. Spectrochimica Acta Part B: Atomic Spectrocopy, 55, 103–150. DOI: 10.1016/S0584-8547(99)00175-5.
Welz, B., Schlemmer, G., & Mudakavi, J. R. (1988). Palladium nitrate-magnesium nitrate modifier for graphite furnace atomic absorption spectrometry Part 2: Determination of arsenic, cadmium, copper, manganese, lead, antimony selenium and thallium in water. Journal of Analytical Atomic Spectrometry, 3, 695–701. DOI: 10.1039/JA9880300695.
Yang, C., Chen, Y., Peng, P., Li, C., Chang, X., & Xie, C. (2005). Distribution of natural and anthropogenic thallium in the soils in an industrial pyrite slag disposing area. Science of the Total Environment, 341, 159–172. DOI: 10.1016/j.scitotenv.2004.09.024.
Ni, Z. M., & Shan, X. Q. (1987). The reduction and elimination of matrix interferences in graphite furnace atomic absorption spectrometry. Spectrochimica Acta Part B: Atomic Spectrocopy,, 42, 937–949. DOI: 10.1016/0584-8547(87)80106-4.
Zhu, D., & Xu, S. K. (2000). Enhancement of thallium response by flow injection hydride generation AAS using palladium and rhodamine B. Atomic Spectroscopy, 21, 136–142.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Medved’, J., Kališ, M., Hagarová, I. et al. Thallium fractionation in polluted environmental samples using a modified BCR three-step sequential extraction procedure and its determination by electrothermal atomic absorption spectrometry. Chem. Pap. 62, 168–175 (2008). https://doi.org/10.2478/s11696-008-0007-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.2478/s11696-008-0007-1