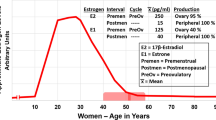
Abstract
The skin expresses estrogen, progesterone, and androgen receptors. In the presence of steroid hormones, such as those contained in oral contraceptives, the skin likely responds to hormonal signals that control the cell cycle, apoptosis, DNA replication, and other cellular functions. Some estrogen-responsive pathways have the potential to promote tumor development, including the augmentation of epidermal growth factor signaling, the expression of proto-oncogenes, and inhibition of apoptosis. The question of whether oral contraceptives increase the risk for the development of skin cancer, particularly melanoma, is still an area of concern. This paper reviews the available evidence, the bulk of which suggests that while the skin responds to estrogens, progestins, and androgens, these responses do not significantly increase the risk of developing skin cancer when estrogen exposure is not excessive.
Similar content being viewed by others
References
Jensen EV, DeSombre ER. Estrogen-receptor interaction. Science. 1973; 182 (108): 126–34
Evans RM. The steroid and thyroid hormone receptor superfamily. Science. 1988; 240 (4854): 889–95
Onate SA, Tsai SY, Tsai MJ, et al. Sequence and characterization of a coactivator for the steroid hormone receptor superfamily. Science. 1995; 270 (5240): 1354–7
Carroll JS, Swarbrick A, Musgrove EA, et al. Mechanisms of growth arrest by c-myc antisense oligonucleotides in MCF-7 breast cancer cells: implications for the antiproliferative effects of antiestrogens. Cancer Res 2002; 62 (11): 3126–31
Webb P, Nguyen P, Valentine C, et al. The estrogen receptor enhances AP-1 activity by two distinct mechanisms with different requirements for receptor transactivation functions. Mol Endocrinol. 1999; 13 (10): 1672–85
Kushner PJ, Agard DA, Greene GL, et al. Estrogen receptor pathways to AP-1. J Steroid Biochem Mot Biol. 2000; 74 (5): 311–7
Kushner PJ, Agard D, Feng WJ, et al. Oestrogen receptor function at classical and alternative response elements. Novartis Found Symp. 2000; 230: 20–6
Horwitz KB, Alexander PS. In situ photolinked nuclear progesterone receptors of human breast cancer cells: subunit molecular weights after transformation and translocation. Endocrinology. 1983; 113 (6: 2195–201
Lessey BA, Alexander PS, Horwitz KB. The subunit structure of human breast cancer progesterone receptors: characterization by chromatography and photoaffinity labeling. Endocrinology. 1983; 112 (4): 1267–74
Arnett-Mansfield RL, deFazio A, Wain GV, et al. Relative expression of progesterone receptors A and B in endometrioid cancers of the endometrium. Cancer Res. 2001; 61 (11): 4576–82
Hanekamp EE, Kuhne EC, Smid-Koopman E, et al. Loss of progesterone receptor may lead to an invasive phenotype in human endometrial cancer. Fur J Cancer. 2002; 38 Suppl. 6: S71–2
Gregory CW, He B, Wilson EM. The putative androgen receptor-A form results from in vitro proteolysis. J Mol Endocrinol. 2001; 27 (3): 309–19
Tilley WD, Marcelli M, Wilson JD, et al. Characterization and expression of a cDNA encoding the human androgen receptor. Proc Natl Acad Sci U S A. 1989; 86 (1): 327–31
Wilson CM, McPhaul MI. A and B forms of the androgen receptor are present in human genital skin fibroblasts. Proc Natl Acad Sci U S A. 1994; 91 (4): 1234–8
Catalano MG, Pfeffer U, Raineri M, et al. Altered expression of androgen-receptor isoforms in human colon-cancer tissues. Int J Cancer. 2000; 86 (3): 325–30
Ho KJ, Liao JK. Nonnuclear actions of estrogen. Arterioscler Thromb Vase Biol. 2002; 22 (12): 1952–61
McCann JP, Mayes JS, Hendricks GR, et al. Subcellular distribution and glycosylation pattern of androgen receptor from sheep omental adipose tissue. J Endocrinol; 2001; 169 (3): 587–93
Harvey BJ, Doolan CM, Condliffe SB, et al. Non-genomic convergent and divergent signaling of rapid responses to aldosterone and estradiol in mammalian colon. Steroids. 2002; 67 (6): 483–91
Singleton DW, Feng Y, Burd CJ, et al. Nongenomic activity and subsequent c-fos induction by estrogen receptor ligands are not sufficient to promote deoxyribonucleic acid synthesis in human endometrial adenocarcinoma cells. Endocrinology. 2003; 144 (1): 121–8
Filardo EJ. Epidermal growth factor receptor (EGFR) transactivation by estrogen via the G-protein-coupled receptor, GPR30: a novel signaling pathway with potential significance for breast cancer. J Steroid Biochem Mol Biol. 2002; 80 (2): 231–8
Filardo EJ, Quinn JA, Frackelton AR, et al. Estrogen action via the G protein-coupled receptor, GPR30: stimulation of adenylyl cyclase and CAMP-mediated attenuation of the epidermal growth factor receptor-to-MAPK signaling axis. Mol Endocrinol. 2002; 16 (1): 70–84
Ahola TM, Alkio N, Manninen T, et al. Progestin and G protein-coupled receptor 30 inhibit mitogen-activated protein kinase activity in MCF-7 breast cancer cells. Endocrinology. 2002; 143 (12): 4620–6
Ahola TM, Manninen T, Alkio N, et al. G protein-coupled receptor 30 is critical for a progestin-induced growth inhibition in MCF-7 breast cancer cells. Endocrinology. 2002; 143 (9): 3376–84
Ahola TM, Purmonen S, Pennanen P, et al. Progestin upregulates G-protein-coupled receptor 30 in breast cancer cells. Eur J Biochem. 2002; 269 (10): 2485–90
Fraser D, Padwick ML, Whitehead M, et al. Presence of an oestradiol receptor-related protein in the skin: changes during the normal menstrual cycle. Br J Obstet Gynaecol. 1991; 98 (12): 1277–82
Pelletier G, El-Alfy M. Immunocytochemical localization of estrogen receptors alpha and beta in the human reproductive organs. J Clin Endocrinol Metab. 2000; 85 (12): 4835–40
Sun J, Baudry J, Katzenellenbogen JA, et al. Molecular basis for the subtype discrimination of the estrogen receptor-beta-selective ligand, diarylpropionitrile. Mol Endocrinol. 2003; 17 (2): 247–58
Sun J, Huang YR, Harrington WR, et al. Antagonists selective for estrogen receptor alpha. Endocrinology. 2002; 143 (3): 941–7
Shiau AK, Barstad D, Radek IT, et al. Structural characterization of a subtype-selective ligand reveals a novel mode of estrogen receptor antagonism. Nat Struct Biol. 2002; 9 (5): 359–64
Harris HA, Katzenellenbogen JA, Katzenellenbogen BS. Characterization of the biological roles of the estrogen receptors, ERα and ERβ, in estrogen target tissues in vivo through the use of an ERa-selective ligand. Endocrinology. 2002; 143 (11): 4172–7
Rickard DJ, Waters KM, Ruesink TJ, et al. Estrogen receptor isoform-specific induction of progesterone receptors in human osteoblasts. J Bone Miner Res. 2002; 17 (4): 580–92
Lindberg MK, Moverare S, Skrtic S, et al. Estrogen receptor (ER)-β reduces ERα-regulated gene transcription, supporting a ‘ying yang’ relationship between ERα and ERβ in mice. Mol Endocrinol. 2003; 17 (2): 203–8
Moverare S, Lindberg MK, Faergemann J, et al. Estrogen receptor-α, but not estrogen receptor-β, is involved in the regulation of the hair follicle cycling as well as the thickness of epidermis in male mice. J Invest Dermatol. 2002; 119 (5): 1053–8
Avila DM, Wilson CM, Nandi N, et al. Immunoreactive AR and genetic alterations in subjects with androgen resistance and undetectable AR levels in genital skin fibroblast ligand-binding assays. J Clin Endocrinol Metab. 2002; 87 (1): 182–8
Alesci S, Bornstein SR. Neuroimmunoregulation of androgens in the adrenal gland and the skin. Horm Res. 2000; 54 (5-6): 281–6
Labrie F, Luu-The V, Labrie C, et al. Intracrinology and the skin. Horm Res. 2000; 54 (5-6): 218–29
Nelson LR, Bulun SE. Estrogen production and action. J Am Acad Dermatol. 2001; 45 (3 Suppl.): S116–24
Izu K, Yamamoto O, Yamaguchi J, et al. A case of autoimmune progesterone dermatitis [in German]. J Uoeh. 2001; 23 (4): 431–6
Uotinen N, Puustinen R, Pasanen S, et al. Distribution of progesterone receptor in female mouse tissues. Gen Comp Endocrinol. 1999; 115 (3): 429–41
Kommoss F, Kiechle-Schwarz M, Dubois A, et al. Co-cultivation of ovarian carcinoma cells with dermal fibroblasts induces fibroblast expression of sex steroid receptor transcripts and protein. Int J Gynecol Cancer. 1995; 5 (2): 101–6
Im S, Lee ES, Kim W, et al. Expression of progesterone receptor in human keratinocytes. J Korean Med Sci. 2000; 15 (6: 647–54
Rossouw JE, Anderson GL, Prentice RL, et al. Risks and benefits of estrogen plus progestin in healthy postmenopausal women: principal results from the Women’s Health Initiative randomized controlled trial. JAMA. 2002; 288 (3): 321–33
Brekelmans CT. Risk factors and risk reduction of breast and ovarian cancer. Curr Opin Obstet Gynecol. 2003; 15 (1): 63–8
Matsumum Y, Ananthaswamy HN. Molecular mechanisms of photocarcinogenesis. Front Biosci. 2002; 7: d765–83
Nataraj AJ, Trent JC, Ananthaswamy HN. p53 gene mutations and photocarcinogenesis. Photochem Photobiol. 1995; 62 (2): 218–30
Nishigori C, Yarosh DB, Donawho C, et al. The immune system in ultraviolet carcinogenesis. J Investig Dermatol Symp Proc. 1996; 1 (2): 143–6
van Steeg H, Kraemer KH. Xeroderma pigmentosum and the role of UV-induced DNA damage in skin cancer. Mol Med Today. 1999; 5 (2): 86–94
McGregor JM, Yu CC, Dublin EA, et al. Aberrant expression of p53 tumour-suppressor protein in non-melanoma skin cancer. Br J Dermatol. 1992; 127 (5): 463–9
Shea CR, McNutt NS, Volkenandt M, et al. Overexpression of p53 protein in basal cell carcinomas of human skin. Am J Pathol. 1992; 141 (1): 25–9
Stephenson TJ, Royds J, Silcocks PB, et al. Mutant p53 oncogene expression in keratoacanthoma and squamous cell carcinoma. Br J Dermatol. 1992; 127 (6): 566–70
Kimonis VE, Goldstein AM, Pastakia B, et al. Clinical manifestations in 105 persons with nevoid basal cell carcinoma syndrome. Am J Med Genet. 1997; 69 (3): 299–308
Pierceall WE, Goldberg LH, Tainsky MA, et al. Ras gene mutation and amplification in human nonmelanoma skin cancers. Mol Carcinog. 1991; 4 (3): 196–202
Pierceall WE, Mukhopadhyay T, Goldberg LH, et al. Mutations in the p53 tumor suppressor gene in human cutaneous squamous cell carcinomas. Mol Carcinog. 1991; 4 (6): 445–9
Dai D, Litman ES, Schonteich E, et al. Progesterone regulation of activating protein-1 transcriptional activity: a possible mechanism of progesterone inhibition of endometrial cancer cell growth. J Steroid Biochem Mol Biol. 2003; 87 (2-3): 123–31
Dai D, Wolf DM, Litman ES, et al. Progesterone inhibits human endometrial cancer cell growth and invasiveness: down-regulation of cellular adhesion molecules through progesterone B receptors. Cancer Res. 2002; 62 (3): 881–6
Rogers GS, Flowers JL, Pollack SV, et al. Determination of sex steroid receptor in human basal cell carcinoma. J Am Acad Dermatol. 1988; 18 (5 Pt 1): 1039–43
Gefeller O, Hassan K, Wille L. Cutaneous malignant melanoma in women and the role of oral contraceptives. Br J Dermatol. 1998; 138 (1): 122–4
Miller JG, Gee J, Price A, et al. Investigation of oestrogen receptors, sex steroids and soluble adhesion molecules in the progression of malignant melanoma. Melanoma Res. 1997; 7 (3): 197–208
Ladanyi A, Timar J, Bocsi J, et al. Sex-dependent liver metastasis of human melanoma lines in SCID mice. Melanoma Res. 1995; 5 (2): 83–6
Kanda N, Watanabe S. 17beta-estradiol, progesterone, and dihydrotestosterone suppress the growth of human melanoma by inhibiting interleukin-8 production. J Invest Dermatol. 2001; 117 (2): 274–83
Richardson B, Price A, Wagner M, et al. Investigation of female survival benefit in metastatic melanoma. Br J Cancer. 1999; 80 (12): 2025–33
Beral V, Ramcharan S, Faris R. Malignant melanoma and oral contraceptive use among women in California. Br J Cancer. 1977; 36 (6): 804–9
Pfahlberg A, Hassan K, Wille L, et al. Systematic review of case-control studies: oral contraceptives show no effect on melanoma risk. Public Health Rev. 1997; 25 (3-4): 309–15
Karagas MR, Stukel TA, Dykes J, et al. A pooled analysis of 10 case-control studies of melanoma and oral contraceptive use. Br J Cancer. 2002; 86 (7): 1085–92
Feskanich D, Hunter DJ, Willett WC, et al. Oral contraceptive use and risk of melanoma in premenopausal women. Br J Cancer. 1999; 81 (5): 918–23
Acknowledgment
Preparation of this article was funded by the Cory Beach Family Fund (KL) and the US National Institutes of Health 1R01CA99908-01 (KL). The authors have no conflicts of interest relevant to this article.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Leslie, K.K., Espey, E. Oral Contraceptives and Skin Cancer. Am J Clin Dermatol 6, 349–355 (2005). https://doi.org/10.2165/00128071-200506060-00002
Published:
Issue Date:
DOI: https://doi.org/10.2165/00128071-200506060-00002