Abstract
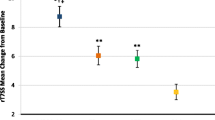
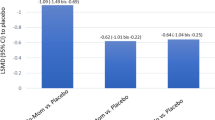
Nasal obstruction, which many patients consider to be the most bothersome symptom of seasonal allergic rhinitis (SAR), is generally refractory to oral anti-histamine therapy. Effective resolution of nasal obstruction associated with SAR may help to prevent lower-airway disorders and other adverse sequelae (e.g. otitis media with effusion). Desloratadine, a nonsedating antihistamine with marked inhibitory effects on the early- and late-phase allergic responses, affords significant relief of sneezing, pruritus and rhinorrhoea, as well as nasal congestion. Using the Vienna Challenge Chamber, a closed system that enables rigorously controlled allergen exposure, we observed that a single 5mg dose of desloratadine rapidly and markedly reduced postexposure nasal obstruction in a pilot study. Separately, three randomised, double-blind, placebo-controlled trials demonstrated that desloratadine significantly reduced nasal blockage, as well as acute SAR symptoms, from baseline as compared with placebo over a 5-hour interval in the pollen chamber. The favourable effects of desloratadine on early-phase symptoms were consistent with evidence from controlled-allergen-exposure trials involving other antihistamines (cetirizine, fexofenadine). However, desloratadine also significantly protected against allergen-induced declines in nasal airflow (as assessed by active anterior rhinomanometry) and reduced nasal secretion weights compared with placebo in a controlled-allergen-exposure paradigm. The consistent decongestant effects of desloratadine in pollen-chamber trials were also concordant with data from clinical trials conducted under natural, ambient exposure conditions. Taken together, these findings support the clinical utility of desloratadine, a nonsedating, long-acting, high-affinity H1 receptor antagonist with decongestant properties.





Similar content being viewed by others
References
Parikh A, Scadding GK. Seasonal allergic rhinitis. BMJ 1997; 314(7091): 1392–5
Sibbald B, Rink E. Epidemiology of seasonal and perennial rhinitis: clinical presentation and medical history. Thorax 1991; 46(12): 895–901
Young T, Finn L, Kim H. Nasal obstruction as a risk factor for sleep-disordered breathing. The University of Wisconsin Sleep and Respiratory Research Group. J Allergy Clin Immunol 1997; 99(2): S757–62
Spector SL. Overview of comorbid associations of allergic rhinitis. J Allergy Clin Immunol 1997; 99(2): S773–80
Fireman P. Otitis media and eustachian tube dysfunction: connection to allergic rhinitis. J Allergy Clin Immunol 1997; 99(2): S787–97
Griffin MP, McFadden ER, Ingram RH. Airway cooling in asthmatic and nonasthmatic subjects during nasal and oral breathing. J Allergy Clin Immunol 1982; 69(4): 354–9
Kirkpatrick MB, Sheppard D, Nadel JA, et al. Effect of the oronasal breathing route on the bronchoconstrictor response to sulfur dioxide in exercising asthmatic subjects. Am Rev Respir Dis 1982; 125: 627–31
Corren J. Allergic rhinitis and asthma: how important is the link? J Allergy Clin Immunol 1997; 99(2): S781–6
Horak F, Stübner UP, Zieglmayer R, et al. Onset and duration of action of desloratadine after controlled pollen challenges in patients with seasonal allergic rhinitis [abstract]. Allergy 2000; 55 Suppl. 63: 279
Horak F, Stübner UP, Zieglmayer R, et al. Decongestant activity of desloratadine versus placebo in allergic rhinitis: results from three single-dose, placebo-controlled, allergen chamber trials [abstract]. Allergy 2001; 56 Suppl. 68: 79
Cartier A, Bernstein IL, Burge PS, et al. Guidelines for bronchoprovocation on the investigation of occupational asthma. Report of the Subcommittee on Bronchoprovocation for Occupational Asthma. J Allergy Clin Immunol 1989; 84(5 Pt 2): 823–9
Bousquet J, Van Cauwenberge P, Khaltaev N (for the WHO) in collaboration with the World Health Organization. Allergic rhinitis and its impact on asthma. ARIA workshop report. 7: diagnosis and management. J Allergy Clin Immunol 2001; 108 Suppl. 5: S208–19
Craig TJ, Teets S, Lehman EB, et al. Nasal congestion secondary to allergic rhinitis as a cause of sleep disturbance and daytime fatigue and the response to topical nasal corticosteroids. J Allergy Clin Immunol 1998; 101: 633–7
Day JH, Briscoe MP, Welsh A, et al. Onset of action, efficacy, and safety of a single dose of fexofenadine hydrochloride for ragweed allergy using an environmental exposure unit. Ann Allergy Asthma Immunol 1997; 79(6): 533–40
Horak F, Stübner P, Zieglmayer R, et al. Controlled comparison of the efficacy and safety of cetirizine 10 mg o.d. and fexofenadine 120 mg o.d. in reducing symptoms of seasonal allergic rhinitis. Int Arch Allergy Immunol 2001; 125(1): 73–9
Day JH, Briscoe M, Widlitz MD. Cetirizine, loratadine, or placebo in subjects with seasonal allergic rhinitis: effects after controlled ragweed pollen challenge in an environmental exposure unit. J Allergy Clin Immunol 1998; 101(5): 638–45
Meltzer EO, the Desloratadine Study Group. Rapid onset of action of desloratadine in patients with seasonal allergic rhinitis [abstract 527]. J Allergy Clin Immunol 2001; 107: S160
Shapiro G, the Desloratadine Study Group. Decongestant effects of desloratadine in patients with seasonal allergic rhinitis and asthma [abstract 530]. J Allergy Clin Immunol 2001; 107: S161
Horak F, Jager S, Toth J, et al. Efficacy and tolerability of astemizole-D and loratadine-D during prolonged, controlled allergen challenge in the Vienna Challenge Chamber. Arzneimittelforschung 1996; 46(11): 1077–81
Horak F, Toth J, Marks B, et al. Efficacy and safety relative to placebo of an oral formulation of cetirizine and sustained-release pseudoephedrine in the management of nasal congestion. Allergy 1998; 53(9): 849–56
Corren J, Harris AG, Aaronson D, et al. Efficacy and safety of loratadine plus pseudoephedrine in patients with seasonal allergic rhinitis and mild asthma. J Allergy Clin Immunol 1997; 100(6 Pt 1): 781–8
Lorber R, Danzig M, the DL-D12 Study Group. Desloratadine and pseudoephedrine provide similar relief of nasal congestion in patients with seasonal allergic rhinitis [poster]. Presented at the XXth Congress of the European Academy of Allergology and Clinical Immunology; 2001 May 9–13; Berlin
Horak F, Stübner P, Zieglmayer R, et al. Effect of desloratadine versus placebo on nasal airflow and subjective measures of nasal obstruction in subjects with grass pollen induced allergic rhinitis in an allergen exposure unit. J Allergy Clin Immunol 2002; 109: 956–61
Settipane RA. Complications of allergic rhinitis. Allergy Asthma Proc 1999; 20(4): 209–13
Henriksen JM, Wenzel A. Effect of an intranasally administered corticosteroid (budesonide) on nasal obstruction, mouth breathing, and asthma. Am Rev Respir Dis 1984; 130(6): 1014–8
Marks MB. Allergy in relation to orofacial dental deformities in children: a review. J Allergy 1965; 36: 293–302
Proctor DF. The upper airways: I. nasal physiology and defense of the lungs. Am Rev Respir Dis 1977; 115: 97–129
Lipworth BJ, Jackson CM. Safety of inhaled and intranasal corticosteroids: lessons for the new millennium. Drug Saf 2000; 23(1): 11–33
Acknowledgements
Funding for these studies was provided by Essex Pharma GmbH (Schering-Plough Pharmaceuticals).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Horak, F., Stübner, P. Decongestant Activity of Desloratadine in Controlled-Allergen-Exposure Trials. Clin. Drug Investig. 22 (Suppl 2), 13–20 (2002). https://doi.org/10.2165/00044011-200222002-00002
Published:
Issue Date:
DOI: https://doi.org/10.2165/00044011-200222002-00002