Abstract
Objective: To identify the reasons for the large variation in renal clearance of mesalazine.
Design: Data were obtained from a randomised crossover bioequivalence study in 18 healthy volunteers.
Methods: Participants received a single 1000mg oral dose of each of two different formulations of prolonged release mesalazine (2 × 500mg tablets). The formulations had similar dissolution profiles in phosphate buffer pH 6.8. Plasma and urine mesalazine and acetylmesalazine concentrations were determined by validated methods involving high performance liquid chromatography analysis with mass spectrometric detection. Lower limits of quantification were 50 μg/L and 50 μg/L in plasma and 0.25 mg/L and 2 mg/L in urine, respectively.
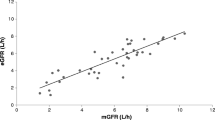
Results: There was a large variability in the release and absorption of mesalazine from both formulations and in the subsequent renal clearance of mesalazine. There was a clear distinction (p = 0.0009) in renal clearance between volunteers who showed slow mesalazine absorption with subsequent low clearance [0.006 to 0.5 L/h (0.1 to 8 ml/min)] and those who showed more extensive absorption followed by higher renal clearance [0.5 to 6 L/h (8 to 100 ml/min)]. Active tubular reabsorption with a saturable maximum must be the explanation for this difference in renal clearance. The metabolite acetylmesalazine is cleared renally via glomerular filtration and active tubular secretion, resulting in a clearance of 12 to 18 L/h (200 to 300 ml/min).
Conclusion: The renal clearance of mesalazine proceeds via the processes of glomerular filtration and active tubular reabsorption. Tubular reabsorption shows saturation at an area under the plasma concentration-time curve of 4 mg/L·h with an excreted amount of 2mg, resulting in a threshold clearance of 0.5 L/h (8 ml/min). This finding explains the dosage-dependent renal clearance of mesalazine reported in the literature, but has no clear clinical implications.





Similar content being viewed by others
References
Ahnfelt-Ronne I, Nielsen OH, Christensen A, et al. Clinical evidence supporting the radical scavenger mechanism of 5-aminosalicylic acid. Gastroenterology 1990; 98 (5 Pt 1): 1162–9
Prakash A, Markham A. Oral delayed-release mesalazine. A review of its use in ulcerative colitis and Crohn’s disease. Drugs 1999; 57: 383–408
Martindale. Reynolds JEF, editor. The extra pharmacopoeia. 31st ed. Gastro-intestinal agents. Mesalazine. London: Pharmaceutical Press, 1996: 1227–8
Yu DK, Morrill B, Eichmeier LS, et al. Pharmacokinetics of 5-aminosalicylic acid from controlled-release capsules in man. Eur J Clin Pharmacol 1995; 48: 273–7
Norlander B, Gotthard R, Ström M. Pharmacokinetics of a 5-aminosalicylic acid enteric-coated tablet in patients with Crohn’s disease or ulcerative colitis and in healthy volunteers. Aliment Pharmacol Ther 1990; 4: 497–505
Bondesen S, Hegnhoj J, Larsen F, et al. Pharmacokinetics of 5-aminosalicylic acid in man following administration of intravenous bolus and per os slow-release formulation. Dig Dis Sci 1991; 36: 1735–40
Brogden RN, Sorkin EM. Mesalazine. A review of its pharmacodynamic and pharmacokinetic properties, and therapeutic potential in chronic inflammatory bowel disease. Drugs 1989; 38: 500–23
Järnerot G. Newer 5-aminosalicylic acid based drugs in chronic inflammatory bowel disease. Drugs 1989; 37: 73–6
Meyers B, Evans DNW, Rhodes J, et al. Metabolism and urinary excretion of mesalazine in healthy volunteers given intravenously or released for absorption at different sites in the gastro-intestinal tract. Gut 1987; 28: 196–200
Allgayer H, Sonnenbichler J, Kruis W, et al. Determination of the pK values of 5-aminosalicylic acid and N-acetylsalicylic acid and comparison of the pH dependent lipid-water partition coefficients of sulphasalazine and its metabolites. Arzneimittelforschung 1985; 35: 1457–9
Nielsen OH, Bondesen S. Kinetics of 5-aminosalicylic acid after jejunal installation in man. Br J Clin Pharmacol 1983; 16: 738–40
Jacobsen BA, Abildgaard K, Rasmussen HH, et al. Availability of mesalazine from enemas and suppositories during steady state conditions. Scand J Gastroenterol 1991; 26: 374–8
Vree TB, Dammers E, Exler PS, et al. Mono and biphasic plasma concentration-time curves of mesalazine from a 500 mg suppository in 24 healthy male volunteers controlled by the time of defecation before dosing. J Pharm Pharmacol 2000. In press
Yu DK, Elvin AT, Morrill B, et al. Effect of food coadministration on 5-aminosalicylic acid oral suspension bioavailability. Clin Pharmacol Ther 1990; 48: 26–33
Norlander B, Gotthard R, Ström M. Steady-state pharmacokinetics of enteric coated 5-aminosalicylic acid tablets in healthy volunteers and in patients with Crohn’s disease or ulcerative colitis. Aliment Pharmacol Ther 1991; 5: 291–300
Gionchetti P, Campieri M, Belluzzi A, et al. Bioavailability of single and multiple doses of a new oral formulation of 5-ASA in patients with inflammatory bowel disease and healthy volunteers. Aliment Pharmacol Ther 1994; 8: 535–40
Vree TB, Hekster YA, Baars AM, et al. Pharmacokinetics of sulphamethoxazole in man: effects of urinary pH and urine flow on metabolism and renal excretion of sulphamethoxazole and its metabolite N4-acetylsulphamethoxazole. Clin Pharmacokinet 1978; 3: 319–29
Vree TB, van Ewijk-Beneken Kolmer EWJ, Verwey-Van Wissen CPWM, et al. Effect of urinary pH on the pharmacokinetics of salicylic acid, with its glycine and glucuronide conjugates in human. Int J Clin Pharmacol Ther 1994; 32: 550–8
Boom, SP, Wouterse AC, Vree TB, et al. Renal tubular excretion of the N4-acetyl metabolites of sulphasomidine and sulphadimethoxine in the dog. J Pharm Pharmacol 1993; 45: 614–7
Boom SP, Hoet S, Russel FG. Saturable urinary excretion kinetics of famotidine in the dog. J Pharm Pharmacol 1997; 49: 288–92
Boom SP, Meyer I, Wouterse AC, et al. A physiologically based kidney model for the renal clearance of ranitidine and the interaction with cimetidine and probenecid in the dog. Biopharm Drug Dispos 1998; 19: 199–208
Bondesen S, Rasmussen SN, Rask-Madsen J, et al. 5-Aminosalicylic acid in the treatment of inflammatory bowel disease. Acta Med Scand 1987; 221: 227–42
Diener U, Tuczek HV, Fischer C, et al. Renal function was not impaired by treatment with 5-aminosalicylic acid in rats and man. Naunyn Schmiedebergs Arch Pharmacol 1984; 326: 278–82
Calder IC, Funder CC, Gree CR, et al. Nephrotoxic lesions from 5-aminosalicylic acid. BMJ 1972; 1: 152–4
Noble E, Janssen L, Dierickx PJ. Comparative cytotoxicity of 5-aminosalicylic acid (mesalazine) and related compounds in different cell lines. Cell Biol Toxicol 1997; 13: 445–51
Fraser JS, Smith D, Lamb E, et al. Prospective study of the effect of 5-aminosalicylic acid (mesalazine) on renal function in inflammatory bowel disease [abstract no. 4377]. Proceedings Digestive Disease Week. 1999 May 16–19; Orlando, Florida, USA; 1999: A-794-5
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Vree, T.B., Dammers, E., Exler, P.S. et al. Saturable Active Tubular Reabsorption in the Renal Clearance of Mesalazine in Human Volunteers. Clin. Drug Investig. 20, 35–42 (2000). https://doi.org/10.2165/00044011-200020010-00005
Published:
Issue Date:
DOI: https://doi.org/10.2165/00044011-200020010-00005