Summary
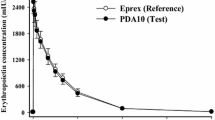
Two studies were performed to assess the tolerability of a new multidose formulation of epoetin beta that contains benzalkonium chloride 0.002% and benzyl alcohol 0.4%. In study 1,12 healthy volunteers received 2ml subcutaneous injections of placebo epoetin beta lyophilisate dissolved in either the multidose solvent or water for injections. There were no significant differences between treatments in the rating of subjective sensations (pain, itching, numbness and tenderness) and local reactions (swelling, induration, redness and heat). Study 2, a single-blind, randomised trial, compared the tolerability and clinical efficacy of multidose epoetin beta (n = 244; subcutaneous 171, intravenous 73) with that of the monodose formulation (n = 62; subcutaneous 44; intravenous 18) in patients with end-stage renal failure. Mild local reactions were experienced by 1 multidose subcutaneous recipient and 3 monodose subcutaneous recipients; no local reactions were recorded following intravenous administration of either formulation. The overall incidence of adverse events was low with both formulations, and there was no difference in clinical efficacy (assessed by haematological parameters and iron turnover) between the two formulations. These results indicate that the multidose formulation of epoetin beta is at least as well tolerated as the monodose formulation.
Similar content being viewed by others
References
Dunn CJ, Markham A. Epoetin beta. A review of its pharmacological properties and clinical use in the management of anaemia associated with chronic renal failure. Drugs 1996; 51(2): 299–318
Eschbach JW, Egrie JC, Downing MR, et al. Correction of the anaemia of end-stage renal disease with recombinant human erythropoietin. N Engl J Med 1987; 316: 73–8
Recormon® Data Sheet 1996
Expert Report on the chemical, pharmaceutical and biological documentation for NeoRecormon 50 000 and 10 000 IU multidose vials and 10 000 and 20 000 IU cartridges. Boehringer Mannheim report no AZ 613, 1995
Samtleben W, Baldamus CA, Bommer J, et al. Indications and contraindications for recombinant human erythropoietin treatment. Results in hemodialysis patients. Nephrol 1989; 76: 193
Scigalla P, Wieczorek L, Bicker U. Treatment of renal anaemia with recombinant human erythropoietin: European experience. In: Garnick MB, editor. Erythropoietin in clinical applications. New York, M Dekker, 1990: 141
Eschbach JW. The future of rhEPO. Nephrol Dial Transplant 1995; lOSuppl. 2: 96–109
Macdougall IC. How to get the best out of rhEPO. Nephrol Dial Transplant 1995; 10Suppl. 2: 85–91
Adverse event reporting on epoetin beta. Boehringer Mannheim Documentation AZ 510
Shannon KM, Keith JF, Mentzer WC, et al. Recombinant human erythropoietin stimulates erythropoiesis and reduces erythrocyte transfusions in very low birth weight preterm infants. Paediatrics 1995; 95(1): 1–9
American Academy of Pediatrics. Benzyl alcohol: toxic agent in neonatal use. Paediatrics 1983; 72: 356–7
Santiero ML. Benzyl alcohol toxicity in newborn infants. J Hosp Pharm 1989; 9: 17–9
Acknowledgements
These studies were sponsored by Boehringer Mannheim GmbH, Germany.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Franke, W., Scherhag, R. Tolerability and Efficacy of Multidose Formulations of Epoetin Beta. Clinical Drug Investigation 13, 199–206 (1997). https://doi.org/10.2165/00044011-199713040-00004
Published:
Issue Date:
DOI: https://doi.org/10.2165/00044011-199713040-00004