Abstract
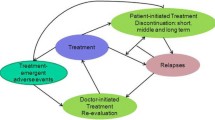
Schizophrenia is one of the most expensive psychiatric conditions because of high direct and indirect costs associated with the nature of the illness, its resistance to treatment and the consequences of relapse. Long-acting risperidone is a new formulation of an atypical antipsychotic drug that also offers the improvements in compliance associated with haloperidol depot. The aim of this simulation study was to compare the benefits and costs of three pharmacological treatment strategies comprising first-line treatment with long-acting risperidone injection, a haloperidol depot or an oral atypical antipsychotic agent, over a 5-year period in Germany. A discrete event simulation model was developed to compare three treatment scenarios from the perspective of major third-party payers (sickness funds and social security ’sozialversicherung’). The scenarios comprised first-line treatment with haloperidol depot (scenario 1), long-acting risperidone (scenario 2) and oral olanzapine (scenario 3). Switches to second or third-line options were allowed when side-effects occurred or a patient suffered more than a fixed number of relapses. The model accounted for fixed patient characteristics, and on the basis of these, simulated patient histories according to several time-dependent variables. The time horizon for this model was limited to 5 years, and in accordance with German guidelines, costs and effects were discounted by between 3 and 10%. Direct costs included medication, type of physician visits and treatment location. Indirect costs were not included. Information on treatment alternatives, transition probabilities, model structure and healthcare utilization were derived from the literature and an expert panel. Outcomes were expressed in terms of the number and duration of psychotic episodes, cumulative symptom scores, costs, and quality-adjusted life-years (QALY). Univariate sensitivity analyses were carried out, as were subgroup analyses based on disease severity and for patients at high risk of being non-compliant. The long-acting risperidone strategy was calculated to avoid 0.23 and 0.33 relapses per patient, decrease the cumulative symptom score by 25 and 33 points, and decrease the costs by €2017 and €6096 per patient (€1608 and €5422 discounted), compared with the haloperidol depot and olanzapine strategies, respectively, over a 5-year period (year of costing 2004). Among high-risk non-compliant patients, long-acting risperidone was estimated to avoid 0.23 and 0.47 relapses and save €4822 and €10 646 per patient (€4107 and €9490 discounted), compared with the haloperidol depot and olanzapine strategies, respectively. Sensitivity analyses showed that the results were robust and mainly sensitive to changes in the reported relative effectiveness of atypical and conventional formulations for preventing symptom recurrence, and in the relative compliance with oral and long-acting formulations. In this model, long-acting risperidone is a dominant strategy compared with a haloperidol depot or oral atypical antipsychotic agent, being both more effective and less costly over a 5-year period. Results for longacting risperidone are even more favourable among patients at high risk of being noncompliant or with more severe disease.








Similar content being viewed by others
References
Kompetenznetz Schizophrenie. Laien und Betroffene: Krankheitsbild: Verlauf [online]. Available from URL: http://www.kompetenznetzschizophrenie.de [Accessed 2 September 2004]
American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders: DSM-IV. Washington, DC, USA: American Psychiatric Publishing, Inc., 1994
Davies LM, Drummond MF. The econimic burden of schizophrenia. Psychiatr Bull 1990; 14: 522–5
McGrath J, Saha S, Welham J, et al. A systematic review of the incidence of schizophrenia: the distribution of rates and the influence of sex, urbanicity, migrant status and methodology. BMC Med 2004; 2: 13 Available from URL: http://www.biomedcentral.com/1741-7015/2/13
Knapp M, Mangalore R, Simon J. The global costs of schizophrenia. Schizophr Bull 2004; 30: 279–93
Salize HJ. Die Kosten der Schizophrenie — was wissen wir (nicht)? Psychiat Prax 2001; 28 (Suppl. 1): 21–8
Almond S, Knapp M, Francois C, et al. Relapse in schizophrenia: costs, clinical outcomes and quality of life. Br J Psychiatry 2004; 184: 346–51
Carr V, Hocking B, Jablensky A, et al. Schizophrenia: costs. An analysis of the burden of schizophrenia and related suicide in Australia. An Access Economics Report for SANE Australia; 2002
American Psychiatric Association. Practice guideline for the treatment of patients with schizophrenia. Am J Psychiatry 1997; 154: 1–63
National Institute for Clinical Excellence. (2002). Schizophrenia: core interventions in the treatment and management of schizophrenia in primary and secondary care. London NICE [online]. Available from URL: http://www.nice.org.uk/page.aspx?o=42461 [Accessed 10 March 2005]
Harrison TS, Goa KL. Long-acting risperidone: a review of its use in schizophrenia. CNS Drugs 2004; 18: 113–32
Newman SCBR. Mortality in a cohort of patients with schizophrenia. Can J Psychiatry 1991; 36: 239–45
Carpenter WT Jr, Buchanan RW. Schizophrenia. N Engl J Med 1994; 330: 681–90
Csernansky JG, Mahmoud R, Brenner R. A comparison of risperidone and haloperidol for the prevention of relapse in patients with schizophrenia. N Engl J Med 2002; 346: 16–22
McIntyre R, Trakas K, Hwang P, et al. Risk of adverse events associated wtih antipsychotic treatment: results from the Canadian National Outcomes Measurement Study in Schizophrenia (CNOMSS). European Neuropsychopharmacology The Journal of the European College of Neuropsychopharmacology Volume 11, Supplement 3, October 2001, S269 (abstract number: P.2.061)
Tollefson GD, Beasley CM Jr, Tran PV, et al. Olanzapine versus haloperidol in the treatment of schizophrenia and schizoaffective and schizophreniform disorders: results of an international collaborative trial. Am J Psychiatry 1997; 154: 457–65
Wirshing DA, Wirshing WC, Kysar L, et al. Novel antipsychotics: comparison of weight gain liabilities. J Clin Psychiatry 1999; 60: 358–63
Bundesverband der Betriebskrankenkassen BKK (Hrsg.). Hauptabteilung Wirtschaft und Statistik. Krankheitsartenstatistik 2001–2002, Essen, Germany, 2003
Davis JM, Chen N, Glick ID. A meta-analysis of the efficacy of second-generation antipsychotics. Arch Gen Psychiatry 2003; 60: 553–64
Remington GJ, Adams ME. Depot neuroleptic therapy: clinical considerations. Can J Psychiatry 1995; 40 (Suppl. 1): S5–11
Gerlach J. Depot neuroleptics in relapse prevention: advantages and disadvantages. Int Clin Psychopharmacol 1995; 9 (Suppl. 5): 17–20
Olfson M, Mechanic D, Hansell S, et al. Predicting medication non-compliance after hospitalization discharge among patients with schizophrenia. Psychiatry Services 2000; 51: 216–22
Chouinard G, Albright PS. Economic and health state utility determinations for schizophrenic patients treated with risperidone or haloperidol. J Clin Psychopharmacol 1997; 17: 298–307
Baron F. Pharmacoeconomic guidelines: per country and international comparisons; current situation and perspectives. Chassieu: GYD Institute, 2002
Hjelmgren J, Berggren F, Andersson F. Health economic guidelines — similarities, differences and some implications. Value Health 2001; 4: 225–50
Rote Liste Service GmbH (ed.) ROTE LISTE1 2004: Arzneimittelverzeichnis für Deutschland (einschlieblich EU-Zulassungen und bestimmter Medizinprodukte). Aulendorf, Germany, 2004
Salize HJ, Rossler W. The cost of comprehensive care of people with schizophrenia living in the community. A cost evaluation from a German catchment area. Br J Psychiatry 1996; 169: 42–8
Barnes TR, Curson DA. Long-term depot antipsychotics. A risk—benefit assessment. Drug Safety 1994; 10: 464–79
Glazer WM, Kane JM. Depot neuroleptic therapy: an underutilized treatment option. J Clin Psychiatry 1992; 53: 426–33
Bestehorn M, Tischer B, Glaser P, et al. Representative study of the epidemiology of schizophrenia patients in health care facilities in Germany [in German]. Fortschr Neurol Psychiatry 1999; 67: 487–92
Gaebel W, Pietzcker A. Prospective study of course of illness in schizophrenia: Part III. Treatment and outcome. Schizophr Bull 1987; 13: 307–16
Weiden P, Glazer W. Assessment and treatment selection for “revolving door” inpatients with schizophrenia. Psychiatr Q 1997; 68: 377–92
Chue P, Devos E, Duchesne I, et al. Hospitalisation rates in patients during long-term treatment with long-acting risperidone injection. The International Journal of Neuropsychopharmacology 2002; 5 (Suppl. 1): S188–9
Foster RH, Goa KL. Risperidone. A pharmacoeconomic review of its use in schizophrenia. PharmacoEconomics 1998; 14: 97–133
De Graeve D, Smet A, Mehnert A, et al. Long-acting risperidone compared with oral olanzapine and haloperidol depot in schizophrenia: a Belgian cost-effectiveness analysis. PharmacoEconomics 2005; 23 (Suppl. 1): 35–47
Heeg BM, van Aalst G, van den Arend IJ, et al. A discrete events model of long-term outcomes and cost of treatment with long acting risperidone in schizophrenia. Value in Health 2003; 6: 515–6
Chue PS, Heeg BMS, Buskens E, et al. Modelling the impact of compliance on the costs and effects of longacting risperidone in Canada. PharmacoEconomics 2005; 23 (Suppl. 1): 62–74
Mantovani LG, Berto P, D’Ausilio A, et al. Costs and effects of long-acting risperidone (LAR) versus atypical antipsychotics in the treatment of schizophrenic patients in Italy. Farmeconomica e percosi terapeutica 2004; 5: 5–11
Acknowledgements
The authors would like to thank the following persons who formed the expert panel: Professors M. Gastpar, J. Klosterkötter, H.-J. Möller, D. Naber and M. Schmauss; Professor J. Fritze, who provided estimates of location costs; and Dr G. van Aalst, who advised on inputs into the core model. They would also like to thank A. van Gestel and E. Buskens for their help during the project. Finally, the authors would like to thank Janssen Pharmaceutica, N.V., Belgium and Janssen-Cilag Gmbtl Germany for financing the development of the model.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Laux, G., Heeg, B.M.S., van Hout, B.A. et al. Costs and effects of long-acting risperidone compared with oral atypical and conventional depot formulations in Germany. Pharmacoeconomics 23 (Suppl 1), 49–61 (2005). https://doi.org/10.2165/00019053-200523001-00005
Published:
Issue Date:
DOI: https://doi.org/10.2165/00019053-200523001-00005