Abstract
Objective:
Although nonsteroidal anti-inflammatory drugs (NSAIDs) are an effective therapy for rheumatoid arthritis, they are associated with significant adverse effects, the management of which imposes additional costs on the healthcare system. Prescribing NSAIDs which have a lower risk of major adverse effects as the first-line NSAID for patients with rheumatoid arthritis and osteoarthritis may be expected to lead to an improvement in clinical outcomes and reduce overall treatment costs. This analysis examines data from a published randomised controlled trial of 5 NSAIDs to explore these hypotheses.
Design and Setting:
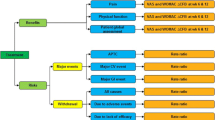
Data from a clinical trial comparing 5 NSAIDs were combined with published cost data to construct 2 clinical decision models, reflecting alternative approaches to the management of major and minor adverse effects in the UK.
Interventions:
The 5 NSAIDs evaluated in the analysis were nabumetone, diclofenac, ibuprofen, piroxicam and naproxen, although only the results for ibuprofen and nabumetone are reported.
Main outcome measures and results:
The total cost of care per patient receiving nabumetone was estimated to be between 25 pounds sterling (£) and £41 more expensive than ibuprofen. In a hypothetical cohort of 100 000 patients, there were between 690 and 821 more major adverse effects using ibuprofen than nabumetone. The cost per life-year gained (LYG) from using nabumetone rather than ibuprofen ranged between £1880 and £2517 (1995 values), depending upon the management of adverse effects.
Conclusions:
These results indicate that: (i) prescribing the newer, currently more expensive, NSAIDs will not necessarily lead to cost savings; (ii) the management of adverse effects can have a significant impact on costs; and (iii) the additional cost may be justifiable in terms of the mortality and morbidity gains associated with the new lower-risk NSAIDs.
Similar content being viewed by others
References
British Medical Association (BMA) and The Royal Pharmaceutical Society of Great Britain. British national formulary number 29. London: BMA, 1995
Wynne HA, Campbell M. Pharmacoeconomics of nonsteroidal anti-inflammatory drugs (NSAIDs). Pharmacoeconomics 1994; 3 (2): 107–23
McIntosh E. The cost of rheumatoid arthritis. Br J Rheumatol 1996; 35 (8): 781–90
Bloor K, Maynard A. Is there scope for improving the costeffective prescribing of nonsteroidal anti-inflammatory drugs? Pharmacoeconomics 1996; 9 (6): 484–96
Eversmeyer W, Poland M, DeLapp RE, et al. Safety experience with Nabumetone versus Diclofenac, Naproxen, Ibuprofen and Piroxicam in osteoarthritis and rheumatoid arthritis. Am J Med 1993; 95 (S2A): 10S–8S
Weinstein MC, Fineberg HV. Clinical decision analysis. Philadelphia: WB Saunders, 1980
Sheldon T. Problems of using modelling in the economic evaluation of health care. Health Econ 1996; 5: 1–11
Lister BJ, Poland M, DeLapp RE. Efficacy of Nabumetone versus Diclofenac, Naproxen, Ibuprofen and Piroxicam in osteoarthritis and rheumatoid arthritis. Am J Med 1993; 95 Suppl. 2A: 2S–9S
Department of Health and Welsh Office. Drug Tariff NHS England and Wales. London: HMSO, 1994
Chemist and Druggist Monthly Price List. Tonbridge: Benn Publications Ltd., 199510. Royal College of Physicians (RCP). Upper gastrointestinal haemorrhage: guideline for good practice and audit of management. London: RCP Publications
Hudson N, Faulkner G, Smith SJ, et al. Morbidity and treatment in elderly patients surviving hospital admission with bleeding peptic ulcer. Gut 1995; 37: 182–6
Department of Health and Personal Social Services. Statistics for England. 1994 ed. London: HMSO, 1995
Allan R, Dykes P. A study of the factors influencing mortality rates from gastrointestinal haemorrhage. Q J Med 1976; 45: 533–50
Hunt PS, Hansky K, Korman MG. Mortality in patients with haematemesis and melaena: a prospective study. BMJ 1979; I: 1239–40
Katschinski BD, Logan RFA, Davies J, et al. Audit of mortality in upper gastrointestinal haemorrhage. Postgrad Med J 1989; 65: 913–7
Department of Health. Register of cost-effectiveness studies. London: HMSO, 1994
Charlton J, Murphy M. The health of adult Britain 1841-1994. Vol. 2. London: HMSO, 19
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
McCabe, C.J., Akehurst, R.L., Kirsch, J. et al. Choice of NSAID and Management Strategy in Rheumatoid Arthritis and Osteoarthritis. Pharmacoeconomics 14, 191–199 (1998). https://doi.org/10.2165/00019053-199814020-00007
Published:
Issue Date:
DOI: https://doi.org/10.2165/00019053-199814020-00007