Summary
Synopsis
Lansoprazole is a proton pump inhibitor that reduces gastric acid secretion. It has proved effective in combination regimens for the eradication of Helicobacter pylori and as monotherapy to heal and relieve symptoms of gastric or duodenal ulcers and gastro-oesophageal reflux. After initial healing, it may be used to prevent recurrence of oesophageal erosions or peptic ulcers in patients in whom H. pylori is not the major cause of ulceration and to reduce basal acid output in patients with Zollinger-Ellison syndrome. Usual dosages are 15 to 60 mg/day, although dosages of ≤ 180 mg/day have been used in patients with hypersecretory states.
In patients with duodenal or gastric ulcer, short term lansoprazole monotherapy was similar to omeprazole and superior to histamine H2 receptor antagonists in achieving healing rates >90%. Lansoprazole was as effective a component of H. pylori eradication regimens as omeprazole, tripotassium dicitrato bismuthate (colloidal bismuth subcitrate) or ranitidine.
Lansoprazole was superior to ranitidine in symptom relief and healing of gastro-oesophageal reflux disease and tended to relieve symptoms more rapidly than omeprazole, although initial healing was similar. As maintenance treatment, lansoprazole was similar to omeprazole and superior to ranitidine in relieving symptoms and preventing relapse. Lansoprazole was also superior to ranitidine in healing and relieving symptoms of oesophageal erosions associated with Barrett’s oesophagus; healing was maintained for a mean of 2.9 years in ≥ 70% of patients. Lansoprazole was also superior to ranitidine in prophylaxis of redilatation of oesophageal strictures.
After ≥ 4 years of use in patients with Zollinger-Ellison syndrome, lansoprazole 60 to 180 mg/day effectively controlled basal acid output. Dosages may be reduced in some patients once healing and symptom relief has been achieved. Preliminary studies of lansoprazole in patients at risk of aspiration pneumonia or stress ulcers show promise. Although studies show lansoprazole is potentially effective in treating gastrointestinal bleeding, future studies should assess patients’ H. pylori status.
Lansoprazole has been well tolerated in clinical trials, with headache, diarrhoea, dizziness and nausea appearing to be the most common adverse effects. Tolerability of lansoprazole does not deteriorate with age and the drug is well tolerated in long term use (≤ 4 years) in patients with Zollinger-Ellison syndrome or reflux disease.
Thus, lansoprazole is an important alternative to omeprazole and H2 receptor antagonists in acid-related disorders. In addition to its efficacy in healing or maintenance treatment, it may provide more effective symptom relief than other comparator agents.
Pharmacological Properties
Lansoprazole provides dose-related inhibition of gastric acid secretion via inhibition of H+/K+-adenosine triphosphatase in gastric parietal cells, with doses of 30 to 60mg producing similar acid suppression to omeprazole 40mg. Mean 24-hour gastric pH levels are higher when lansoprazole is given in 2 divided doses rather than 1 dose daily. Lansoprazole is 2 to 8 times more potent than omeprazole in inhibitory effects on H. pylori in vitro and is associated with dose-dependent killing of the bacterium.
Orally administered lansoprazole is well absorbed, with peak plasma concentrations (Cmax) linear over the dose range from 15 to 60mg. Sucralfate does reduce and food may reduce absorption of lansoprazole, and lansoprazole may reduce concentrations of theophylline. Both lansoprazole and the 14-hydroxy metabolite of clarithromycin are found in greater concentrations in plasma when administered together with amoxicillin. The drug is hepatically biotransformed and excreted through the bile and kidneys. The elimination half-life is ≈1 hour. Although clearance is reduced in poor metabolisers of S-mephenytoin, elderly patients and those with hepatic cirrhosis or undergoing haemodialysis, dosage adjustments are usually not considered except for patients with severe liver failure. For ease of administration, the enteric-coated granules in lansoprazole capsules may be mixed with apple sauce or juice before administration, with no major changes in pharmacokinetics.
Clinical Efficacy
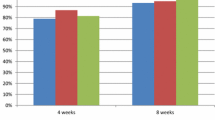
In peptic ulcer healing and symptom relief, lansoprazole 15 to 60 mg/day monotherapy for 4 to 8 weeks was similar or superior in efficacy to omeprazole 20 to 40 mg/day. Lansoprazole 30 mg/day was similar or superior to ranitidine and superior to famotidine in ulcer healing. When used in combination with ≥2 antibacterials, lansoprazole 30 to 60 mg/day was an effective part of regimens capable of achieving eradication of H. pylori in ≥90% of patients, although lansoprazole monotherapy was ineffective. The probability of achieving eradication in ≥90% of patients was highest in regimens incorporating ≥2 antibacterial agents and using ≤4 weeks of lansoprazole 60 mg/day. Healing rates seen in a subset of ≥1300 patients from these H. pylori eradication studies (receiving mono-, dual or triple therapy) were similar to those seen in earlier lansoprazole monotherapy trials. Healing was not affected by the number of agents in the regimen, the lansoprazole dosage or the ability of the regimen to eradicate H. pylori. As part of regimens with ≥2 antibacterial agents, lansoprazole 60 mg/day was as effective as omeprazole 40 mg/day, tripotassium dicitrato bismuthate 120mg 4 times daily or ranitidine 300 mg/day in H. pylori eradication and was similar to omeprazole and tripotassium dicitrato bismuthate but superior to cimetidine in achieving ulcer healing.
As initial treatment of gastro-oesophageal reflux disease (GORD), lansoprazole 15 to 60 mg/day is superior to ranitidine 150mg twice daily in 2-week symptom relief and 4-week healing. Lansoprazole 30 mg/day and famotidine 40 mg/day for 4 weeks achieved similar healing rates, but relapse was more common in patients subsequently maintained on famotidine. No statistically significant differences in GORD healing rates were seen between omeprazole 20 or 40 mg/day and lansoprazole 15 or 30 mg/day, but lansoprazole tended to relieve symptoms more rapidly than omeprazole. Although clear dose-response effects have not been seen in GORD healing across trials, superior symptom relief appears to accompany 30 or 60 mg/day dosages. Following initial GORD healing, lansoprazole 15 and 30 mg/day or omeprazole 20 mg/day for 12 months did not differ in prevention of symptoms or relapse, although both drugs were superior to ranitidine or placebo. Lansoprazole also heals and maintains healing of erosions resistant to H2 receptor antagonists.
Greater rates of endoscopically confirmed healing and relief of symptoms of oesophageal erosions associated with Barrett’s oesophagus were found with lansoprazole 30 to 60 mg/day than with ranitidine 150mg twice daily for 8 weeks. 70% of patients achieved healing and symptom relief during long term (mean 2.9 years) treatment of Barrett’s oesophagus with lansoprazole 60 mg/day. Fewer lansoprazole 30 mg/day than ranitidine 300 mg/day recipients with oesophageal strictures required redilatation during 12 months of treatment, but most of those required the procedure during the first 6 months of treatment.
In patients treated for up to 4 years, lansoprazole 60 to 180 mg/day effectively reduced basal acid output by 95% or to <10 mmol/h and healed ulcers in patients with Zollinger-Ellison syndrome. Preliminary studies of lansoprazole in patients at risk of stress ulcers or of aspiration pneumonia suggest that this treatment has promise; lansoprazole 30mg on the evening before and morning of surgery was similar in efficacy to ranitidine 150mg on the morning of surgery in preventing aspiration pneumonitis. Preliminary results showing that lansoprazole 30 to 60 mg/day can halt or reduce the risk of gastrointestinal bleeding are encouraging for future studies in this indication.
Tolerability
In clinical trials involving >5000 patients receiving lansoprazole 7.5 to 60 mg/day monotherapy or comparators only 2.1 % of lansoprazole recipients withdrew from treatment (vs 2.6% receiving ranitidine, 1.2% on famotidine or 1.6% on omeprazole). The most common adverse events included headache (8.8%), diarrhoea (3.5%), nausea (2.0%), dizziness (1.6%), vomiting (1.4%) and constipation (1.0%). However, when non-drug-related adverse events were excluded, only headache, diarrhoea, dizziness and nausea occurred in >1 % of patients. There was no increase in adverse events in elderly patients, in whom headache (4%), diarrhoea (4.7%), nausea (2.9%), dizziness (2.9%) and constipation (2.9%) were most common. Serious adverse events occurred in only 0.6% of lansoprazole recipients in clinical trials and no patterns occurred to suggest a specific mechanism for these events.
Lansoprazole is generally well tolerated in long term treatment in patients with GORD or Zollinger-Ellison syndrome. No significant differences in the number of adverse events with lansoprazole compared with omeprazole, ranitidine or placebo have been observed in long term studies. Diarrhoea appeared to be the most commonly reported adverse event associated with long term lansoprazole treatment.
Pharmacoeconomic Assessment
In H. pylori eradication regimens in Canada, lansoprazole and omeprazole triple therapies appear less expensive than other triple therapy, dual therapy or H2 receptor antagonist monotherapy regimens. A UK model assessing direct medical costs after lansoprazole, ranitidine or omeprazole therapy reported these 3 agents to be similarly cost effective in peptic ulcer or dyspepsia patients. Lansoprazole, however, was clearly more cost effective than the other 2 agents in the treatment of GORD.
In the US, lansoprazole was shown to be a less costly treatment of GORD than omeprazole, laparoscopic fundoplication surgery or brandname H2 receptor antagonists. However, generic ranitidine or cimetidine were less expensive than lansoprazole in GORD treatment.
In the prevention of GORD relapse, lansoprazole was more cost effective than high dose H2 receptor antagonists and similar to standard doses of H2 receptor antagonists in some patients.
Dosage and Administration
Usual dosages of lansoprazole for use in the treatment of gastric or duodenal ulcer (ulcer healing or H. pylori eradication) or reflux oesophagitis healing are 15 to 60 mg/day for 4 to 8 weeks, with dosages of 15 to 30 mg/day for maintenance treatment, where indicated. The drug is generally administered before meals and is given either once daily in the morning or in divided doses morning and evening. Patients with Zollinger-Ellison syndrome usually receive 60 mg/day initially, with the dose then titrated to reduce basal acid output to <5 or <10 mmol/L. Dosage adjustments may be considered in patients with severe liver disease, but are not required for elderly patients or those with renal insufficiency. The use of lansoprazole in children has not been fully studied.
Similar content being viewed by others
References
Barradell LB, Faulds D, McTavish D. Lansoprazole: a review of its pharmacodynamic and pharmacokinetic properties and its therapeutic efficacy in acid-related disorders. Drugs 1992; 44: 225–50
Spencer CM, Faulds D. Lansoprazole: a reappraisal of its pharmacodynamic and pharmacokinetic properties, and its therapeutic efficacy in acid-related disorders. Drugs 1994 Sep; 48: 404–30
Nagaya H, Satoh H, Kubo K, et al. Possible mechanism for the inhibition of gastric (H+ + K+)-adenosine triphosphatase by the proton pump inhibitor AG-1749. J Pharmacol Exp Ther 1989; 248: 799–805
Timmer W, Ripke H, Kleist P, et al. Effect of four lansoprazole dose levels and one dosage regimen of omeprazole on 24-hour intragastric pH in healthy subjects. Methods Find Exp Clin Pharmacol 1995; 17(7): 489–95
Blum RA, Vermeire GD, Shi H, et al. Gastric pH levels with lansoprazole 15 mg and 30 mg qd, omeprazole 20 mg qd, and ranitidine 150 mg qid [abstract]. Gastroenterology 1997 Apr; 112(4): A73
Tolman KG, Sanders SW, Buchi KN. Gastric pH levels after 15mg and 30mg of lansoprazole and 20mg omeprazole [abstract]. Gastroenterology 1994 Apr; 106 Suppl.: 197
Sakaguchi M, Ashida K, Umegaki E, et al. Suppressive action of lansoprazole on gastric acidity and its clinical effect in patients with gastric ulcers: comparison with famotidine. J Clin Gastroenterol 1995; 20 Suppl. 2: S27–31
Florent C, Forestier S. Twenty-four-hour monitoring of intragastric acidity: comparison between lansoprazole 30mg and pantoprazole 40mg. Eur J Gastroenterol Hepatol 1997; 9: 195–200
Dammann H-G, Fuchs W, Richter G, et al. Lansoprazole versus omeprazole: influence on meal-stimulated gastric acid secretion. Aliment Pharmacol Ther 1997; 11: 359–64
Blum RA, Hunt RH, Kidd SL, et al. Pharmacodynamics of various doses of lansoprazole [abstract no. 75]. Am J Gastroenterol 1994; 89(9): 1634
Yamamoto I, Fukuda Y, Okui M, et al. Proton pump inhibitors for Helicobacter pylori eradication in patients with peptic ulcer. J Clin Gastroenterol 1995; 20 Suppl. 1: S38–42
Vogt K, Hahn H. Bactericidal activity of lansoprazole versus omeprazole in vitro [abstract]. Gut 1996; 39 Suppl. 2: 9–10
Vicari F, Foliguet B, Conroy MC, et al. Comparative in vitro activity of 8 metal salts, lansoprazole and cefotiam against clinical isolates of H. pylori [abstract]. Gastroenterology 1995; 108(4) Suppl.: A936
Figura N, Crabtree JE, Datillo M. In-vitro activity of lansoprazole against Helicobacter pylori. J Antimicrob Chemother 1997; 39: 589–90
Tateno M, Nakamura N. Phase I study of lansoprazole (AG-1749) antiulcer agent: capsule form. Rinsho Iyaku 1991; 7(1): 51–62
Gerloff J, Mignot A, Barth H, et al. Pharmacokinetics and absolute bioavailability of lansoprazole. Eur J Clin Pharmacol 1996 Jun; 50: 293–7
Nakamura M, Oda M, Akiba Y, et al. Autoradiographic demonstration of lansoprazole uptake sites in rat antrum and colon. J Clin Gastroenterol 1995; 20 Suppl. 2: S8–S13
Pearce RE, Rodrigues AD, Goldstein JA, et al. Identification of the human P450 enzymes involved in lansoprazole metabolism. J Pharmacol Exp Ther 1996 May; 277: 805–16
Sohn D-R, Kwon J-T, Kim H-K. Influence of S-mephenytoin phenotype on the lansoprazole metabolism [abstract]. Clin Pharmacol Ther 1996 Feb; 59: 137
Delhotal-Landes B, Flouvat B, Duchier J, et al. Pharmacokinetics of lansoprazole in patients with renal or liver disease of varying severity. Eur J Clin Pharmacol 1993; 45: 367–71
Katsuki H, Yagi H, Arimori K, et al. Determination of R(+)- and S(−)-lansoprazole using chiral stationary-phase liquid chromatography and their enantioselective pharmacokinetics in humans. Pharm Res 1996 Apr; 13: 611–5
Danz-Neeff H, Brunner G. Comparative kinetic studies with the three proton pump inhibitors omeprazole, lansoprazole and pantoprazole in patients with complete liver cirrhosis [abstract]. Gastroenterology 1996 Apr; 110 Suppl.: A90
Siringo S, Bolondi L, Gramantieri L, et al. Pharmacokinetic of lansoprazole in cirrhotics with different degree of liver function derangement [abstract]. Gastroenterology 1995 Apr; 108 Suppl.: A221
Karol MD, Machinist JM, Cavanaugh JM. Lansoprazole pharmacokinetics in subjects with various degrees of kidney function. Clin Pharmacol Ther 1997; 61: 450–8
Karol MD, Machinist JM, Cavanaugh JM. Pharmacokinetics of lansoprazole in hemodialysis patients. J Clin Pharmacol 1995 Aug; 35: 815–20
Barclay ML, Begg EJ, Robson RA, et al. The pharmacokinetics of lansoprazole in normal volunteers and patients with reflux oesophagitis [abstract]. Clin Exp Pharmacol Physiol 1993 Suppl. 1: 3
Granneman GR, Karol MD, Locke CS. Pharmacokinetic interaction between lansoprazole and theophylline. Ther Drug Monk 1995 Oct; 17: 460–4
Kokufu T, Ihara N, Sugioka N, et al. Effects of lansoprazole on pharmacokinetics and metabolism of theophylline. Eur J Clin Pharmacol 1995; 48(5): 391–5
Cavanaugh J, Locke C, Shi H, et al. Assessment of the effect of sucralfate on the bioavailability of lansoprazole and omeprazole [abstract]. Am J Gastroenterol 1995 Sep; 90: 1577
Bergstrand R, Grind M, Nyberg G, et al. Decreased oral bioavailability of lansoprazole in healthy volunteers when given with a standardised breakfast. Clin Drug Invest 1995 Feb; 9: 67–71
Brummer R-JM, Geerling BJ, Stockbrügger RW. Acute and chronic effect of lansoprazole and omeprazole in relation to food intake [abstract no. T127]. Gut 1995; 37 Suppl. 2: A32
Mainz D, Borner K, Koeppe P, et al. Pharmacokinetics of lansoprazole, amoxicillin and clarithromycin when given single and concomitant [abstract no. 3066]. Presented at the 20th International Congress of Chemotherapy; 1997 Jun–Jul; Sydney
St Peter JV, Awni WM, Granneman GR, et al. The effects of lansoprazole on the disposition of antipyrine and indocyanine green in normal human subjects. Am J Ther 1995 Aug; 2: 561–8
Rizzo N, Padoin C, Palombo S, et al. Omeprazole and lansoprazole are not inducers of cytochrome P4501A2 under conventional therapeutic conditions. Eur J Clin Pharmacol 1996; 49(6): 491–5
Cavanaugh JH, Schneck DW, Mukherji D, et al. Lansoprazole does not alter the pharmacokinetics nor chronotropic effect of single dose propanolol [abstract]. 10th World Congress on Gastroenterology; 1994: Abstracts II: 654P
Karol MD, Mukherji D, Cavanaugh JH. Concomitant multi-dose lansoprazole does not alter single-dose phenytoin pharmacokinetics in subjects [abstract]. 10th World Congress on Gastroenterology 1994; Abstracts II: 654P
Karol MD, Mayersohn M, Eason CJ, et al. Evaluation of interaction potential between lansoprazole and clarithromycin (Biaxin Rm) in normal subjects [abstract]. Am J Gastroenterol 1995 Sep; 90: 1583
Cavanaugh JH, Karol MD. Lack of pharmacokinetic interaction after administration of lansoprazole or omeprazole with prednisone. J Clin Pharmacol 1996 Nov; 36: 1064–71
Girre C, Coutelle C, David P, et al. Lack of effect of lansoprazole on the pharmacokinetics of ethanol in male volunteers [abstract]. Gastroenterology 1994 Apr; 106 Suppl.: A504
Fuchs W, Sennewald R, Klotz U. Lansoprazole does not affect the bioavailability of oral contraceptives. Br J Clin Pharmacol 1994 Oct; 38: 376–80
TAP Holdings Inc. Data on file.
Chun AHC, Eason CJ, Shi HH, et al. Lansoprazole: an alternative method of administration of a capsule dosage formulation. Clin Ther 1995 May–Jun; 17: 441–7
Chun AHC, Shi HH, Achari R. Lansoprazole: administration of the contents of a capsule dosage formulation through a nasogastric tube. Clin Ther 1996 Sep-Oct; 18: 833–42
Tucker GT. The interaction of proton pump inhibitors with cytochromes P450. Aliment Pharmacol Ther 1994; 8 Suppl 1: 33–8
Avner DL, Dorsch ER, Jennings DE, et al. A comparison of three doses of lansoprazole (15, 30 and 60 mg) and placebo in the treatment of duodenal ulcer. Aliment Pharmacol Ther 1995 Oct; 9: 521–8
Avner DL, Movva R, Nelson KJ, et al. Comparison of once daily doses of lansoprazole (15, 30, and 60 mg) and placebo in patients with gastric ulcer. Am J Gastroenterol 1995 Aug; 90: 1289–94
Iwasaki A, Uno A, Okano N, et al. Helicobacter pylori eradication method for peptic ulcers — comparison of lansoprazole plus single- and double-antibacterial agent treatment regimens [abstract no. API 48]. 10th Asian-Pacific Congress on Gastroenterology 1996 Sep 19: 211
Petite J-P, Slama J-L, Licht H, et al. Lansoprazole and omeprazole in the treatment of duodenal ulcer: a multicentric double-blind comparative trial [in French]. Gastroenterol Clin Biol 1993; 17: 334–40
Ekström P, Carling L, Unge P, et al. Lansoprazole versus omeprazole in active duodenal ulcer: a double-blind, randomized, comparative study. Scand J Gastroenterol 1995 Mar; 30: 210–5
Dobrilla G, Italian Duodenal Ulcer Group for the study of Lansoprazole. Lansoprazole vs omeprazole in healing and relapse prevention of duodenal ulcer [abstract]. Gastroenterology 1996 Apr; 110 Suppl.: A96
Florent C, Audigier JC, Boyer J, et al. Efficacy and safety of lansoprazole in the treatment of gastric ulcer: a multicentre study. Eur J Gastroenterol Hepatol 1994; 6: 1135–9
Lanza F, Goff J, Scowcroft C, et al. Double-blind comparison of lansoprazole, ranitidine, and placebo in the treatment of acute duodenal ulcer. Am J Gastroenterol 1994 Aug; 89: 1191–200
Bardhan KD, Crowe J, Thompson RPH, et al. Lansoprazole v ranitidine maintenance treatment for prevention of duodenal ulcer relapse [abstract]. Gastroenterology 1996; 110(4) Suppl.: A56
Bardhan KD, Ahlberg J, Hislop WS, et al. Rapid healing of gastric ulcers with lansoprazole. Aliment Pharmacol Ther 1994; 8: 215–20
Michel P, Lemaire M, Colin R, et al. Short report: treatment of gastric ulcer with lansoprazole or ranitidine: a multicentre clinical trial. Aliment Pharmacol Ther 1994; 8: 119–22
Poynard T, Lemaire M, Agostini H. Meta-analysis of randomized clinical trials comparing lansoprazole with ranitidine or famotidine in the treatment of acute duodenal ulcer. Eur J Gastroenterol Hepatol 1995 Jul; 7: 661–5
Russo A, Dattilo M. Bedtime administration of lansoprazole does not alter its superior efficacy versus ranitidine in the acute and long-term treatment of duodenal ulcer. Results from a multicentre, randomised, double blind clinical trial. Wyeth S.p.A., Aprilia, Italy. 1997. (Data on file)
Robinson M, Campbell DR, Sontag S. Treatment of erosive reflux esophagitis resistant to H2-receptor antagonist therapy: lansoprazole, a new proton pump inhibitor. Dig Dis Sci 1995 Mar; 40: 590–7
van Rensburg CJ, Louw JA, Girdwood AH, et al. A trial of lansoprazole in refractory gastric ulcer. Aliment Pharmacol Ther 1996; 10: 381–6
Harlet L, Penson J, Boesaart E, et al. Lansoprazole in maintenance therapy for resistant duodenal ulcers [abstract no. 1392]. Gut 1995; 37 Suppl. 2: A163
Lanza F, Goff J, Silvers D, et al. Lansoprazole for one year prevents recurrence of duodenal ulcer [abstract]. 10th World Congress on Gastroenterology 1994; Abstracts II: 27P
Kovacs TOG, Campbell DR, Richter JE, et al. Lansoprazole (LAN) is safe and effective for one year in preventing recurrence of duodenal ulcer (DU) disease [abstract]. Am J Gastroenterol 1995; 90(9): 1584
Campbell DR, Kovacs TOG, Sontag SJ. Lansoprazole (LAN) is safe and effective for one year in preventing recurrence of gastric ulcer (GU) disease [abstract no. 95]. Am J Gastroenterol 1995; 90(9): 1577
Lind T, Veldhuyzen van Zanten SJO, Unge P, et al. The Mach 1 study: optimal one-week treatment for H. pylori defined? [abstract]. Gut 1995; 37 Suppl. 1: A4
Digestive Health InitiativeSM of the American Digestive Health FoundationSM. The report of the Digestive Health InitiativeSM International Update Conference on Helicobacter pylori. Presented at Digestive Disease Week; 1997 May 14: Washington, DC
Chey WD, Fey D, Scheiman JM, et al. The role of acid suppression in the effects of lansoprazole and ranitidine on the 14C-urea breath test [abstract]. Gastroenterology 1996; 110(4) Suppl.: A80
Ogoshi K, Kato T, Sakagawa T. Peptic ulcer therapy with lansoprazole and Helicobacter pylori eradication. J Clin Gastroenterol 1995; 20 Suppl. 2: S97–9
Eberl T, Barnert J, Wagner T, et al. Combination therapy with lansoprazole and roxithromycin in Helicobacter pylori eradication [abstract]. Gastroenterology 1996 Apr; 110 Suppl.: A100
Fernandez JA, Suarez A, Aponte L. Dual therapy with azithromycin and lansoprazole for the treatment of duodenal ulcers associated with Helicobacter pylori [abstract no. 295]. Am J Gastroenterol 1996 Sep; 91: 1959
Medina N, Reymunde A. Helicobacter pylori eradication with clarithromycin and lanzoprazole in short term period (14 days) [abstract no. 148]. Am J Gastroenterol 1996; 91(9): 1920
Sierra F, Gutierrez O, Molano B, et al. Eradication of Helicobacter pylori with lansoprazole and clarithromycin [abstract]. Gastroenterology 1995; 108(4) Suppl.: A219
Takimoto T, Kimura K, Taniguchi Y, et al. Dual therapy with lansoprazole and clarithromycin for eradication of Helicobacter pylori. Eur J Gastroenterol Hepatol 1995 Aug; 7 Suppl. l: S63–6
Chahin NJ, Rigo GP, Perini M, et al. Short triple therapy for Helicobacter pylori (HP) eradication in duodenal ulcer (DU) patients: could it be even shorter? [abstract]. Gastroenterology 1996; 110(4) Suppl.: A78
Schütze K, Hentschel E. Duodenal ulcer healing after 7-day treatment: a pilot study with lansoprazole, amoxicillin and clarithromycin. Z Gastroenterol 1995; 33: 651–3
Sierra F, Otero W, Gutierrez O, et al. One week full-dose triple therapy: new standard for Helicobacter pylori treatment in Latin America [abstract]. Gastroenterology 1996; 110(4) Suppl.: A258
Egawa S, Suzaki Y, Murakawa Y, et al. A comparative study of the early and late combined therapy with lansoprazole and amoxicillin in patients with H. pylori-positive peptic ulcer [in Japanese]. Shinyaku to Rinsho 1996 Feb; 45: 390–6
Hatlebakk JG, Nesje LB, Hausken T, et al. Lansoprazole capsules and amoxicillin oral suspension in the treatment of peptic ulcer disease. Scand J Gastroenterol 1995 Nov; 30: 1053–7
Kato M, Asaka M, Kudo M, et al. Effects of lansoprazole plus amoxycillin on the cure of Helicobacter pylori infection in Japanese peptic ulcer patients. Aliment Pharmacol Ther 1996; 10: 821–7
Katoh M, Asaka M, Kudoh M, et al. Clinical efficacy of lansoprazole in eradication of Helicobacter pylori. J Clin Gastroenterol 1995; 20 Suppl. 2: SI12–4
Kohli Y, Kato T, Azuma T, et al. Lansoprazole treatment of Helicobacter pylori-positive peptic ulcers. J Clin Gastroenterol 1995; 20 Suppl 1: S48–51
Louw JA, van Rensburg C, Moola S, et al. One versus two weeks single dose lansoprazole based combination therapy: efficacy of H. pylori eradication and duodenal ulcer healing [poster no. 4]. S Afr Med J 1996; 86(9): 1122
Parente F, Maconi G, Bargiggia S, et al. Comparison of two lansoprazole-antibiotic combinations (amoxycillin or classical triple therapy) for treatment of H. pylori infection in duodenal ulcer patients. Aliment Pharmacol Ther 1996 Apr; 10: 211–3
Shimoyama T, Munakata A, Mizuki I, et al. Eradication of Helicobacter pylori with lansoprazole and clarithromycin in gastric ulcer patients. J Clin Gastroenterol 1995; 20 Suppl. 2: S125–7
Sugiyama T, Hisano K, Ochiai T, et al. Lansoprazole versus lansoprazole plus amoxicillin treatment for eradication of Helicobacter pylori in patients with gastric ulcer. J Clin Gastroenterol 1995; 20 Suppl. 2: S104–6
Phull PS, Price AB, Halliday D, et al. Lansoprazole plus clarithromycin: evaluation of a new dual therapy for Helicobacter pylori eradication. Br J Clin Pract 1996; 50(7): 360–2
Tsai T-C, Lin H-H, Huang L-C, et al. Lansoprazole-containing triple therapy on Helicobacter pylori associated gastric ulcers. Tzu Chi Med J 1996; 8(3): 191–6
Saggioro A, GISU (Interdisciplinary Group for Ulcer Study). A one-week triple therapy vs a two-week dual therapy for eradication and healing of H. pylori (Hp) positive duodenal ulcers (DU): results from a randomized, double blind clinical trial [abstract no. 2208]. Digestive Disease Week; 1997 May 10–16: Washington, DC
Rehmann I, Kadus W. Low-dose lansoprazole short-term triple regimen for eradication of H. pylori [abstract]. Gastroenterology 1997 Apr; 112(4): A38
Jaup BH, Norrby A. Comparison of two low-dose one-week triple therapy regimens with and without metronidazole for cure of H. pylori infection. Aliment Pharmacol Ther 1996; 10: 275–7
Lamouliatte H, Florent C, Vicari F, et al. Effect of lansoprazole and amoxicillin plus metronidazole on the eradication of Helicobacter pylori: a randomized, double blind, multicenter study [abstract]. Gastroenterology 1996; 110(4) Suppl.: A170
Nakata H, Itoh H, Nishioka S. Efficacy of lansoprazole and amoxicillin in eradicating Helicobacter pylori: evaluation using 13C-UBT and monoclonal H. pylori antibody testing. J Clin Gastroenterol 1995; 20 Suppl. 2: S118–20
Shirai T, Takagi A, Kurumada T, et al. Eradication of Helicobacter pylori with lansoprazole, amoxicillin, and plaunotol in duodenal ulcer patients. J Clin Gastroenterol 1995; 20 Suppl. 2: S128–31
Yonei Y, Tsukada N, Inagaki Y, et al. Effect of lansoprazole/ sucralfate and clarithromycin on Helicobacter pylori infection and recurrence in the patients with peptic ulcer [in Japanese]. Shinyaku to Rinsho 1996; 45(7): 1385–404
Harris AW, Gummett PA, Logan RPH, et al. Eradication of Helicobacter pylori with lansoprazole and clarithromycin. Aliment Pharmacol Ther 1995 Apr; 9: 201–4
Matsukura N, Onda M, Shirakawa T, et al. High eradication rate of Helicobacter pylori with lansoprazole + amoxicillin + ecabet sodium: new triple therapy with novel mucosal protective agent for the treatment of peptic ulcer [abstract]. Gastroenterology 1996; 110(4) Suppl.: A190
Murakami M, Saita H, Takahashi Y, et al. Therapeutic effects of lansoprazole on peptic ulcers in elderly patients. J Clin Gastroenterol 1995; 20 Suppl. 2: S79–82
Saita H, Murakami M, Dekigai H, et al. Efficacy of two weeks combination therapy with lansoprazole and amoxillin to eradicate H. pylori [abstract]. 10th World Congress on Gastroenterology 1994; Abstracts II: 74P
Schwartz H, Krause R, Sahba B. Triple vs. dual therapy with lansoprazole, clarithromycin and/or amoxicillin in the eradication of Helicobacter pylori [abstract no. 641], Am J Gastroenterol 1996; 91(9): 2047
Schwartz H, Krause R, Sahba B. Triple vs. dual therapy with lansoprazole (L), clarithromycin (C), and/or amoxicillin (A) in the eradication of Helicobacter pylori [abstract no. 641]. Am J Gastroenterol 1997 Sep; 91(9): 2047
Ohashi T, Sakata J, Haraguchi Y, et al. Effect of lansoprazole on peptic ulcers. J Clin Gastroenterol 1995; 20 Suppl. 2: S83–5
Gisbert JP, Boixeda D, Alvarez Baleriola I, et al. Eradicating therapy of Helicobacter pylori with lansoprazole and amoxicillin in duodenal ulcer patients [in Spanish]. Rev Clin Esp 1996; 196: 588–93
Chey WD, Fisher L, Elta GH, et al. One week, bid therapy for H. pylori: a randomized comparison of 2 strategies [abstract]. Gastroenterology 1997 Apr; 112(4): A87
Bolin TD, Korman MG, Engelman JL, et al. Lansoprazole and bismuth triple therapy in the eradication of Helicobacter pylori [abstract]. Gastroenterology 1997 Apr; 112(4): A76
Saita H, Murakami M, Dekigai H, et al. Two weeks combination therapy with lansoprazole and amoxicillin for eradication of H. pylori [abstract]. Gastroenterology 1994; 106(4) Suppl.: A171
Vakil N, Peutz T. Helicobacter pylori eradication treatment with dual therapy (lansoprazole and clarithromycin) in an office-based study [abstract]. 3rd International Congress on the Macrolides Azalides and Streptogramins 1996 Jan: 148–9
Barbezat G, Schlup M, Lubcke R, et al. Lansoprazole, tetracycline and metronidazole — an effective Helicobacter pylori eradication regime [abstract]. J Gastroenterol Hepatol 1995; 10 Suppl. 2: A47
Burette A, Lamy V, Ramdani B, et al. Lansoprazole (30 mg OD vs BID) with amoxicillin and clarithromycin to cure Helicobacter pylori infection [abstract]. Gut 1996; 39 Suppl. 2: 33
Cayla R, Zerbib F, de Mascarel A, et al. High dose of lansoprazole in combination with amoxicillin and clarithromycin twice daily for H. pylori eradication: results of a pilot study [abstract]. Am J Gastroenterol 1994 Aug; 89: 1367
Harris AW, Pryce DI, Gabe SM, et al. Lansoprazole, clarithromycin and metronidazole for seven days in Helicobacter pylori infection. Aliment Pharmacol Ther 1996 Dec; 10: 1005–8
Schwartz H, Krause R, Siepman N, et al. Seven-day triple therapy with lansoprazole, clarithromycin, and metronidazole for the cure of Helicobacter pylori infection: a short report. Helicobacter 1996; 1(4): 251–5
Riff DS, Kidd S, Rose P, et al. Triple therapy with lansoprazole, clarithromycin, and amoxicillin for the cure of Helicobacter pylori infection: a short report. Helicobacter 1996; 1(4): 238–42
Isomoto H, Matsunaga K, Ofukuji M, et al. Effect of lansoprazole and clarithromycin plus metronidazole on eradication of Helicobacter pylori [abstract no. API24]. 10th Asian-Pacific Congress on Gastroenterology 1996 Sep 19: 199
Kodama M, Tsukada H, Fukuda K, et al. Comparative study of successful and unsuccessful cases of Helicobacter pylori eradication [abstract no. AP141]. 10th Asian-Pacific Congress on Gastroenterology 1996 Sep 19: 208
Kumar N, Shanker A. Study of new short term triple therapy in Helicobacter pylori associated antral gastritis [abstract no. APV24]. 10th Asian-Pacific Congress on Gastroenterology 1996 Sep 19: 121
Lamouliatte H, Courrier A, Mion F, et al. Triple therapy with roxithromycin-amoxicillin and lansoprazole for Helicobacter pylori eradication: results of an open multicentre study [abstract]. Gastroenterology 1996 Apr; 110 Suppl. A: A171
Nakata H, Imoto K, Kasamatsu T, et al. Effect of lansoprazole and clarithromycin plus polaprezinc on the eradication of Helicobacter pylori [abstract]. 10th Asian-Pacific Congress on Gastroenterology 1996 Sep 19: 202
Ozmen MM, Johnson CD. Is short-term triple therapy with lansoprazole, clarithromycin, and metronidazole a definitive answer for Helicobacter pylori eradication? [letter]. Am J Gastroenterol 1995; 90(9): 1542–3
Pryce DI, Harris AW, Gabe SM, et al. One week of lansoprazole, clarithromycin and metronidazole eradicates Helicobacter pylori [abstract]. Gastroenterology 1996; 110(4) Suppl.: A235
Salomaa A, Kosunen TU, Rautelin H, et al. Screening and eradication of H. pylori infection in adult population of Vammala, Finland [abstract no. 3A:08]. Gut 1996; 39 Suppl. 2: A22–23
Takahara N, Nakata H, Tanaka T, et al. Effect of lansoprazole and clarithromycin plus amoxicillin on the eradication of Helicobacter pylori [abstract no. API 29]. 10th Asian-Pacific Congress on Gastroenterology 1996 Sep 19: 202
Tursi A, Cammarota G, Papa A, et al. Short-term low-dose triple therapy with lansoprazole plus amoxicillin and clarithromycin for Helicobacter pylori eradication [letter]. Am J Gastroenterol 1996; 91(8): 1668–9
De Boer WA, Van Etten RJXM, Schade RWB, et al. One-day intensified lansoprazole-quadruple therapy for cure of Helicobacter pylori infection. Aliment Pharmacol Ther 1997; 11: 109–12
de Boer WA, van Etten RJXM, Schade RWB, et al. 4-Day lansoprazole quadruple therapy: a highly effective cure for Helicobacter pylori infection. Am J Gastroenterol 1996 Sep; 91: 1778–82
Nakata H, Imoto K, Kasamatsu T, et al. Eradication of Helicobacter pylori with a combination of lansoprazole, amoxicillin, clarithromycin and polaprezinc [abstract no. API31]. 10th Asian-Pacific Congress on Gastroenterology 1996 Sep 19: 203
Bateson MC, Diffey BL. New treatments for duodenal ulcer [abstract]. Gastroenterology 1996; 110(4) Suppl.: A59
Cammarota G, Tursi A, Papa A, et al. Helicobacter pylori eradication using one-week low-dose lansoprazole plus amoxycillin and either clarithromycin or azithromycin. Aliment Pharmacol Ther 1996 Dec; 10: 997–1000
Cayla R, de Mascarel A, Zerbib F, et al. High dose of lansoprazole plus amoxicillin versus high dose of lansoprazole plus amoxicillin and clarithromycin for Helicobacter pylori (H. pylori) infection [abstract]. Gastroenterology 1995 Apr; 108 Suppl.: A68
Chey WD, Fisher L, Elta GH, et al. One week, bid therapy for H. pylori: a randomized comparison of 2 strategies [abstract no. 98]. Am J Gastroenterol 1996 Sep; 91: 1906
De Korwin JD, Joubert M, Bazin N, et al. Lansoprazole versus lansoprazole plus antibiotics in the treatment of Helicobacter pylori gastric infection: a randomised multicenter trial [in French]. Ann Med Nancy Est 1994; 33(2): 123–5
Hazell SL, Daskalopoulous G, Mitchell HM, et al. Lansoprazole and amoxycillin: observations on the treatment of Helicobacter pylori infection. J Gastroenterol Hepatol 1997; 12: 93–9
Caselli M, Trevisani L, Tursi A, et al. Short-term low-dose triple therapy with azithromycin, metronidazole and lansoprazole appears highly effective for the eradication of Helicobacter pylori. Eur J Gastroenterol Hepatol 1997; 9: 45–8
Catalano F, Privitera U, Branciforte G, et al. Omeprazole vs two different doses of lansoprazole in triple therapy on H. pylori positive duodenal ulcer [abstract no. 4A:08]. Gut 1996; 39 Suppl. 2: A32
Daskalopoulos G, Lian JX, Ho YY. Randomized controlled study comparing triple therapy containing a proton pump inhibitor to standard triple therapy for the eradication of H. pylori [abstract]. Gastroenterology 1995 Apr; 108 Suppl.: A79
Deltenre M, Jonas C, Burette A, et al. Bazzoli-like schemes are not optimal treatment for HP eradication in Brussels, Belgium [abstract]. Gastroenterology 1996 Apr; 110 Suppl.: A93
Ho AS, Lee SC, Hsu CT. What’s the clinically favored triple therapy? [abstract no. 4A:09]. Gut 1996; 39 Suppl. 2: A32–3
Jonas C, DeReuck M, Otero J, et al. Low-dose, one-week triple therapies for HP: a randomized study [abstract]. Gastroenterology 1996 Apr; 110 Suppl.: A145
Kihira K, Kimura K, Satoh K, et al. Effect of 1-week triple therapy for Helicobacter pylori infection with lansoprazole or ranitidine and clarithromycin and metronidazole [abstract]. Gastroenterology 1996 Apr; 110 Suppl. A: A154
Liu WZ, Xiao SD, Xu WW, et al. Efficacy of one-week triple therapy with clarithromycin, furazolidone plus lansoprazole or colloidal bismuth subcitrate for eradication of Helicobacter pylori [abstract no. 4A:10]. Gut 1996; 39 Suppl. 2: A33
Misiewicz JJ, Harris AW, Bardhan KD, et al. One week low-dose triple therapy for eradication of H. pylori: a large multi-centre, randomised trial [abstract no. W4]. Gut 1996; 38 Suppl. 1: A1
Müllhaupt B, Flückiger T, Fröhli P, et al. One week low dose triple therapies with lansoprazole or omeprazole are equally effective for H. pylori eradication and ulcer healing [abstract]. Gastroenterology 1996; 110(4) Suppl.: A202
Sito E, Konturek PC, Bielanski W, et al. One week treatment with omeprazole, clarithromycin and tinidazole or lansoprazole, amoxicillin and metronidazole for cure of Helicobacter pylori infection in duodenal ulcer patients. J Physiol Pharmacol 1996 Mar; 47(1): 221–8
Spinzi GC, Colombo E, Bortoli A, et al. Lansoprazole vs omeprazole: one week triple therapy in peptic ulcer (preliminary results) [abstract]. Gastroenterology 1997 Apr; 112(4): A298
Yang JC, Yang CK, Wang JT, et al. Use of lansoprazole with and without antibiotics in the healing of duodenal ulcer and eradication of Helicobacter pylori [abstract no. 1988]. Gut 1995; 37 Suppl. 2: A232–3
Bardhan KD, Hawkey CJ, Long RG, et al. Lansoprazole versus ranitidine for the treatment of reflux oesophagitis. Aliment Pharmacol Ther 1995 Apr; 9: 145–51
Benhaim MC, Evereux M, Salducci J, et al. Lansoprazole and ranitidine in treatment of reflux oesophagitis: double blind comparative trial [abstract]. Gastroenterology 1990; 98: A20
Jansen JBMJ, Hazenberg BP, Tan TG, et al. Lansoprazole (30 mg) is more effective than high-dose ranitidine (2 × 300 mg) in moderate to severe reflux esophagitis — a Dutch multi-center trial [abstract]. Gastroenterology 1996; 110 Suppl.: 143
Plein K, Stolte M, Fuchs W, et al. Lansoprazole vs. ranitidine — efficacy in healing acute reflux esophagitis and influence on hyperregenerative esophagopathy [abstract]. Gut 1995; 37 Suppl. 2: A38
Robinson M, Sahba B, Avner D, et al. A comparison of lansoprazole and ranitidine in the treatment of erosive oesophagitis. Aliment Pharmacol Ther 1995 Feb; 9: 25–31
Castell DO, Richter JE, Robinson M, et al. Efficacy and safety of lansoprazole in the treatment of erosive reflux esophagitis. Am J Gastroenterol 1996 Sep; 91: 1749–57
Corallo J, Vicari F, Forestier S, et al. Lansoprazole in acute treatment of reflux esophagitis [abstract]. Gastroenterology 1993; 28: A58
Hatlebakk JG, Berstad A, Carling L, et al. Lansoprazole versus omeprazole in short-term treatment of reflux oesophagitis: results of a Scandinavian multicentre trial. Scand J Gastroenterol 1993; 28: 224–8
Mee AS, Rowley JL, Lansoprazole Clinical Research Group. Rapid symptom relief in reflux oesophagitis: a comparison of lansoprazole and omeprazole. Aliment Pharmacol Ther 1996; 10: 757–63
Mulder CJ, Dekker W, Gerretsen M. Lansoprazole 30mg versus omeprazole 40mg in the treatment of reflux oesophagitis grade II, III and IVa: a Dutch multicentre trial. Eur J Gastroenterol Hepatol 1996 Nov; 8: 1101–6
Sekiguchi T, Horikoshi T, Nishioka T, et al. Clinical effect of proton pump inhibitors on reflux esophagitis [in Japanese]. Nip Rinsho 1992 Jan; 50: 131–7
Rampai P, Courrier A, Lemerez M, et al. Efficacy and safety of lansoprazole 30 mg versus omeprazole for 21 days treatment of acute esophagitis [abstract]. Gastroenterology 1995; 108 Suppl.: A200
Umeda N, Miki K, Hoshino E. Lansoprazole versus famotidine in symptomatic reflux esophagitis: a randomized, multicenter study. J Clin Gastroenterol 1995; 20 Suppl. 1: S17–23
Sontag SJ, Kogut DG, Fleischmann R, et al. Lansoprazole heals erosive reflux esophagitis resistant to histamine H2-receptor antagonist therapy. Am J Gastroenterol 1997; 92(3): 429–37
Dorsch E, Jones J, Rose P, et al. Lansoprazole provides superior symptom relief for patients with non-erosive reflux esophagitis [abstract]. Gastroenterology 1996 Apr; 110 Suppl.: A98
Hatlebakk JG, Berstad A. Lansoprazole 15 and 30 mg daily in maintaining healing and symptom relief in patients with reflux oesophagitis. Aliment Pharmacol Ther 1997; 11: 365–72
Poynard T, Staub JL, Lemerez M, et al. Efficacy and safety of lansoprazole 15 mg OAD or 30mg OAD as one year maintenance treatment for erosive reflux esophagitis: a randomized trial [abstract]. Gastroenterology 1995 Apr; 108 Suppl.: A195
Robinson M, Lanza F, Avner D, et al. Effective maintenance treatment of reflux esophagitis with low-dose lansoprazole: a randomized, double-blind, placebo-controlled trial. Ann Intern Med 1996 May 15; 124: 859–67
Sontag SJ, Kogut DG, Fleischmann R, et al. Lansoprazole prevents recurrence of erosive reflux esophagitis previously resistant to H2-RA therapy. Am J Gastroenterol 1996 Sep; 91: 1758–65
Baldi F, Bardhan KD, Borman BC, et al. Lansoprazole maintains healing in patients with reflux esophagitis [abstract]. Gastroenterology 1996 Apr; 110 Suppl.: A55
Carling L, Axelsson C, Forsell H, et al. Lansoprazole versus omeprazole in long term maintenance treatment of reflux oesophagitis: a Scandinavian multicentre trial [abstract no. 1036] [abstract]. Gut 1996; 39 Suppl. 3: 182
Gough AL, Long RG, Cooper BT, et al. Lansoprazole versus ranitidine in the maintenance treatment of reflux oesophagitis. Aliment Pharmacol Ther 1996 Aug; 10: 529–39
Feldman M, Harford WV, Fisher RS, et al. Treatment of reflux esophagitis resistant to H2-receptor antagonists with lansoprazole, a new H+/K+-ATPase inhibitor: a controlled, double-blind study. Am J Gastroenterol 1993; 88: 1212–7
Sontag SJ, Schnell TG, Chejfec G, et al. Lansoprazole heals erosive reflux oesophagitis in patients with Barrett’s oesophagus. Aliment Pharmacol Ther 1997; 11: 147–56
Sampliner RE. Effect of up to 3 years of high-dose lansoprazole on Barrett’s esophagus. Am J Gastroenterol 1994 Oct; 89: 1844–8
Savarino V, Mela GS, Zentilin P, et al. Once or twice daily doses of proton pump inhibitor in treating Barrett’s esophagus? [letter]. Am J Gastroenterol 1995 May; 90: 845–6
Swarbrick ET, Gough AL, Foster CS, et al. Prevention of recurrence of oesophageal stricture, a comparison of lansoprazole and high-dose ranitidine. Eur J Gastroenterol Hepatol 1996 May; 8: 431–8
Mignon M, Hochlaf S, Forestier S, et al. Acid inhibitory effect of graded doses of lansoprazole in patients with Zollinger-Ellison syndrome [in French]. Gastroenterol Clin Biol 1994 Jan; 18: 13–6
Pospari D, Forestier S, Vissuzaine CH, et al. Long-term lansoprazole (LAN) treatment in patients with Zollinger-Ellison syndrome (ZES) [abstract]. 10th World Congress on Gastroenterology 1994; Abstracts II: 25P
Hirschowitz BI, Mohnen J, Shaw S. Long-term treatment with lansoprazole for patients with Zollinger-Ellison syndrome. Aliment Pharmacol Ther 1996 Aug; 10: 507–22
Otani Y, Kitajima M, Sugiyama M, et al. Inhibitory effects of intravenous lansoprazole on gastric acid hypersecretion in patients with postoperative stress. J Clin Gastroenterol 1995; 20 Suppl 2: S22–6
Hett DA, Scott RC, Risdall JE. Lansoprazole in the prophylaxis of acid aspiration during elective surgery. Br J Anaesth 1995 May; 74: 614–5
Mikawa K, Nishina K, Maekawa N, et al. Lansoprazole reduces preoperative gastric fluid acidity and volume in children. Can J Anaesth 1995 Jun; 42: 467–72
Nishina K, Mikawa K, Maekawa N, et al. A comparison of lansoprazole, omeprazole, and ranitidine for reducing preoperative gastric secretion in adult patients undergoing elective surgery. Anesth Analg 1996 Apr; 82: 832–6
Nishioka S, Ogoshi K, Ogawa N. Clinical study of lansoprazole (AG-1749) injection on upper gastrointestinal bleeding: hemostatic effect of lansoprazole injection and preventive effect of lansoprazole capsules on re-bleeding [in Japanese]. Rinsho Iyaku 1996; 12(13): 2927–52
Cole AT, Maclntyre AS, Hawkey GM, et al. Lansoprazole and tranexamic acid alone and in combination for upper gastrointestinal bleeding [abstract]. Gastroenterology 1995 Apr; 108 Suppl.: A74
Michel P, Duhamel C, Bazin B, et al. Prevention of early rebleeding from gastric and duodenal peptic ulcer with lansoprazole or ranitidine. Randomized multicentre trial [in French]. Gastroenterol Clin Biol 1994 Dec; 18: 1102–5
Colin-Jones DG. Safety of lansoprazole. Br J Clin Pract 1994 May–Jun Suppl. 75: 58-66
TAP Pharmaceuticals Inc. PrevacidR (lansoprazole) delayed release capsules. In: Physicians’ Desk Reference. Montvale: Medical Economics, 1996: 2562–4
Penson J, Puttemans M, Fourie E. Safety of lansoprazole in maintenance therapy for reflux oesophagitis [abstract]. Gut 1995; 37 Suppl. 2: A163
Freston JW. Long-term acid control and proton pump inhibitors: interactions and safety issues in perspective. Am J Gastroenterol 1997; 92(4): 51S–7S
Jones RH, Bosanquet N, Johnson NJ, et al. Cost-effective management strategies for acid-peptic disorders. Br J Med Econ 1994; 7: 99–114
Sadowski DC, Chiba N, Manzi P. An economic comparison of Helicobacter pylori treatment regimens in Canada [abstract]. Gastroenterology 1997 Apr; 112(4): A274
Heudebert GR, Wilcox CM, Marks R. Impact of lansoprazole in the management of gastroesophageal reflux disease (GERD)): a cost-utility analysis [abstract]. Am J Gastroenterol 1996 Sep; 91: 1889
Zagari M, Villa KF, Freston JW. Proton pump inhibitors versus H2-receptor antagonists for the treatment of erosive gastroesophageal reflux disease: a cost-comparative study. Am J Managed Care 1995 Nov-Dec; 1: 247–55
Harris RA, Kupperman M, Richter JE. The cost effectiveness of proton pump inhibitors vs histamine-2 receptor antagonists for the prevention of recurrences of erosive reflux esophagitis [abstract]. Presented at the 18th Annual Scientific Meeting of the Society for Medical Decision Making (SMDM). 13–16 October 1996, Toronto, Canada.
Harris RA, Kuppermann M, Richter JE. Prevention of recurrences of erosive reflux esophagitis: a cost-effectiveness analysis of maintenance proton pump inhibition. Am J Med 1997 Jan; 102: 78–88
Bardhan KD. Management issues and economic implications in erosive oesophagitis — a case-illustrated discussion. Eur J Clin Res 1997; 9: 85–9
Hopkins RJ, Girardi LS, Turney EA. Relationship between Helicobacter pylori eradication and reduced duodenal and gastric ulcer recurrence: a review. Gastroenterology 1996 Apr; 110: 1244–52
Leon-Barua R, Recavarren-Arce S, Gilman RH, et al. Can eradication of Helicobacter pylori prevent gastric cancer? Drugs 1993 Sep; 46: 341–6
Hackelsberger A, Malfertheiner P. A risk-benefit assessment of drugs used in the eradication of Helicobacter pylori infection. Drug Saf 1996; 15(1): 30–52
Walsh JH, Peterson WL. The treatment of Helicobacter pylori infection in the management of peptic ulcer disease. N Engl J Med 1995 Oct 12; 333: 984–91
Anonymous. Release of new European H. pylori eradication guidelines. Inpharma 1996 Nov 9; 1062: 2
Soll AH, Practice Parameters Committee of the American College of Gastroenterology. Medical treatment of peptic ulcer disease: practice guidelines. JAMA 1996; 275(8): 622–9
Caspary WF, Arnold R, Bayerdörffer E, et al. Diagnosis and therapy of Helicobacter pylori infection [in German]. Z Gastroenterol 1996 Jun; 34: 392–401
Agence Nationale pour le Développement de l’Évaluation Médicale. Anti-ulcer agents: recommendations and medical references [in French]. Gastroenterol Clin Biol 1996; 20: 991–1008
de Boer WA. How to achieve a near 100% cure rate for H. pylori infection in peptic ulcer patients: a personal viewpoint. J Clin Gastroenterol 1996; 22(3): 313–6
Graham DY. A reliable cure for Helicobacter pylori infection? Gut 1995; 37: 154–6
Brady WM, Ogorek CP. Gastro-esophageal reflux disease: the long and the short of therapeutic options. Postgrad Med 1996 Nov; 100(85-86 & 89): 76–80
Hunt RH. The relationship between the control of pH and healing and symptom relief in gastro-oesophageal reflux disease. Aliment Pharmacol Ther 1995; 9 Suppl. 1: 3–7
Best regimens for H pylori eradication. Scrip 1997 Jun 17(2241): 21
Jensen RT, Fraker DL. Zollinger-Ellison syndrome: advances in treatment of gastric hypersecretion and the gastrinoma. JAMA 1994 May 11; 271: 1429–35
Author information
Authors and Affiliations
Corresponding author
Additional information
Various sections of the manuscript reviewed by: F.-Y. Chang, Division of Gastroenterology, Veterans General Hospital-Taipei, Taipei, Taiwan, Republic of China; C. Florent, Gastro-enterologie, Hôpital Saint-Antoine, Paris, France; Y. Fukuda, Department of Internal Medicine 4, Hyogo College of Medicine, Hyogo, Japan; A.W. Harris, Parkside Helicobacter Study Group, Central Middlesex Hospital, London, England; J.G. Hatlebakk, Division of Gastroenterology, Medical Department A, Haukeland University Hospital, Bergen, Norway; R.H. Hunt, Division of Gastroenterology, Department of Medicine, McMaster University, Chedoke-McMaster Hospitals, Hamilton, Ontario, Canada; R.T. Jensen, Digestive Disease Branch, National Institute of Diabetes and Digestive and Kidney Diseases, National Institutes of Health, Public Health Service, Department of Health and Human Services, Bethesda, Maryland, USA; R. León-Barúa; Universidad Peruana Cayetano Heredia, Instituto de Medicina Tropical ‘Alexander von Humboldt’, Lima, Peru; C. O’Morain, Department of Gastroenterology, The Adelaide & Meath Hospital, Dublin, Ireland; P. Phull, GI Unit, Aberdeen Royal Infirmary, Aberdeen, Scotland; R. E. Sampliner, Section of Gastroenterology, Department of Medicine, College of Medicine, The University of Arizona Health Sciences Center, Tucson, Arizona, USA; A.H. Soll, CURE Digestive Disease Research Center, West Los Angeles Veterans Affairs Medical Center, Los Angeles, California, USA.
Rights and permissions
About this article
Cite this article
Langtry, H.D., Wilde, M.I. Lansoprazole. Drugs 54, 473–500 (1997). https://doi.org/10.2165/00003495-199754030-00010
Published:
Issue Date:
DOI: https://doi.org/10.2165/00003495-199754030-00010