Abstract
Synopsis
Epirubicin is the 4′ epimer of the anthracycline antibiotic doxorubicin, and has been used alone or in combination with other cytotoxic agents in the treatment of a variety of malignancies. Comparative and noncomparative clinical trials have demonstrated that regimens containing conventional doses of epirubicin achieved equivalent objective response rates and overall median survival as similar doxorubicin-containing regimens in the treatment of advanced and early breast cancer, non-small cell lung cancer (NSCLC), small cell lung cancer (SCLC), non-Hodgkin’s lymphoma, ovarian cancer, gastric cancer and nonresectable primary hepatocellular carcinoma. Recently, dose-intensive regimens of epirubicin have achieved high response rates in a number of malignancies including early and advanced breast cancer and lung cancer.
The major acute dose-limiting toxicity of anthracyclines is myelosuppression. In vitro and clinical studies have shown that, at equimolar doses, epirubicin is less myelotoxic than doxorubicin. The lower haematological toxicity of epirubicin, as well as the recent introduction of supportive measures such as colony-stimulating factors, has allowed dose-intensification of epirubicin-containing regimens, which is particularly significant because of the definite dose-response relationship of anthracyclines. Cardiotoxicity, which is manifested clinically as irreversible congestive heart failure and/or cardiomyopathy, is the most important chronic cumulative dose-limiting toxicity of anthracyclines. Epirubicin has a lower propensity to produce cardiotoxic effects than doxorubicin, and its recommended maximum cumulative dose is almost double that of doxorubicin, thus allowing for more treatment cycles and/or higher doses of epirubicin.
In summary, dose-intensive epirubicin-containing regimens, which are feasible due to its lower myelosuppression and cardiotoxicity, have produced high response rates in early breast cancer, a potentially curable malignancy, as well as advanced breast, and lung cancers. Furthermore, there is evidence to suggest that improved response rates can improve quality of life in some clinical settings, but whether this leads to prolonged survival has not yet been determined. Recently implemented supportive measures such as colony-stimulating factors, prophylactic antimicrobials and peripheral blood stem cell support may help achieve other potential advantages of dose-intensive epirubicin-containing regimens such as reductions in morbidity and length of hospital admissions.
Pharmacodynamic Properties
Epirubicin is the epimer of doxorubicin, with inversion of the 4′-hydroxyl group on the sugar moiety. Epirubicin is a cell cycle phase non-specific anthracycline, with maximal cytotoxic effects in the S and G2 phases. In vitro studies showed that epirubicin possesses cytotoxicity at least equivalent to that of doxorubicin against a variety of animal and human tumour cell lines including those derived from breast, liver, lung, gastric, colorectal, squamous cell, cervical, bladder, ovarian carcinomas, neuroblastoma and leukaemia. In an in vitro murine model, epirubicin was at least as effective as doxorubicin in inhibiting basement membrane degradation, a property deemed necessary to prevent development of metastases. Significant correlations have been detected between the in vitro activity of epirubicin and other anthracyclines against various tumour specimens, and therapeutic response. Multidrug resistance to anthracyclines, vinca alkaloids, dactinomycin and epidopodophyllotoxins has been reported in a variety of tumour cell lines. In vivo antitumour activity of epirubicin was also comparable with that of doxorubicin against a variety of human tumour xenografts in mice, including breast, lung, ovarian, prostate and testicular neoplasms.
The mechanism of antitumour action for epirubicin has not been completely elucidated; however, anthracyclines appear to form a complex with DNA by intercalation between the DNA strands, thus inhibiting replication and transcription. This action may be attributed, at least in part, to interference with topoisomerase-DNA ‘cleavable complex’ and helicase activity by anthracyclines. Reduction of anthracyclines to semiquinone free radicals may cause damage to DNA, cell membrane lipids and mitochondria.
Myelotoxicity, which seems to be mediated, at least in part, via chromatid breaks in bone marrow cells, was lower with epirubicin than doxorubicin in pharmacodynamic studies. Anthracyclines appear to exert their cardiotoxic effects through a variety of mechanisms including impairment of heart mitochondrial function, depressed adenosine diphosphate-stimulated respiration, and changes in membrane structure and function. In a number of acute and chronic studies which measured these effects objectively in animals, epirubicin demonstrated a significantly lower propensity to produce cardiotoxic effects than doxorubicin, possibly due to a more rapid myocardiai release of epirubicin during the post-infusion period.
Pharmacokinetic Properties
Following rapid intravenous administration, epirubicin undergoes triphasic plasma elimination. Epirubicin exhibits a rapid initial (α) distribution phase (t½α = 1.8 to 4.8 minutes), followed by an intermediate (β) phase (t½β = 0.5 to 2.6 hours) and a much slower (γ) terminal elimination phase (t½γ = 15 to 45 hours). The t½γ of doxorubicin was approximately 40 to 70% longer than that of epirubicin in the majority of comparative pharmacokinetic studies in cancer patients. However, peak plasma drug concentrations were similar following intravenous administration of equimolar doses.
Epirubicin undergoes extensive tissue distribution; volume of distribution values were high and variable (13 to 52 L/kg), but similar to those reported for doxorubicin. Area under the plasma concentration versus time curve values adjusted for dose were 30 to 70% higher for doxorubicin than epirubicin following single-dose intravenous administration. Following intravenous administration, epirubicin is rapidly metabolised to 2 glucuronides, plus epirubicinol and 4 aglycones. Epirubicin is eliminated primarily via the hepatobiliary system, with approximately 11 to 15% of a dose eliminated in the urine as unchanged drug and metabolites. Patients with moderate to severe hepatic dysfunction exhibited reduced clearance of epirubicin and elevated plasma drug concentrations.
Therapeutic Use
At conventional doses, the combination of FEC (fluorouracil, epirubicin 50 mg/m2 plus cyclophosphamide) versus FAC (fluorouracil, doxorubicin 50 mg/m2 plus cyclophosphamide) administered every 3 weeks achieved similar objective response rates of approximately 55% and median overall survival of about 15 to 20 months in patients with advanced breast cancer in large randomised multicentre trials. Recently, dose-intensive epirubicin-containing regimens have achieved higher response rates in women with metastatic breast cancer, usually ranging from about 60 to 80%, predominantly in noncomparative studies. Dose-intensive FEC regimens often included epirubicin ⩾ 75 mg/m2 in combination with 500 mg/m2 of both fluorouracil and cyclophosphamide, while others combined epirubicin ⩾ 60 mg/m2 with higher doses of fluorouracil and cyclophosphamide administered every 3 weeks. Other dose-intensive regimens combined epirubicin with different cytotoxic drugs, used shorter intervals between treatment cycles and/or employed granulocyte colony-stimulating factor (G-CSF).
A number of studies have been, and continue to be, conducted with dose-intensive epirubicin-containing regimens, such as FEC, in the adjuvant and neoadjuvant treatment of early breast cancer. Data are generally preliminary and an advantage in favour of epirubicin dose-intensification has not yet been demonstrated in terms of survival. Interim results of a large study in premenopausal node-positive women with early stage breast cancer comparing adjuvant therapy with standard doses of FEC versus a combined regimen of cyclophosphamide, methotrexate and fluorouracil (CMF) demonstrated similar local relapse rates between treatment groups. However, a trend towards a lower rate of metastasis and fewer deaths from metastatic progression were noted among FEC recipients. In a large study evaluating neoadjuvant therapy, regimens of FEC, FAC and CMF were equally effective in producing cytoreduction sufficient to avoid radical mastectomy in patients with early breast cancer.
Noncomparative studies in previously untreated patients with small cell lung cancer (SCLC) reported objective response rates of approximately 65% with epirubicin 50 to 90 mg/m2 every 3 to 4 weeks in combination with cyclophosphamide and vincristine. Median survival was 10 to 14 months which was similar to that achieved with other active regimens. Epirubicin in combination with 1 of more other cytotoxic drugs such as carboplatin, etoposide, cisplatin, cyclophosphamide, ifosfamide and/or vincristine, generally demonstrated slightly higher response rates (52 to 95%), but median survival was essentially equivalent (7 to 16 months). Preliminary results of a dose-finding study showed a response rate of approximately 75% in patients with SCLC receiving epirubicin 100 to 140 mg/m2 in combination with cisplatin 100 mg/m2 every 3 weeks plus granulocyte-macrophage colony-stimulating factor (GM-CSF). Single-agent epirubicin 100 to 140 mg/m2 every 3 to 4 weeks was apparently less effective than combination regimens. Patients with non-small cell lung cancer (NSCLC) responded poorly to single-agent epirubicin < 120 mg/m2 every 3 or 4 weeks. In previously untreated patients, epirubicin monotherapy 120 to 180 mg/m2 every 3 to 4 weeks achieved response rates of approximately 20 to 35%, and overall survival ranged from 6 to 10 months. Similarly, such patients with NSCLC receiving epirubicin 50 to 100 mg/m2 every 3 to 4 weeks in conjunction with 1 or more other agents such as cisplatin, cyclophosphamide or etoposide, exhibited objective response rates of approximately 30 to 40%, and overall survival ranged from 5 to 11 months. High-dose epirubicin (120 mg/m2) plus cisplatin 60 mg/m2 every 4 weeks achieved an objective response rate of 54% and median overall survival of 9 months among 35 previously untreated patients with NSCLC. In another study, epirubicin 135 mg/m2 combined with etoposide offered no advantage over high dose epirubicin as a single agent and was apparently more toxic.
Epirubicin-containing combination regimens achieved complete remission rates of approximately 45 to 65% in patients with non-Hodgkin’s lymphoma. In studies of patients with intermediate- or high-grade non-Hodgkin’s lymphoma, epirubicin 50 to 75 mg/m2 was typically administered every 3 or 4 weeks in combination with cyclophosphamide, vincristine and prednisone, with or without bleomycin (CEOP or CEOP-B). Response rates and survival were equivalent between patients receiving CEOP or CEOP-B and those receiving similar regimens containing doxorubicin (CHOP or CHOP-B). Among patients with previously untreated Hodgkin’s disease, complete response was achieved in approximately 80 to 90% of adults and 62% of children receiving epirubicin-containing regimens.
In previously untreated patients with gastric cancer, objective response rates generally ranged from approximately 25 to 55%, and overall median survival ranged from about 3 to 11 months following treatment with approximately 40 to 60 mg/m2 of epirubicin every 3 or 4 weeks in specific combinations with other cytotoxic agents such as fluorouracil plus mitomycin, or epirubicin 50 to 120 mg/m2 plus fluorouracil. The higher response rates were achieved with higher epirubicin dose-intensities.
In patients with superficial bladder cancer, single or repeated intravesical administration of epirubicin 30 to 80mg following transurethral resection (TUR) achieved response rates of approximately 45 to 85% at ⩾ 1 year follow-up. Instillation of epirubicin plus TUR was more effective than surgery alone, and achieved remission rates comparable with a regimen of intravesically administered mitomycin plus TUR. Selected regimens combining epirubicin plus interferon-α2b intravesically following surgery may improve the efficacy of epirubicin in patients with superficial bladder cancer. A number of preliminary reports demonstrated promising efficacy of neoadjuvant treatment with epirubicin 30 to 60 mg/m2 intravenously every 3 or 4 weeks in combination with other antineoplastic agents, such as cisplatin plus methotrexate plus vinblastine, administered to patients with locally advanced or invasive bladder cancer. Objective response rates with neoadjuvant epirubicin-containing treatment generally ranged from approximately 45 to 90%, but appeared to be related, at least in part, to the epirubicin dosage administered. Data from 2 studies demonstrated 1.5- and 2-year survival rates of 81 and 74%, respectively, with epirubicin-containing neoadjuvant therapy.
First-line treatment of patients with advanced ovarian cancer using epirubicin 50 to 75 mg/m2 every 3 to 4 weeks in platinum-based combination regimens achieved objective response rates ranging from approximately 40 to 90%, which were similar to those obtained using doxorubicin in platinum-based chemotherapy regimens (approximately 55 to 90%) in comparative trials. Overall median survival was 14.0 to 24.9 months among epirubicin recipients and 24.3 to 26.5 months among the doxorubicin treatment groups. In patients with advanced prostatic cancer who had relapsed after hormonal treatment, epirubicin, with or without medroxyprogesterone acetate, achieved equivalent or superior improvements in performance status and/or pain relief compared with estramustine. Furthermore, preliminary results of a large study demonstrated that patients with hormone-resistant prostatic cancer were more likely to remain progression-free following epirubicin plus medroxyprogesterone acetate than with estramustine. In noncomparative trials, patients with primary hepatocellular carcinoma (PHCC) receiving intravenous or intrahepatic arterial administration of epirubicin (approximately 40 to 90 mg/m2 every 3 weeks to 3 months), with or without other antineoplastic drugs, generally exhibited objective response rates of approximately 15% and median survival of about 2 to 4 months, which were similar to those reported for doxorubicin-containing regimens. However, epirubicin plus cisplatin via intrahepatic chemoembolisation achieved objective response rates of about 50 to 60% in 2 small studies; median survival was 9 months in 1 trial and 55% of patients were alive after 1 year in the other study. In a comparative trial, median survival for patients receiving epirubicin via the hepatic artery was significantly longer than that for patients receiving doxorubicin intra-arterially (205 vs 100 days; p = 0.0036). Limited results indicate that epirubicin may also be useful as a component of chemotherapy in patients with soft tissue sarcomas, nasopharyngeal carcinoma or advanced pancreatic carcinoma.
Tolerability
Myelosuppression is the major acute dose-limiting toxicity of epirubicin, while cardiotoxicity is the most important chronic cumulative dose-limiting toxicity. Myelosuppression consists predominantly of leucopenia, which reaches a median white blood cell (WBC) nadir of about 3 × 109/L 10 to 14 days following administration of epirubicin 75 to 90 mg/m2 and usually resolves within 21 days of epirubicin administration. Approximately 3% of patients exhibit prolonged moderate to severe leucopenia (WBC < 3 × 109/L) at these doses. Comparative clinical trials and in vitro studies have demonstrated that the haematological toxicity of epirubicin is lower than that of equimolar doses of doxorubicin, thus allowing administration of higher epirubicin doses. Addition of G-CSF to high-dose epirubicin-containing regimens has been used to reduce the severity of leucopenia, and antimicrobial prophylaxis reduced the incidence of hospitalisation due to febrile neutropenia in patients receiving a dose-intensive FEC regimen as adjuvant treatment for breast cancer.
Severe irreversible congestive heart failure and/or cardiomyopathy may develop during or after epirubicin treatment, particularly with cumulative doses approaching 1000 mg/m2. However, based on in vitro studies and comparative data from clinical trials, the frequency and severity of cardiotoxicity is lower with epirubicin than doxorubicin, which is reflected in their maximum recommended cumulative doses (1000 vs 550 mg/m2). This allows for more treatment cycles and/or higher doses of epirubicin than doxorubicin. Pathological cardiac changes include myofibrillar loss, sarcoplasmic vacuolisation and fibrosis.
Most patients receiving epirubicin without adequate antiemetic therapy will experience nausea and vomiting within the first 24 hours after administration. The majority of epirubicin recipients will also develop reversible alopecia. Other adverse events include fever, diarrhoea, radiation recall and local reactions such as severe cellulitis, and the development of tissue necrosis and pain if extravasation occurs.
Dosage and Administration
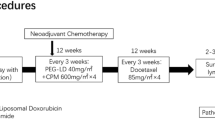
Epirubicin has been administered intravenously in various dosage regimens, alone or in combination with other antineoplastic drugs. As single-agent therapy for advanced breast cancer, epirubicin was usually administered as 75 to 90 mg/m2 every 3 to 4 weeks, although doses up to 180 mg/m2 have been used. Lower doses of epirubicin, such as 50 mg/m2 every 3 to 4 weeks, have conventionally been used in combination regimens such as FEC, but recently higher doses of 75 to 100 mg/m2 have been employed, and epirubicin 120 mg/m2 as a component of FEC was administered to a small number of patients in a dose-finding study. Similarly, dose-intensive FEC regimens have recently been employed in the adjuvant treatment of early breast cancer; epirubicin 50 mg/m2 has conventionally been used in FEC regimens, but doses up to 120 mg/m2 every 3 to 4 weeks have been become a more aggressive treatment option for this potentially curable disease.
Patients with SCLC usually received epirubicin 50 to 90 mg/m2 every 3 to 4 weeks in combination with other agents such as cyclophosphamide plus vincristine or carboplatin plus etoposide, although doses as high as 120 mg/m2 have been used in combination with cisplatin or cyclophosphamide. Epirubicin 50 to 120 mg/m2 every 3 to 4 weeks has been used in combination with 1 or more cytotoxic agents such as cisplatin, cyclophosphamide or etoposide in the treatment of NSCLC. Patients with non-Hodgkin’s lymphoma were most commonly treated with epirubicin 50 to 75 mg/m2 every 3 to 4 weeks, usually combined with cyclophosphamide, vincristine and prednisone, with or without bleomycin. In patients with Hodgkin’s disease, similar regimens of epirubicin, bleomycin, vinblastine and prednisone were used. Recommended doses for patients with advanced gastric carcinoma include monotherapy with epirubicin 75 to 100 mg/m2 every 3 to 4 weeks, or combined therapy using epirubicin 80 mg/m2 plus fluorouracil. Clinical trials have employed epirubicin doses up to 120 mg/m2 in combination regimens. Single or repeated intravesical administration of epirubicin 30 to 80mg, usually diluted in 50ml sterile solution and instilled for 1 to 2 hours following TUR, was used in patients with superficial bladder cancer. In clinical trials, patients with ovarian cancer generally received recommended epirubicin doses of 50 to 90 mg/m2 every 3 to 4 weeks combined with cisplatin plus cyclophosphamide. Epirubicin has been used at fractionated doses (approximately 25 mg/m2) every week, often in conjunction with medroxyprogesterone acetate, for the treatment of advanced prostatic carcinoma. Patients with nonresectable PHCC have been treated with epirubicin 40 to 90 mg/m2 every 3 weeks by intravenous or intrahepatic arterial administration, alone or in combination with other cytotoxic agents.
The maximum recommended cumulative dose of epirubicin is 1000 mg/m2, although this is reduced in patients previously treated with other cardiotoxic agents, mediastinal radiotherapy, and in those with a history or presence of cardiac disease. Epirubicin is usually administered intravenously over at least 3 to 5 minutes into the tubing of a free-flowing infusion of normal saline or 5% dextrose solution, and care must be taken to avoid extravasation.
Similar content being viewed by others
References
Aapro MS, Forni M, Krauer F, Alberto P, Wählbys. Preliminary analysis of combined treatment with prednimustine and 4’epidoxorubicin in advanced breast cancer. Seminars in Oncology 13: 35–38, 1986
Abate G, Castello G, Tafuto S, Romano AM, Ruffolo P. MOPP/EBVD combination chemotherapy in advanced Hodgkin’s disease: preliminary results. European Journal of Cancer 27 (Suppl. 2): S239, 1991a
Abate G, Romano AM, Tafuto S, Gailli E, Picone A. 3-year results of advanced stage poor histology NHL treated with CEOP/PEB alternating chemotherapy. European Journal of Cancer 27 (Suppl. 2): S243, 1991b
Abs L, Mahjoubi R, Bachouchi M, Azli N, Fandi K, et al. BEC (bleomycin-epirubicin-cisplatin) in metastatic (MTS)/recurrent (REC) undifferentiated nasopharyngeal cancer (UCNT). European Journal of Cancer 27 (Suppl. 2): S148, 1991
Agata N, Hirano S, Hara Y, Iguchi H, Shirai M, et al. A comparison of pirarubicin with other anthracyclines on the cardiovascular system. Oyo Yakura/Pharmacometrics 41: 467–474, 1991
Airoldi M, Pedani F, Brando V, Gabriele P, Giordano C. Cisplatin, epirubicin and 5-fluorouracil combination chemotherpay for recurrent carcinoma of the salivary gland. Tumori 75: 252–256, 1989
Ajani JA, Kanojia MD, Bodey GP. Phase II evaluation of 4’-epidoxorubicin in patients with metastatic colorectal carcinoma. Cancer Treatment Reports 68: 1507–1510, 1984
Alberola V, Conde JG, Jimeno J, Martos CF, Herranz C, et al. Phase II study with high doses of epirubicin in patients with advanced rectal cancer. Tumori 76: 503–504, 1990
Alberola V, Camps C, Munoz MA, Gomez-Codina J, Montalar J, et al. Ifosfamide, epirubicin, etoposide, combination chemotherapy for small cell lung cancer (SCLC). A multicentric phase II study. Proceedings of the American Society of Clinical Oncology 11: 308, 1992
Alberola V, Gomez-Codina J, Munoz MA, Montalar J, Guillen V, et al. Combination chemotherapy of small cell lung cancer (SCLC): a phase II-trial with epirubicin (E), ifosfamide (I) and VP16 (V). European Journal of Cancer 27 (Suppl. 2): S187, 1991
Alderton PM, Gross J, Green MD. Comparative study of doxorubicin, mitoxantrone, and epirubicin in combination with ICRF-187 (ADR-529) in a chronic cardiotoxicity animal model. Cancer Research 52: 194–201, 1992
Al-Ismail SAD, Whittaker JA, Gough J. Combination chemotherapy including epirubicin for the management of non-Hodgkin’s lymphoma. European Journal Cancer and Clinical Oncology 23: 1379–1384, 1987
Altavilla G, Adamo V, Alafaci E, Buemi B, De Stefano P, et al. High dose epirubicin in the treatment of advanced adenocarcinoma of the pancreas. European Journal of Cancer 27 (Suppl. 2): S74, 1991
Aitavilla G, Adamo V, Alafaci E, Buemi B, Caristi N, et al. VP16, epirubicin and procarbazine in the treatment of advanced nonsmall-cell lung cancer. Tumori 75: 168–170, 1989
Andersson M, Domellöf L, Eksborg S, Häggmark S, Johansson G, et al. Pharmacokinetics and central haemodynamic effects of doxorubicin and 4’epi-doxorubicin in the pig. Acta Oncologica 28: 709–714, 1989
Anderström C, Eddeland A, Folmerz P, Hansson R. Epirubicine and medroxyprogesteron acetate (MPA) or estramustine phosphate in the treatment of hormone resistant prostatic cancer. European Journal of Cancer 27 (Suppl. 2): SI 16, 1991
Anon. WHO handbook for reporting results of cancer treatment. Geneva, WHO Offset Publication No. 48, WHO, pp. 1–45, 1979
Antón A, Rosell R, Gómez-Codina J, Vadell C, Jimeno J, et al. Escalating-dose epirubicin plus cisplatin in small-cell lung cancer with granulocyte-macrophage colony-stimulating (GM-CSF) use where appropriate. Abstract no. 158. Annals of Oncology 3 (Suppl. 5): 40, 1992
Aoki K, Shimada Y, Tajiri H, Shirao K, Fukuda H, et al. Phase II study of epirubicin in advanced adenocarcinoma of the pancreas. Nip-chir-gakk Nippon Gan Chiryo Gakkai-Shai 27: 487, 1992
Aravantinos G, Linardou H, Christodoulou Ch, Samanta D, Pikramenos D, et al. M-VEP chemotherapy in transitional cell urothelial carcinoma (T.C.U.C.). European Journal of Cancer 27 (Suppl. 2): S102, 1991
Arcamone F, Lazzati M, Vicario GP, Zini G. Disposition of Relabelled 4’-epidoxorubicin and doxorubicin in the rat. A comparative study. Cancer Chemotherapy and Pharmacology 12: 157–166, 1984
Arcamone F. Properties of antitumor anthracyclines and new developments in their application: Cain Memorial Award Lecture. Cancer Research 45: 5995–5999, 1985
Ardizzoni A, Rosso R, Salvati F, Scagliotti G, Soresi E, et al. Combination chemotherapy and interferon α2b in the treatment of advanced non-small-cell lung cancer. American Journal of Clinical Oncology 14: 120–123, 1991
Armitage JO, Cheson BD. Interpretation of clinical trials in diffuse large-cell lymphoma. Journal of Clinical Oncology 6: 1335–1347, 1988
Athlin L, Domellöf L, Norberg B. Effect of therapeutic concentrations of anthracyclines on monocyte phagocytosis of yeast cells. European Journal of Clinical Pharmacology 36: 155–159, 1989
Azuma J, Sperelakis N, Hasegawa H, Tanimoto T, Vogel S, et al. Adriamycin cardiotoxicity: possible pathogenic mechanisms. Journal of Molecular and Cellular Cardiology 13: 381–397, 1981
Bachouchi M, Cvitkovic E, Azli N, Gasmi J, Cortes-Funes H et al. High complete response in advanced nasopharyngeal carcinoma with bleomycin, epirubicin, and cisplatin before radiotherapy. Journal of the National Cancer Institute 82: 616–620, 1990
Bachur NR, Yu F, Johnson R, Hickey R, Wu Y, et al. Helicase inhibition by anthracycline anticancer agents. Molecular Pharmacology 41: 993–998, 1992
Bachur NR, Gordon SL, Gee MV, Kon H. NADPH cytochrome P-450 reductase activation of quinone anticancer agents to free radicals. Proceedings of the National Academy of Sciences of the United States of America 76: 954–957, 1979
Bachur NR, Gordon SL, Gee MV. Anthracycline antibiotic augmentation of microsomal electron transport and free radical formation. Molecular Pharmacology 13: 901–910, 1977
Bachur NR, Gordon SL, Gee MV. A general mechanism for microsomal activation of quinone anticancer agents to free radicals. Cancer Research 38: 1745–1750, 1978
Bader GR, Costa MA, Lamarca JE, Salgado R, Froimtchuk MJ. Primary treatment of poor prognosis locally advanced breast cancer with etoposide, 4-epirubicin, platinum (EEP): preliminary results. Proceedings of the American Society of Clinical Oncology 11: 66, 1992
Bagnara GP, Rocchi P, Bonsi L, Valvassori L, Marini M, et al. The in vitro effect of epirubicin on human normal and leukemic hemopoietic cells. Anticancer Research 7: 1197–1200, 1987
Bailey NP, Clegg JA, Barton CM, Stanley A, Elias E, Neuberger J, et al. Is there a role for combination chemotherpay in hepatobilliary and pancreatic carcinoma: a phase II study of combined epirubicin infusion together with mitoxantrone bolus therapy. Abstract no. 100. Annals of Oncology 3 (Suppl. 5): 26, 1992
Baltali E, Içli F, Onat H, Mandel N, Berk Ö, et al. Phase II study of weekly doses of epirubicin in metastatic breast cancer. European Journal of Cancer 27 (Suppl. 2): S59, 1991
Baltali E, Kars A, Oner Z, Ozisik Y, Guler N, et al. CEF combination as neoadjuvant therapy in stage III breast cancer patients. Abstract no. 384. Annals of Oncology 3 (Suppl. 5): 99, 1992
Banham SW, Henderson AF, Bicknell S, Hughes J, Milroy R. High dose epirubicin chemotherapy in untreated poorer prognosis small cell lung cancer. Respiratory Medicine 84: 241–244, 1990
Barker GH, Wiltshaw E. Use of high dose cis-dichlorodiammine platinum (II) (NSC-119875) following failure on previous chemotherapy for advanced carcinoma of the ovary. British Journal of Obstetrics and Gynaecology 88: 1192–1199, 1981
Barone C, Cassano A, Astone A, Ricevuto E, Fontana T, et al. Association of epirubicin, etoposide and cisplatin in gastric cancer. Oncology 48: 353–355, 1991
Bartkowiak D, Hemmer J, Röttinger E. Dose dependence of the cytokinetic and cytotoxic effects of epirubicin in vitro. Cancer Chemotherapy and Pharmacology 30: 189–192, 1992
Bastholt L, Dalmark M, Gjedde S, Pfeiffer P, Petersen D, et al. Epirubicin at four different dose levels in metastatic breast cancer. A randomized trial. Proceedings of the American Society of Clinical Oncology 11: 56, 1992
Bastholt L, Mouridsen HT. How to improve cytotoxic therapy in advanced breast cancer. Acta Oncologica 29: 349–355, 1990
Becher R, Hayungs J, Abelein G, Hartwich G, Bartels H, et al. Epirubicin and ifosfamide in advanced breast cancer. Proceedings of the American Association of Cancer Research 33: 214, 1992
Becher R. Evaluation of the optimal duration of chemotherapy in advanced breast cancer. Onkologie 14 (Suppl. 3): 18–19, 1991
Becouarn Y, Bui BN, Kerbrat P, Chauvergne J, Brunet R, et al. Traitement des sarcomes des tissus mous de l’adulte par une association de vindesine et de cisplatine avec doxorubicine ou epirubicine: une etude pilote. Bulletin du Cancer 74: 109–116, 1987
Bellamy WT, Dalton WS, Kailey JM, Gleason MC, McCloskey TM, et al. Verapamil reversal of doxorubicin resistance in multidrug-resistant human myeloma cells and association with drug accumulation and DNA damage. Cancer Research 48: 6365–6370, 1988
Benedetto P, Ahmed T, Needles B, Watson RC, Yagoda A. Phase II trial of 4’epi-adriamycin for advanced hypernephroma. American Journal of Clinical Oncology 6: 553–554, 1983
Berens ME, Saito T, Welander CE, Modest EJ. Antitumor activity of new anthracycline analogues in combination with interferon alfa. Cancer Chemotherapy and Pharmacology 19: 301–306, 1987
Beretta G, Locatelli C, Tabiadon D, Labianca R, Fraschini P, et al. Epirubicin treatment of advanced breast carcinoma with the weekly low-dose regimen. Oncology 44: 6–12, 1987
Beretta G, Villa E, Tancini G, Frontini L, Arnoldi E, et al. Clinical evaluation of an intensive polychemotherapy FEC 1+8 for advanced/metastatic breast carcinoma (ABC). Proceedings of the American Society of Clinical Oncology 11: 79, 1992
Berman E, Casper ES, Howard J, Wittes RE. Phase II trial of 4’-epi-doxorubicin in patients with advanced malignant melanoma. Cancer Treatment Reports 68: 679–680, 1984
Bertazzoli C, Rovero C, Ballerini L, Lux B, Balconi F, et al. Experimental systemic toxicology of 4’-epidoxorubicin, a new, less cardiotoxic anthracycline antitumor agent. Toxicology and Applied Pharmacology 79: 412–422, 1985
Bezwoda WR, Dansey R, Seymour L. High-dose 4’-epiadriamycin for treatment of breast cancer refractory to standard dose anthracycline chemotherapy: achievement of second responses. Oncology 47: 4–8, 1990
Bezwoda WR. Treatment of advanced ovarian cancer: a randomised trial comparing adriamycin or 4’epi-adriamycin in combination with cisplatin and cyelophosphamide. Medical and Pediatric Oncology 14: 26–29, 1986
Bigotte L, Olsson Y. Distribution and toxic effects of intravenously injected epirubicin on the central nervous system of the mouse. Brain 112: 457–469, 1989
Blackstein M, Eisenhauer EA, Wierzbicki R, Yoshida S. Epirubicin in extensive small-cell lung cancer: a phase II study in previously untreated patients: a national cancer institute of Canada Clinical Trials Group Study. Journal of Clinical Oncology 8: 385–389, 1990
Blomqvist C, Elomaa I, Rissanen P, Hietanen P, Nevasaari K, et al. FEC (5-fluorouracil-epirubicin-cyclophosphamide) monthly versus FEC weekly in metastatic breast cancer. Acta Oncologica 31: 231–236, 1992
Boccardo F, Rubagotti A, Amoroso D, Sismondi P, Genta F, et al. Chemotherapy versus tamoxifen versus chemotherapy plus tamoxifen in node-positive, oestrogen-receptor positive breast cancer patients. An update at 7 years of the 1st GROCTA (Breast Cancer Adjuvant Chemo-hormone Therapy Cooperative Group) Trial. European Journal of Cancer 28: 673–680, 1992
Boccardo F, Rubagotti A, Bruzzi P, Cappellini M, Isola G, et al. Chemotherapy versus tamoxifen versus chemotherapy plus tamoxifen in node-positive, estrogen receptor-positive breast cancer patients: results of a multicentric Italian study. Journal of Clinical Oncology 8: 1310–1320, 1990
Bokemeyer C, Harstrick A, Schmoll H-J, Könne-Wömpner CH, Schöffski P, et al. Epirubicin/dacarbazine rapidly alternated with ifosfamide in the treatment of metastatic soft tissue sarcomas. Annals of Oncology 3: 487–488, 1992
Bolognesi A, Villa E, Tancini G, Beretta G. Intensive anthracycline polychemotherapy-FCE-for advanced metastatic breast carcinoma (BC). European Journal of Cancer 27 (Suppl. 2): S55, 1991
Bonadonna G, Veronesi U, Brambilla C, Ferrari L, Luini A, et al. Primary chemotherapy to avoid mastectomy in tumors with diameters of three centimeters or more. Journal National Cancer Institute 82: 1539–1545, 1990
Bonfante V, Ferrari L, Brambilla C, Rossi A, Fabrizio V, et al. New anthracycline analogs in advanced breast cancer. European Journal of Cancer and Clinical Oncology 22: 1379–1385, 1986
Bonfante V, Villani F, Bonadonna G. Toxic and therapeutic activity of 4’-epidoxorubicin. Tumori 68: 105–111, 1982
Bouleuc C, Rousseau F, Extra JM, Giachetti S, Espie M, et al. Intensive induction chemotherapy (IC) with cyelophosphamide (C) and epirubicin (E) in patients (PTS) with metastatic breast cancer (MBC). Abstract no. 438. Annals of Oncology 3 (Suppl. 5): 114, 1992
Boven E, Schlüper M, Erkelens CAM, Pinedo HM. Doxorubicin compared with related compounds in a nude mouse model for human ovarian cancer. European Journal of Cancer 26: 983–986, 1990
Brambilla C, Rossi A, Bonfante V, Ferrari L, Villani F, et al. Phase II study of doxorubicin versus epirubicin in advanced breast cancer. Cancer Treatment Reports 70: 261–265, 1986
Brandwein JM, Callum J, Sutcliffe SB, Scott JG, Keating A. Evaluation of cytoreductive therapy prior to high dose treatment with autologous bone marrow transplantation in relapsed and refractory Hodgkin’s disease. Bone Marrow Transplantation 5: 99–103, 1990
Brincker H. Distant recurrence in breast cancer. Survival expectations and first choice of chemotherapy regimen. Acta Oncologica 27: 729–732, 1988
Bristow MR, Kantrowitz NE, Harrison WD, Minobe WA, Sageman WS, et al. Mediation of subacute anthracycline cardiotoxicity in rabbits by cardiac histamine release. Journal of Cardiovascular Pharmacology 5: 913–919, 1983
Bristow MR, Minobe WA, Billingham ME, Marmor JB, Johnson GA, et al. Anthracycline-associated cardiac and renal damage in rabbits. Laboratory Investigation 45: 157–168, 1981
Bristow MR, Sageman WS, Scott RH, Billingham ME, Bowden RE, et al. Acute and chronic cardiovascular effects of doxorubicin in the dog: the cardiovascular pharmacology of drug-induced histamine release. Journal of Cardiovascular Pharmacology 2: 487–515, 1980
Broggini M, Colombo T, Martini A, Donelli MG. Studies on the comparative distribution and biliary excretion of doxorubicin and 4’-epi-doxorubicin in mice and rats. Cancer Treatment Reports 64: 897–904, 1980
Bruckner HW, Cohen CJ, Goldberg JD, Kabakow B, Wallach R, et al. Improved chemotherapy for ovarian cancer with Cisdiamminedichloroplatinum and adriamycin. Cancer 47: 2288–2294, 1981
Budman DR, Wood W, Henderson IC, Korzun AH, Cooper R, et al. Initial findings of CALGB 8541: A dose and dose intensity trial of cyelophosphamide (C), doxorubicin (A), and 5-fluorouracil (F) as adjuvant treatment of stage II, node+, female breast cancer. 28th Annual Meeting of the American Society of Clinical Oncology, Abstract no. 29. San Diego, May 17–19, 1992
Burk K, Sropp C, Schultze-Seemann W, de Riese W. Chemotherapy of hormone-resistant carcinoma of the prostrate: 4-Epi-rubicin weekly v.s. DMF monthly. In Prostate cancer, Part B: imaging techniques, radiotherapy, chemotherapy, and management issues, pp. 243–246, Alan R. Liss, Inc., 1987
Buzzoni R, Bonadonna G, Valagussa P, Zambetti M. Adjuvant chemotherapy with doxorubicin plus cyclophosphamide, methotrexate, and fluorouracil in the treatment of resectable breast cancer with more than three positive axillary nodes. Journal of Clinical Oncology 9: 2134–2140, 1991
Calais da Silva F, Denis L, Bono A, Bollack C, Bouffioux C. Intravesical chemoresection with 4’-epi-doxorubicin in patients with superficial bladder tumors. European Urology 14: 207–209, 1988
Calais da Silva F, Ferrito F, Brandão T, Santos A. 4’-epidoxorubicin versus mitomycin C intravesical chemoprophylaxis of superficial bladder cancer. European Urology 21: 42–44, 1992
Calero F, Asins-Codoner E, Jimeno J, Rodriguez-Escudero F, Mendana J, et al. Epirubicin in advanced endometrial adenocarcinoma: a phase II study of the Grupo Ginecologico Espanol para el Tratamiento Oncologico (GGETO). European Journal of Cancer 27: 864–866, 1991
Calero F, Rodriguez-Escudero F, Jimeno J, Mendana, Iglesias J, et al. Single agent epirubicin in squamous cell cervical cancer. Acta Oncologica 30: 325–327, 1991
Calero F, Jimeno J, Rodriguez-Escudero F, Codoner JA, Mendana J, et al. Epirubicin: clinical toxicity during the phase II program in endometrial and cervical cancer. European Journal of Gynaecological Oncology 13: 83–89, 1992
Camaggi CM, Comparsi R, Strocchi E, Testoni F, Angelelli B, et al. Epirubicin and doxorubicin comparative metabolism and pharmacokinetics. Cancer Chemotherapy and Pharmacology 21: 221–228, 1988
Camaggi CM, Strocchi E, Comparsi R, Testoni F, Angelelli B, et al. Biliary excretion and pharmacokinetics of 4’epidoxorubicin (epirubicin) in advanced cancer patients. Cancer Chemotherapy and Pharmacology 18: 47–50, 1986
Camaggi CM, Strocchi E, Martoni A, Angelelli B, Comparsi R, et al. Epirubicin plasma and blood pharmacokinetics after single i.v. bolus in advanced cancer patients. Drugs Under Experimental and Clinical Research 11: 285–294, 1985
Camaggi CM, Strocchi E, Tamassia V, Martoni A, Giovannini M et al. Pharmacokientic studies of 4’-epi-doxorubicin in cancer patients with normal and impaired renal function and with hepatic metastases. Cancer Treatment Reports 66: 1819–1824, 1982
Campora E, Nobile MT, Sertoli MR, Rosso R. Phase II study of 4’-epi-doxorubicin in advanced breast cancer. Cancer Treatment Reports 68: 1285–1286, 1984
Cantoni O, Sestili P, Cattabeni F, Geroni C, Giuliani F. Comparative effects of doxorubicin. and 4’-epi-doxorubicin on nucleic acid metabolism and cytotoxicity in a human tumor cell line. Cancer Chemotherapy and Pharmacology 27: 47–51, 1990
Cantoni O, Sestili P, Cattabeni F, Geroni C, Grandi M, et al. Cellular and molecular pharmacology of 4’-epidoxorubicin in HeLa cells. Journal of Cancer Research and Clinical Oncology 115: 373–378, 1989
Cantwell BMJ, Bozzino JM, Corris P, Harris AL. The multidrug resistant phenotype in clinical practice: evaluation of cross resistance to ifosfamide and mesna after VP16–213, doxorubicin and vincristine (VPAV) for small cell lung cancer. European Journal of Cancer and Clinical Oncology 24: 123–129, 1988
Carmo-Pereira J, Costa FO, Miles DW, Henriques E, Richards MA, et al. High-dose epirubicin as primary chemotherapy in advanced breast carcinoma: a phase II study. Cancer Chemotherapy and Pharmacology 27: 394–396, 1991
Carmo-Pereira J, Henriques E, Costa FO. 5-Fluorouracil (F), epirubicin (E), and cyclophosphamide (C) [FEC] as first-line chemotherapy in disseminated breast carcinoma, with or without granulocyte colony stimulating factor (G-CSF). Preliminary results. Proceedings of the American Society of Clinical Oncology 11: 74, 1992
Caroni P, Villani F, Carafoli E. The cardiotoxic antibiotic doxorubicin inhibits the Na+/CA2+ exchange of dog heart sarcolemmal vesicles. Febs Letters 130: 184–186, 1981
Carli N, Flamant F, Treuner J, Prascella E, Stevens M, et al. High dose epirubicin (EPI) in combination with carboplatin (CBDCA) and vincristine (VCR) in stage IV childhood malignant mesenchymal tumors (MMT). Proceedings of the American Society of Clinical Oncology 11: 369, 1992
Carrion RP, Jimeno JM, Jimenez V, Guardiola L, Donnay O, et al. Traitement du cancer avancé du sein par une polychimiothérapie de type FEC utilisant l’épirubicine à la dose de 75 mg/m2 (FEC 75). Bulletin du Cancer 77: 1033–1035, 1990
Cartei G, Nascimben O, Veronesi A, Foladore S, Delia Valentina M, et al. Cyclophosphamide, epirubicin and vincristine (CEV) vs cisplatin and etoposide (PE): a randomized study. European Journal of Cancer 27 (Suppl. 2): S185, 1991
Casadio M, Lelli G, Giordani S, Boltri B, Blotta A, et al. Small cell bronchogenic carcinoma: a cyclical alternating combination of epirubicin plus cisplatin and cyclophosphamide plus etoposide. Journal of Chemotherapy 2: 199–202, 1990
Casali P, Pastorino U, Zucchinelli P, Devizzi L, Azzarelli A, et al. Epirubicin plus ifosfamide and dacarbazine (EID) in advanced soft tissue sarcomas. Annals of Oncology 3: 125–126, 1992
Case Jr DC, Ervin TJ, Gams R, Sonneborn HL, Paul SD, et al. Phase I-II study of epirubicin in multiple myeloma. Cancer Research 48: 6246–6248, 1988
Cassinelli G, Configliacchi E, Penco S, Rivola G, Arcamone F, et al. Separation, characterization, and analysis of epirubicin (4’-epi-doxorubicin) and its metabolites from human urine. Drug Metabolism and Disposition 12: 506–511, 1984
Cazap E, Estevez R, Bruno M, Levy D, Algamiz C, et al. Phase II trial of 4’-epi-doxorubicin in locally advanced or metastatic gastric cancer. Tumori 74: 313–315, 1988
Cellerino R, Tummarello D, Guidi F, Isidori P, Raspugli M, et al. A randomized trial of alternating chemotherapy versus best supportive care in advanced non-small-cell lung cancer. Journal of Clinical Oncology 9: 1453–1461, 1991
Cersosimo RJ, Hong WK. Epirubicin: a review of the pharmacology, clinical activity, and adverse effects of an adriamycin analogue. Journal of Clinical Oncology 4: 425–439, 1986
Chadjaa M, Izzo J, Levin FM, Riggi M, Armand JP, et al. Preliminary data on 4’epiadriamycine (EPI)-vinorelbine (VNB): a new active combination in advanced breast cancer (ABC). Proceedings of the American Association of Cancer Research 33: 214, 1992
Chauffert B, Pelletier H, Corda C, Solary E, Bedenne L, et al. Potential usefulness of quinine to circumvent the anthracycline resistance in clinical practice. British Journal of Cancer 62: 395–397, 1990
Cheirsilpa A, Kosiyatrakul T, Benjachai W, Tangkrutt S, Puriphat S. Primary chemotherapy for locally advanced uterine cervical carcinoma. Gan to Kagaku Ryoho 19: 1224–1232, 1992
Chen ZM, Colombo T, Conforti L, Donelli MR, Fiedorowicz RJ, et al. Effects of three new anthracyclines and doxorubicin on the rat isolated heart. Journal of Pharmacy and Pharmacology 39: 947–950, 1987
Cheson BD. New chemotherapeutic agents for non-Hodgkin’s lymphomas. Drugs of Today 27: 157–176, 1991
Chevallier B, Leyvraz S, Olivier JP, Fargeot P, Facchini T, et al. Epirubicin and ifosphamide in advanced soft tissue sarcoma (ASTS): a phase II study. European Journal of Cancer 27 (Suppl. 2): S162, 1991
Chevallier B, Montcuquet P, Fachini T, Bui BN, Kerbrat P, et al. Phase II study of epirubicin in advanced soft tissue sarcoma. Bulletin du Cancer 77: 991–995, 1990
Cocconi G, Bella M, Zironi S, Algeri R, Bartolocci R, et al. A prospective randomized trial comparing FAM combination with PELF combination in advanced gastric carcinoma. Proceedings of the American Society of Clinical Oncology 11: 163, 1992
Cognetti F, Schornagel J, Wildiers J, De Mulder P, Verweij J, et al. Phase II trial of epirubicin (EPI) and cisplatin (DDP) in advanced undifferentiated nasopharingeal carcinoma (UNCP). European Journal of Cancer 27 (Suppl. 2): S148, 1991
Coleman R, Towlson K, Wiltshaw E, Slevin M, Blake P, et al. Epirubicin for pretreated advanced ovarian cancer. European Journal of Cancer 26: 850–851, 1990
Colucci G, Maiello E, Valori V, Pezzella G, Giuliani F. A randomized study with 5-FU + folinic acid (FA) alone or with epirubicin (E) in advanced gastric carcinoma. European Journal of Cancer 27 (Suppl. 2): S81, 1991
Cornelia P, Abate G, Fiore M, Di Finizio G, Polverino W, et al. Cyclophosphamide, epirubicin, vincristine and prednisone (CEOP) in the treatment of advanced stage non-Hodgkin’s lymphomas with poor histology. Haematologica 73: 509–512, 1988
Conte PF, Pronzato P, Rubagotti A, Alama A, Amadori D, et al. Conventional versus cytokinetic polychemotherapy with estrogenic recruitment in metastatic breast cancer: results of a randomized cooperative trial. Journal of Clinical Oncology 5: 339–347, 1987
Cosolo W, Zimet A, McKendrick J, Zalcberg JR. The effect of age on the pharmacokinetics of epirubicin. European Journal of Cancer 27 (Suppl. 2): S199, 1991
Cova D, Sassano M, Monti E, Piccinini F. DNA damage induced by doxorubicin, 4’-epidoxorubicin and their copper (II) complexes. Archives of Toxicology 64: 597–598, 1990
Cruciani G, Fornarola V, Emiliani E, Montanari F, Dazzi C, et al. Conservative treatment in T2-T4 bladder cancer, with radiotherapy and rescue-m-vec: a pilot study. European Journal of Cancer 27 (Suppl. 2): S103, 1991
Cruz JJ, Garcia J, Fonseca E, Gomez A, Martin G, et al. MEV (mitomycin C, epirubicine and vincristine) as 2° line after CMF cyclophosphamide, methotrexate and 5-fluoracil) in the treatment of advanced breast cancer. European Journal of Cancer 27 (Suppl. 2): S65, 1991
Cumming JA, Kirk D, Newling DW, Hargreave TB, Whelan P. A multi-centre phase two study of intravesical epirubicin in the treatment of superficial bladder tumour. European Urology 17: 20–22, 1990
Cvitkovic E, Bachouchi M, Armand J-P. Nasopharyngeal Carcinoma. Biology, natural history, and therapeutic implications. Head and Neck Cancer 5: 821–839, 1991
Dahl O. Hyperthermic potentiation of doxorubicin and 4’-epi-doxorubicin in a transplantable neurogenic rat tumor (BT4A) in BD IX rats. International Journal of Radiation Oncology Biology Physics 9: 203–207, 1983
Dardir M, Herman EH, Ferrans VJ. Effects of ICRF-187 on the cardiac and renal toxicity of epirubicin in spontaneously hypertensive rats. Cancer Chemotherapy and Pharmacology 23: 269–275, 1989a
Dardir MD, Ferrans VJ, Mikhael YS, El-Guindy MS, El-Aasar A-B, et al. Cardiac morphologic and functional changes induced by epirubicin chemotherapy. Journal of Clinical Oncology 7: 947–958, 1989b
Decorti G, Klugmann FB, Candussio L, Baldini L. Characterization of histamine secretion induced by anthracyclines in rat peritoneal mast cells. Biochemical Pharmacology 35: 1939–1942, 1986
Delaere KP, Leliefeld H, Peulen F, Stapper EW, Smeets J, et al. Phase II study of epirubicin in advanced hormone-resistant prostatic carcinoma. British Journal of Urology 70: 641–642, 1992
De Lena M, Maiello E, Lorusso V, Brandi M, Calabrese P, et al. Comparison of CHOP-B vs CEOP-B in ‘poor prognosis’ non-Hodgkin’s lymphomas. A randomized trial. Medical Oncology and Tumor Pharmacotherapy 6: 163–169, 1989
Delfino C, Caccia G, Fein L, Chirino M, et al. Cisplatin (C), epirubicin (E) and 5-fluoruracil (F) in patients (pts) with advanced gastric cancer (AGC). European Journal of Cancer 27 (Suppl. 2): S81, 1991
De Marinis F, Noseda MA, Migliorino MR, Pallotta G. Phase II trial of high-dose epirubicin and cisplatin with vindestine in unresectable non small-cell lung cancer (NSCLC). European Journal of Cancer 27 (Suppl. 2): S171, 1991b
De Marinis F, Nunziati F, Noseda MA, Signora M, Vaccarino M, et al. Phase II trial of high dose epirubicin and ifosfamide in adanced non small-cell lung cancer (NSCLC). European Journal of Cancer 27 (Suppl. 2): S174, 1991a
Demicheli R, Bonciarelli G, Jirillo A, Lonardi F, Balli M. Bleomycin, vincristine, mitomycin and cisplatin alternated with cyclophosphamide, 4’-epidoxorubicin and procarbazine in advanced non-small-cell lung cancer. Tumori 74: 563–566, 1988
De Oliveira CF, Brandão T, Gervasio H, Gordilho J, Rosa F, et al. Treatment of advanced and/or recurrent breast cancer with 5-fluoruracil (F) + novantrone (N) + metotrexate (M), FNM) versus, 5-fluoruracil (F) + 4’-epirubicin (E) + cyclophosphamide (C), (FEC). A randomized phase II study. Proceedings of the American Society of Clinical Oncology 11: 72, 1992
DeVita Jr VT, Hubbard SM, Young RC, Longo DL. The role of chemotherapy in diffuse aggressive lymphomas. Seminars in Hematology 25: 2–10, 1988
de Vries EG, Greidanus J, Mulder NH, Nieweg MB, Postmus PE, et al. A phase I and pharmacokinetic study with 21-day continuous infusion of epirubicin. Journal of Clinical Oncology 5: 1445–1451, 1987
de Vries EGE, Nanninga AG, Greidanus J, Oremus EThHGJ, Verschueren RCJ, et al. A phase II study of a 21 day continuous infusion schedule with epirubicin in advanced gastric cancer. European Journal of Cancer and Clinical Oncology 25: 1509–1510, 1989
Diaz-Maqueo JC, Aviles A, Talavera A, Garcia EL, Guzman R, et al. Triple alternating chemotherapy (TAC) versus TAC plus interferon alfa 2b induction therapy in high risk multiple myeloma. European Journal of Cancer 27 (Suppl. 2): S249, 1991
Diaz-Rubio E, Jimeno J, Aranda E, Massuti B, Camps C, et al. Etoposide (E) + epirubicin (E) + cisplatin (P) combination chemotherapy (EEP) in advanced gastric cancer: negative impact on clinical outcome. Annals of Oncology 3: 861–863, 1992
Di Lauro L, Natali M, Gionfra T, Vici P, Casali A, et al. Fluo-rouracil (F), epirubicin (E) and cisplatin (P) (FEP) for advanced gastric carcinoma (AGC). European Journal of Cancer 27 (Suppl. 2): S80, 1991
Di Marco A, Casazza AM, Gambetta R, Supino R, Zunino F. Relationship between activity and amino sugar stereochemistry of daunorubicin and adriamycin derivatives. Cancer Research 36: 1962–1966, 1976
Di Marco A, Zunino F, Silvestrini R, Gambarucci C, Gambetta RA. Interaction of some derivatives with deoxyribonucleic acid and their biological activity. Biochemical Pharmacology 20: 1323–1328, 1971
Dixon AR, Robertson JFR, Athanassiou E, Jackson L, Blarney RW. Weekly low dosage epirubicin in advanced breast cancer. European Journal of Cancer 26: 847–848, 1990
Djaldetti M, Gilgal R, Shainberg A, Klein B, Zahavi I. SEM observations on the effect of anthracycline drugs on cultured newborn rat cardiomyocytes. Basic Research in Cardiology 83: 672–677, 1988
Dobbs NA, Twelves CJ, Gillies H, Richards MA, Rogers HJ, et al. Comparative pharmacokinetics and metabolism of doxorubicin (DOX) and epidoxorubicin (EPI) in relation to liver biochemistry tests. British Journal of Cancer 63 (Suppl. 8): 46, 1991
Drings P, Bülzebruck H, Hruska D, Manke HG, Schuler G. EPICO für die Behandlung des kleinzelligen Bronchialkarzinoms. 3. Zwischenanalyse. Onkologie 9 (Suppl. 1): 14–20, 1986
Early Breast Cancer Trialists’ Collaborative Group. Effects of adjuvant tamoxifen and of cytotoxic therapy on mortality in early breast cancer. New England Journal of Medicine 319: 1681–1692, 1988
Early Breast Cancer Trialists’ Collaborative Group. Systemic treatment of early breast cancer by hormonal, cytotoxic, or immune therapy. Part 2. Lancet 339: 1–15, 1992
Ebbs SR, Saunders JA, Graham H, A’Hern RP, Bates T, et al. Advanced breast cancer. A randomised trial of epidoxorubicin at two different dosages and two administration systems. Acta Oncologica 28: 887–892, 1989
Eckhardt S, Juhos E, Hindy I, Jelic S, Koza I, et al. Activity of epirubicin and dibromodulcitol in advanced breast cancer. Oncology 45: 409–412, 1988
Eckhardt S, Kolaric K, Vukas D, Kánitz E, Schoket Zs, et al. Phase II study of 4’-epi-doxorubicin in patients with untreated, extensive small cell lung cancer. Medical Oncology and Tumor Pharmacotherapy 7: 19–23, 1990
Ejlertsen B, Pfeiffer P, Pedersen D, Mouridsen HT, Rose C, et al. Loss of efficacy observed reducing duration of CEF from 18 to 6 months in the treatment of advanced breast cancer. IV Scandinavian Breast Cancer Symposium, Finland p. 25, 3–5 June, 1991
Eksborg S, Andersson M, Domellöf L, Lönroth U. A pharmacokinetic study of adriamycin and 4’epi-adriamycin after simultaneous intra-arterial liver administration. Medical Oncology and Tumor Pharmacotherapy 3: 105–110, 1986b
Eksborg S, Hardell L, Bengtsson N-O, Sjödin M, Elfsson B. Epirubicin as a single agent therapy for the treatment of breast cancer — a pharmacokinetic and clinical study. Medical Oncology and Tumour Pharmacotherapy 9; 75–80, 1992
Eksborg S, Mattson K. Pharmacokinetics of epirubicin in man. Non-influence of alpha interferon. Medical Oncology and Tumor Pharmacotherapy 5: 131–133, 1988
Eksborg S, Stendahl U, Lönroth U. Comparative pharmacokinetic study of adriamycin and 4’Epi-adriamycin after their simultaneous intravenous administration. European Journal of Clinical Pharmacology 30: 629–631, 1986a
Eksborg S. Anthracycline pharmacokinetics. Acta Oncologica 29: 339–342, 1990
Eliopoulos A, Kerr DJ, Spandidos DA. The effect of doxorubicin, daunorubicin and 4’-epidoxorubicin on the exogenous c-myc promoter in mouse erythroleukemia cells. Anticancer Research 11: 2153–215, 1991
Elisson LO, Björkman S. Congestive heart failure in rabbits after a single intrapleural administration of a low dose of doxorubicin or epirubicin. Pharmacology and Toxicology 62: 84–89, 1988
El Mawla NG, Hamza MR, El Khodari A, Khaled H, Gaafar R, et al. A phase II study of epirubicin in breast cancer. Anti-Cancer Drugs 2: 371–374, 1991.
Elomaa I, Kellokumpu-Lehtinen P, Rannikko S, Alfthan O. Hormone-resistant metastatic prostate cancer. European Urology 19: 12–15, 1991
Epirubicin Study Group for Hepatocellular Carcinoma. Intra-arterial administration of epirubicin in the treatment of nonresectable hepatocellular carcinoma. Cancer Chemotherapy and Pharmacology 19: 183–189, 1987
Erazo A, Olguin A, Cervantes G, Guizar R, Capdeville D, et al. Neo-adjuvant chemotherapy in locally advanced breast cancer. Stage III. A randomized, prospective, comparative study of FAC (5-fluorouracil (FU), adriamycin (A), cyclophosphamide (C) vs FEC (5-flourouracil (FU), epirubicin (E), cyclophosphamide (C). European Journal of Cancer 27 (Suppl. 2): S32, 1991
Evans TRJ, Glees JP, Knight MJ, Leicester RJ, Fiennes A, et al. The use of a new combination chemotherapy: epirubicin, cisplatin and continous infusion 5-fluorouracil (ECF) in upper gastrointestinal (GI) tumours. Abstract no. 52. Annals of Oncology 3 (Suppl. 5): 14, 1992b
Evans TRJ, Millard FJC, Sanders KB, Coombes RC, Mansi JL. A randomised trial of 3 versus 6 cycles of combination chemotherapy with etoposide and epirubicin (EE) in small cell lung cancer (SCLC). Abstract no. 154. Annals of Oncology 3 (Suppl. 5): 39, 1992a
Extra JM, Espie M, Culine S, Morvan M, Bourstyn E, et al. High dose short interval cyclophosphamide epirubicin induction phase in inflammatory breast cancer. 4th International Conference on Adjuvant Therapy of Primary Breast Cancer, St Gallen, pp. 83, 26–29 Feb. 1992
Feld R, Ginsberg RJ, Payne DG. Treatment of small cell lung cancer. In Roth JA (Ed.) Thoracic oncology, pp. 229–262, 1989
Fedeli SL, Cascinu S, Fedeli A, Casadei V, Fabrizi G, et al. Neoadjuvant chemotherapy with cyclophosphamide, epirubicin and cisplatin (CEP) for invasive bladder cancer. European Journal of Cancer 27 (Suppl. 2): S105, 1991
Feld R, Wierzbicki R, Walde PLD, Shepherd FA, Evans WK, et al. Phase I-II study of high-dose epirubicin in advanced non-small-cell lung cancer. Journal of Clinical Oncology 10: 297–303, 1992
Fernandez-Giachella O, Gálvez C, Rufino C, Rufino A, Morera F, et al. Sequential versus alternating regimens in the treatment of advanced breast cancer. Tumori 75: 137–140, 1989
Ferrari P, Castagnetti G, Pollastri CA, Ferrari G, Tavoni F, et al. Chemoimmunotherapy for prophylaxis of recurrence in superficial bladder cancer: interferon-α2b versus interferon-α2b with epirubicin. Anti-Cancer Drugs 3 (Suppl. 1): 25–27, 1992
Ferrazzi E, Pappagallo GL, Nicoletto O, Fornasiero A, Refatti F, et al. Phase II evaluation of,4’-epi-doxorubicin in patients with metastatic colorectal carcinoma. Tumori 70: 297–300, 1984
Findlay M, Mansi JL, Ford HT, Nash AT, Cunningham D. Epirubicin, cisplatin and 5-fluorouracil (ECF) is highly effective in gastric cancer. European Journal of Cancer 27 (Suppl. 2): S71, 1991
Fisher B, Bauer M, Margolese R, Poisson R, Pilch Y, et al. Five-year results of a randomized clinical trial comparing total mastectomy and segmental mastectomy with or without radiation in the treatment of breast cancer. New England Journal of Medicine 312: 665–673, 1985
Fisher B, Redmond C, Wickerham DL, Bowman D, Schipper H, et al. Doxorubicin-containing regimens for the treatment of stage II breast cancer: the national surgical adjuvant breast and bowel project experience. Journal of Clinical Oncology 7: 572–582, 1989
Flechtner H, Queisser W, Heim ME, Henss H, Arnold H, et al. 5-Fluorouracil, 4-epidoxorubicin, and mitomycin C (FEM) combination chemotherapy for advanced gastric carcinoma. A phase-II trial by the ‘Chemotherapiegruppe Gastrointestinaler Tumoren (CGT)’. Onkologie 10: 67–71, 1987
Fountzilas G, Skarlos D, Pavlidis NA, Makrantonakis P, Tsavaris N, et al. High-dose epirubicin as a single agent in the treatment of patients with advanced breast cancer. Tumori 77: 232–236, 1991
Fossa SD, Wik B, Bae E, Lien HH. Phase II study of 4’-epi-doxorubicin in metastatic renal cancer. Cancer Treatment Reports 66: 1219–1221, 1982
Fossa S, Splinter T, Roozendaal KJ, Veenhof KHN, Pavone-Macaluso M, et al. A phase II study of 4-epi-adriamycin in advanced urothelial transitional cell cancer. European Journal of Cancer and Clinical Oncology 25: 389–390, 1989
Frassoldati A, Federico M, Barbieri F, Brausi M, Pollastri C, et al. Methotrexate, vinblastine, epidoxorubicin and cisplatin (MVEC) in patients with locally advanced transitional bladder cancer. Medical Oncology and Tumor Pharmacotherapy 8: 99–103, 1991
French Epirubicin Study Group. A prospective randomized phase III trial comparing combination chemotherapy with cyclophosphamide, fluorouracil, and either doxorubicin or epirubicin. Journal of Clinical Oncology 6: 679–688, 1988
French Epirubicin Study Group. A prospective randomized trial comparing epirubicin monochemotherapy to two fluorouracil, cyclophosphamide, and epirubicin regimens differing in epirubicin dose in advanced breast cancer patients. Journal of Clinical Oncology 9: 305–312, 1991
Fritze D, Harjung H, Katz R, Kober B. Ergebnisse der palliativen Chemotherapie bei fortgeschrittenem kleinzelligem bronchialkarzinom. Deutsche Medizinische Wochenschrift 43: 1663–1668, 1988
Fuith LC, Bazzanella A, Hetzel H. Combination chemotherapy with epirubicin and cisplatin in ovarian carcinoma. Archives of Gynecology and Obstetrics 244: 15–21, 1988
Fumoleau P, Berthaud P, Maugard-Louboutin C, Guillard Y, Cussac A. Optimal, dose schedule combining cyclophosphamide, epirubicin and cisplatin (CEP) in advanced ovarian carcinoma (ADOVCA) with residual tumor after first look laparotomy. Proceedings of the American Society of Clinical Oncology 11: 233, 1992
Fumoleau P, Devaux Y, Vo Van ML, Kerbrat P, Fargeot P, et al. Premenopausal patients with node positive resectable breast cancer — preliminary results of a randomised trial comparing three adjuvant regimens: FEC 50 × 6 cycles vs FEC 50 × 3 cycles vs FEC 75 × 3 cycles. Drugs (Suppl.), in press, 1993
Galvez CA, Bonamassa M, Pire P. 5-Fluorouracil (F) + 4’epiadriamycin (E) + cisplatin (C) vs cisplatin (C) + etoposide (E) + 5-fluorouracil (F) in patients with advanced gastric cancer. Preliminary report. European Journal of Cancer 27 (Suppl. 2): S79, 1991
Ganzina F, Di Pietro N, Magni O. Clinical toxicity of 4’-epi-doxorubicin (epirubicin). Tumori 71: 233–240, 1985
Gasmi J, Bachouchi M, Cvitkovic E, Boussen H, Azli N, et al. Nasopharyngeal carcinoma: a medical oncology viewpoint. The Gustave Roussy experience. Annals of Oncology 1: 245–253, 1990
Gasparini G, Dal Fior S, Panizzoni GA, Favretto S, Pozza F. Weekly epirubicin versus doxorubicin as second line therapy in advanced breast cancer. American Journal of Clinical Oncology 14: 38–44, 1991
Gastrointestinal Tumor Study Group. Phase II trials of the single agents baker’s antifol, diaziquone, and epirubicin in advanced pancreatic cancer. Cancer Treatment Reports 71: 865–867, 1987
Gennatas C, Dimas C, Dardoufas C. Low-dose weekly epirubicin as second-line chemotherapy in metastatic breast cancer. European Journal of Cancer 27 (Suppl. 2): S60, 1991
Gerard JP, Hery M, Gedouin D, Monnier A, Goudier MJ, et al. Postmenopausal patients with node positive resectable breast cancer: tamoxifen vs FEC 50 (6 cycles) vs FEC 50 (6 cycles) plus tamoxifen vs control — preliminary results of a 4-arm randomised trial. Drugs (Suppl.), in press, 1993
Gessner T, Robert J, Bolanowska W, Hoerni B, Durand M, et al. Effects of prior therapy on plasma levels of adriamycin during subsequent therapy. Journal of Medicine 12: 183–193, 1981
Ghavamzadeh A. Treatment of metastatic breast cancer with the combination of ifosfamide, epirubicin and 5-fluorouracil. Cancer Chemotherapy and Pharmacology 26 (Suppl.): S66–S68, 1990
Giaccone G, Donadio M, Bonardi G, Iberti V, Calciati A. 4’-epidoxorubicin in advanced lung cancer, Investigational New Drugs 8: 393–396, 1990
Giannakakis Th, Samelis G, Tsiryannis P, Loukatou M, Samanda D, et al. Efficacy and toxicity of 5-fluorouracil-epirubicin-cyclophosphamide (FEC) combination in metastatic breast cancer (MBC). Abstract no. 339. Annals of Oncology 3 (Suppl. 5): 87, 1992
GICOG (Gruppo Interegionale Cooperativo Oncologico Ginecologia). Randomised comparison of cisplatin with cyclophosphamide/cisplatin and with cyclophosphamide/doxorubicin/cisplatin in advanced ovarian cancer. Lancet 2: 353–359, 1987
Giglio R, Cabalar E, Mickiewicz E, Caccia G, Maniago O, et al. Phase II study of high dose epirubicin as single agent (HDESA) followed by radiotherapy (RT) in patients (pts) with locally advanced head and neck tumor (HNT). Proceedings of the American Society of Clinical Oncology 11: 245, 1992
Gindrey-Vie B, Genoa JY, Switsers O, Olivier JM, Rivière A, et al. Combination chemotherapy with navelbine, epirubicin and mitomycin C: a pilot study in advanced breast cancer (ABC). European Journal of Cancer 27 (Suppl. 2): S61, 1991
Giuliani FC, Coirin AK, Rice MR, Kaplan NO. Effect of 4’-doxorubicin analogs on heterotransplantation of human tumors in congenitally athymic mice. Cancer Treatment Reports 65: 1063–1075, 1981
Glisson BS, Ross WE. DNA topoisomerase II: a primer on the enzyme and its unique role as a multidrug target in cancer chemotherapy. Pharmacology and Therapeutics 32: 89–106, 1987
Goel R, Stewart DJ. Phase I study of epirubicin (EPI) plus biochemical modulators. Proceedings of the American Society of Clinical Oncology 11: 133, 1992
Gómez-Codina J, Anton A, Rosell R, Vadell C, Jimeno J, et al. Determining the optimal dose of epirubicin (EPI) when combined with cisplatin (CP) in patients (pts) with small cell lung cancer (SCLC). Proceedings of the American Society of Clinical Oncology 11: 302, 1992
Goncalves E, Chastang C, de Bock R, da Costa L, Eghbali H, et al. EBVVP: an intensive anthracycline/podophylotoxin containing chemotherapy. Feasibility as salvage treatment for Hodgkin’s disease. European Journal of Cancer 27 (Suppl. 2): S237, 1991
Goormaghtigh E, Vandenbranden M, Ruysschaert JM, De Kruijff B. Adriamycin inhibits the formation of non-bilayer lipid structures in cardiolipin-containing model membranes. Biochimica et Biophysica Acta 68: 137–143, 1982
Goormaghtigh E, Pollakis G, Ruysschaert JM. Mitochondrial membrane modifications induced by adriamycin-mediated electron transport. Biochemical Pharmacology 32: 889–893, 1983
Grankvist K, Stendahl U, Henriksson R. Comparative study of demethoxydaunorubicin with other anthracyclines on generation of oxygen radicals and clonogenic survival of fibroblasts. Pharmacology and Toxicology 65: 40–44, 1989
Greidanus J, Willemse PHB, Sleijfer DTH, Mulder NH, Verschueren RCJ, et al. Phase II study of a 21-day continuous infusion schedule with epirubicin in metastatic colorectal cancer. European Journal of Cancer and Clinical Oncology 24: 801–802, 1988
Gridelli C, Airoma G, Incoronato P, Pepe R, Palazzolo G, et al. Mitomycin C plus vindesine or cisplatin plus epirubicin in previously treated patients with symptomatic advanced non-smallcell lung cancer. Cancer Chemotherapy and Pharmacology 30: 212–214, 1992a
Gridelli C, Ianniello G, Maiorino A, Curcio C, D’Aprile M, et al. Carboplatin, epirubicin and VP-16 chemotherapy in the treatment of small cell lung cancer (SCLC). Proceedings of the American Society of Clinical Oncology 11: 301, 1992b
Gridelli C, Maiorino A, Curcio C, D’Aprile M, Ianniello G, et al. Carboplatin, epirubicin and VP-16 in the treatment of extensive small cell lung cancer (SCLC). European Journal of Cancer 27 (Suppl. 2): S185, 1991a
Gridelli C, Maiorino A, Curcio C, D’Aprile M, Ianniello G, et al. Carboplatin plus epirubicin plus VP-16, concurrent radiotherapy and adjuvant surgery for limited SCLC. European Journal of Cancer 27 (Suppl. 2): S186, 1991b
Guaraldi M, Martoni A, Rossi AP, Romagnoli D, Occhiuzzi L, et al. Treatment of advanced head and neck tumors with PEV-B regimen: a pilot study. Chemioterapia 7: 341–344, 1988
Guillem V, Camps C, Forner E, Munoz MA, Vicent J, et al. A feasibility study incorporating epirubicin (EPI) to M-VAC chemotherpay in patients (pt) with advanced bladder cancer (BC). Abstract no. 426. Annals of Oncology 3 (Suppl. 5): 110, 1992
Gundersen S, Kvinnsland S, Klepp O, Lund E, Host H. Weekly adriamycin® vs 4’-epidoxorubicin every second week in advanced breast cancer. A randomized trial. European Journal of Cancer 26: 45–48, 1990
Habeshaw T, Paul J, Jones R, Stallard S, Stewart M, Kaye SB, et al. Epirubicin at two dose levels with predisolone as treatment for advanced breast cancer: the results of a randomized trial. Journal of Clinical Oncology 9: 295–304, 1991
Hacker-Klom UB, Meistrich ML, Göhde W. Effect of doxorubicin and 4’-epi-doxorubicin on mouse spermatogenesis. Mutation Research 160: 39–46, 1986
Hall KS, Endresen L, Huitfeldt HS, Rugstad HE. Induction of in vitro resistance to 4’-epi-doxorubicin and cis-dichlorodiammineplatinum in hepatoma cells. Anticancer Research 11: 817–824, 1991
Hall KS, Endresen L, Rugstad HE. Verapamil potentiates 4’-epidoxorubicin cytotoxicity in a rat hepatoma cell line and in a substrain resistant to 4’-epidoxorubiein. Pharmacology and Toxicology 67: 402–405, 1990
Hansen HH, Rørth M. Lung Cancer. In Pinedo HM, et al. (Eds) Cancer chemotherapy and biological response modifiers annual. 10, Elsevier Science Publishers B.V, pp. 222–240, 1988
Harstrick A, Casper J, Köhne-Wömpner H, Wilke H, Schmoll H-J, et al. Comparative activity of four anthracyclines against heterotransplanted germ cell tumor lines. Investigational New Drugs 8 (Suppl): S19–S24, 1990a
Harstrick A, Schmoll H-J, Wilke H, Schöber C, Stahl M, et al. High dose epirubicin in refractory or relapsed non-seminomatous testicular cancer. A phase-II study. Annals of Oncology 1: 375–376, 1990b
Hasan I, Wickenhöfer R, Görich J, Hartlapp JH, Reiser M. Palliative Behandlung des primären Leberzellkarzinoms mit Chemoembolisation. Deutsche Medizinische Wochenschrift 117: 576–580, 1992
Hausmaninger H, Lehnert M, Steger G, Sevelda P, Michlmayr G, et al. Vindesine-epirubicin versus vindesine-mitoxantrone in metastatic breast cancer. Onkologie 12: 225–229, 1989
Hayward JL, Rubens RD, Carbone PP, Heuson J-C, Kumaoka S, et al. Assessment of response to therapy in advanced breast cancer. British Journal of Cancer 35: 292–298, 1977
Heidemann E, Steinke B, Hartlapp J, Schumacher K, Possinger K, et al. Randomized clincial trial comparing mitoxantrone with epirubicin and with doxorubicin, each combined with cyclophosphamide in the first-line treatment of patients with metastatic breast cancer. Onkologie 13: 24–27, 1990
Hemmer J. Cellular proliferation of oral carcinomas during chemotherapy assessed by DNA flow cytometry and bromodeoxyuridine labeling. Tumor Diagnostik und Therapie 12: 16–20, 1991
Hemmer J. Rapid in vitro bromodeoxyuridine labeling method for monitoring of therapy response in solid human tumors. Cytometry 11: 603–609, 1990
Hemmer J, Kreidler J, Thein T, Haase S. Monitoring of intra-arterial treatment with epirubicin and cisplatin in oral carcinoma by DNA flow cytometry. Chemotherapy 37: 150–156, 1991
Henriksson R, Grankvist K. Protective effect of iron chelators on epirubicin-induced fibroblast toxicity. Cancer Letters 43: 179–183, 1988
Hernádi Z, Juhász B, Póka R, Lampé LG. Randomised trial comparing combinations of cyclophosphamide and cisplatin without or with doxorubicin or 4’-epi-doxorubicin in the treatment of advanced ovarian cancer. International Journal of Gynecology and Obstetrics 27: 199–204, 1988
Hill BT, Whelan RDH. A comparison of the lethal and kinetic effects of doxorubicin and 4’-epi-doxorubicin in vitro. Tumori 68: 29–37, 1982
Hochster HS, Green MD, Speyer J, Fazzini E, Blum R, et al. 4’-Epidoxorubicin (Epirubicin): activity in hepatocellular carcinoma. Journal of Clinical Oncology 3: 1535–1540, 1985
Hochster H, Green MD, Speyer JL, Wernz JC, Blum RH, et al. Activity of epirubicin in pancreatic carcinoma. Cancer Treatment Reports 70: 299–300, 1986
Hoerni B, Orgerie MB, Eghbali H, Blanc CM, David B, et al. Nouvelle association d’epirubicine, bleomycine, vinblastine et prednisone (EBVP II) avant radiotherapie dans les stades localises de maladie de Hodgkin. Essai de phase II chez 50 malades. Bulletin du Cancer 75: 789–794, 1988
Hoffmann W, Weidmann B, Migeod F, Könner J, Seeber S. Epirubicin and ifosfamide in patients with refractory breast cancer and other metastatic solid tumours. Cancer Chemotherapy and Pharmacology 26 (Suppl.): S69–S70, 1990
Holdener EE, Hansen HH, Host H, Bruntsch U, Cavalli F, et al. Epirubicin in colorectal cancer. A phase II study of the early clinical trials group (EORTC). Investigational New Drugs 3: 63–66, 1985
Homesley HD, Harry DS, O’Toole RV, Hoogstraten B, Franklin EW, et al. Randomized comparison of cisplatin plus epirubicin or doxorubicin for advanced epithelial ovarian carcinoma. American Journal of Clinical Oncology 15: 129–134, 1992
Hortobagyi GN. Comprehensive management of loeally advanced breast cancer. Cancer 66: 1387–1391, 1990
Hortobagyi GN, Buzdar AU. Present status of anthracyclines in the adjuvant treatment of breast cancer. Drugs (Suppl.), in press, 1993
Hortobagyi GN, Yap H-Y, Kau SW, Fraschini G, Ewer MS, et al. A comparative study of doxorubicin and epirubicin in patients with metastatic breast cancer. American Journal of Clinical Oncology 12: 57–62, 1989
Hu OY-P, Chang S-P, Jame J-M, Chen K-Y. Pharmacokinetic and pharmacodynamic studies with 4’-epi-doxorubicin in nasopharyngeal carcinoma patients. Cancer Chemotherapy and Pharmacology 24: 332–337, 1989
Hubman R, Haider A. Cytostatic treatment of hormone-resistant prostatic cancer with 4-epirubicin (Farmorubicin). Aktuelle Urologie 21 (Suppl.): 18–21, 1990
Huovinen R, Nikkanen V. Low dose epidoxorubicin (LDEPI) as a second or third line chemotherapy in metastatic breast cancer (BC). European Journal of Cancer 27 (Suppl. 2): S64, 1991
Hurteloup P, Cappelaere P, Armand JP, Mathé G. Phase II clinical evaluation of 4’-epi-doxorubicin. Cancer Treatment Reports 67: 337–341, 1983
Ianniello GP, De Cataldis G, Cornelia G, Catalano G, Maiorino A, et al. Enhancement of C.E.V. combination (cisplatin, epirubicin, vindesine) in advanced NSCLC by lonidamine. Preliminary data of a phase II randomized trial. Abstract no. 118 Annals of Oncology 3 (Suppl. 5): 30, 1992
Iigo M, Nishikata K, Nakajima Y, Hoshi A. Effects of anthracycline derivatives on hepatic neoplastic nodules of Lewis lung carcinoma and colon adenocarcinoma 26. British Journal of Cancer 63: 363–366, 1991
Italia C, Paglia L, Trabattoni A, Luchini S, Villas F, et al. Distribution of 4’-epi-doxorubicin in human tissues. British Journal of Cancer 47: 545–547, 1983
Italian Multicentre Breast Study with Epirubicin. Phase III randomized study of fluorouracil, epirubicin, and cyclophosphamide v fluorouracil, doxorubicin, and cyclophosphamide in advanced breast cancer: an Italian Multicentre Trial. Journal of Clinical Oncology 6: 976–982, 1988
Itoh T, Sakata Y, Tamura Y, Furukohori N, Saitoh S, et al. Effect of combination of 5-fluorouracil, epirubicin and mitomycin C (FEM) on advanced gastric carcinoma. Nippon Gan Chiryo Gakkai-Shi 24: 1551–1556, 1989
Izzo J, Chadjaa M, May-Levin F, Riggi M, Armand JP, et al. Preliminary data on 4’epiadriamycine (EPI)- vinorelbine (VNB): a new active combination in advanced breast cancer (ABC). Abstract no. 319. Annals of Oncology 3 (Suppl. 5): 82, 1992
Jain KK, Casper ES, Geller NL, Hakes TB, Kaufman RJ, et al. A prospective randomized comparison of epirubicin and doxorubicin in patients with advanced breast cancer. Journal of Clinical Oncology 3: 818–826, 1985
Jakobsen P, Steiness E, Bastholt L, Dalmark M, Lorenzen A, et al. Multiple-dose pharmacokinetics of epirubicin at four different dose levels: studies in patients with metastatic breast cancer. Cancer Chemotherapy and Pharmacology 28: 63–68, 1991
Jassem J, Karnicka-Mlodkowska H, Drozd-Lula M, Strug A, Pilarska-Machowicz A, et al. Combination chemotherapy with vincristine, epirubicin and cyclophosphamide in small cell lung carcinoma. European Journal of Cancer 28: 473–476, 1992
Jelic S, Vuletic L, Milanovic N, Tomasevic Z, Kovcin V. Highdose epirubicin-cisplatin chemotherapy for advanced soft tissue sarcoma. Tumori 76: 467–471, 1990
Johnson DH. Primary lung cancer. In Rakel RE (Ed.) Latest approved methods of treatment for the practicing physician pp. 150–155, WB Saunders Company, Philadelphia, 1992
Jones WG. Mattsson W. Phase II study of weekly low-dose 4’-epidoxorubicin in advanced postmenopausal breast cancer. Cancer Treatment Reports 68: 675–677, 1984
Jones SE, Mennel RG, Peters GN, Westrick MA, Brooks BD, et al. Phase I trial of adjuvant chemotherapy with cyclophosphamide, epirubicin and 5-fluorouracil (CEF) for stage II breast cancer. Investigational New Drugs 6: 195–198, 1988
Jones WG, Fossa SD, Bono AY, Klijn JGM, De Pauw M, et al. European organization for research and treatment of cancer (EORTC) phase II study of low-dose weekly epirubicin in metastatic prostate cancer. Cancer Treatment Reports 71: 1317–1318, 1987
Jones WG. Effective palliation of advanced breast cancer with weekly low dose epirubicin. European Journal of Cancer and Clinical Oncology 25: 357–360, 1989
Joss RA, Hansen HH, Hansen M, Renards J, Rozencweig M. Phase II trial of epirubicin in advanced squamous, adeno- and large cell carcinoma of the lung. European Journal of Cancer and Clinical Oncology 20: 495–499, 1984
Jovanovic DM, Muzikravic Lj, Stosic J, Bojanovic, Jovanovic DV, et al. 4-epi-doxorubicin and high doses of 5-fluorouracil in treatment of advanced gastric cancer. Abstract no. 56. Annals of Oncology 3 (Suppl. 5): 15, 1992
Jungi WF, Morant R, Thürlimann B, de Stoutz N, Ries G, et al. VECOP-B-RT: highly effective chemo-radiothefapy for aggressive non Hodgkin’s lymphoma. Proceedings of the American Society of Clinical Oncology 11: 322, 1992
Kajanti MJ, Pyrhönen SO. Phase II intravenous study of epirubicin with 5-fluorouracil in patients with advanced hepatocellular carcinoma. European Journal of Cancer 27: 1620–1622, 1991
Kajanti M, Pyrhönen S, Mäntylä M, Rissanen P. Intra-arterial and intravenous use of 4’-epidoxorubicin combined with 5-fluorouracil in primary hepatocellular carcinoma. American Journal of Clinical Oncology 15: 37–40, 1992
Kánitz É, Kolaric K, Jassem J, Mechl Z, Pawlicki M, et al. Randomized phase II trial of high-dose 4’-epi-doxorubicin + cyclophosphamide versus high-dose 4’-epi-doxorubicin + cisplatin in previously untreated patients with extensive small cell lung cancer. Oncology 49: 327–332, 1992
Karakiulakis G, Missirlis E, Maragoudakis ME. Basement membrane collagen-degrading activity from a malignant tumor is inhibited by anthracycline antibiotics. Biochimica et Biophysica Acta 1035: 218–222, 1990
Kaye S, Merry S. Tumour cell resistance to anthracyclines. A review. Cancer Chemotherapy and Pharmacology 14: 96–103, 1985
Kimura K, Suga S, Kano H, Sakakibara K, Hayashi T, et al. Phase II study of epirubicin on gastric cancer. A cooperative study of the Tokai Cancer Chemotherapy Group. Japanese Journal of Cancer Chemotherapy 13: 2440–2445, 1986
Klastersky J. Therapy of small cell lung cancer: anything new? European Journal of Clinical Oncology 24: 107–112, 1988
Klein LA. Prostatic carcinoma. New England Journal of Medicine 300: 824–833, 1979
Klugmann FB, Decorti G, Candussio L, Mallardi F, Grill V, et al. Amelioration of 4’-epidoxorubicin-induced cardiotoxicity by sodium cromoglycate. European Journal of Cancer Clinical Oncology 25: 361–368, 1989
Knuchel R, Hofstadter F, Jenkins WEA, Masters JRW. Sensitivities of monolayers and spheroids of the human bladder cancer cell line MGH-U1 to the drugs used for intravesical chemotherapy. Cancer Research 49: 1397–1401, 1989
Kohnoe S, Yoshida M, Takahashi I, Emi Y, Maehara Y, et al. Epirubicin is equivalent to adriamycin in vitro against many cancer cells but more effective against gastric cancer cells. Anticancer Research 12: 389–392, 1992
Kolaric K, Roth A, Ban J, Bistrovic M, Dujmovic I. Combination of 4’-epi-doxorubicin and irradiation — a new approach in the treatment of locoregionally advanced inoperable esophageal cancer. Tumori 72: 89–94, 1986a
Kolaric K, Potrebica V, Stanovnik M. Controlled phase III clinical study of 4’-epi-doxorubicin + 5-fluorouracil versus 5-fluorouracil alone in metastatic gastric and rectosigmoid cancer. Oncology 43: 73–77, 1986b
Kontturi M, Sotarauta M, Tammela T, Lukkarinen O, Romppainen M-L, et al. Sequentially alternating hormone chemotherapy with high-dose medroxy-progesterone acetate and low-dose epirubicin for the treatment of hormone-resistant metastatic prostatic cancer. European Urology 15: 43–47, 1988
Kornek G, Depisch D, Schulz F, Pidlich P, Rosen H, Sebesta C, et al. Phase II trial of epirubicin (EPI), fluorouracil (FU) and leucovorin (LV) in advanced metastatic adenocarcinoma of the stomach. Onkologie 14 (Suppl. 2): 89, 1991
Kosmidis PA, Fountzilas G, Tsavaris N, Kalogera A, Karabelis A, et al. Alternative administration of epirubicin, vincristine and mitoxantrone, vincristine in advanced breast cancer. Journal of Experimental and Clinical Cancer Research 9: 215–219, 1990
Kretzschmar A, Drings P. Epirubicin weekly in combination chemotherapy with cyclophosphamide and vincristin in untreated small cell lung cancer: a phase II trial. Onkologie 13: 141–142, 1990
Kurth KH, Mross K. ten Kate F, Weissglas G, Carpentier PJ, et al. Phase I/II study of intravesical epirubicin in patients with carcinoma in situ of the bladder. In Uro-oncplogy: current status and future trends, pp. 41–59, Wiley-Liss, 1990
Kurth K, v.d. Vijgh WJF, ten Kate F, Bogdanowicz JF, Carpentier PJ, et al. Phase 1/2 study of intravesical epirubicin in patients with carcinoma in situ of the bladder. The Journal of Urology 146: 1508–1513, 1991
Lahtinen R, Kuikka J, Nousiainen T, Uusitupa M, Länsimies E. Cardiotoxicity of epirubicin and doxorubicin. A double-blind randomized study. European Journal of Haematology 46: 301–305, 1991
Lange OF, Scheef W, Haase KD, Heckmann M, Leyendecker R, et al. Palliative chemo-radiotherapy with ifosfamide and epirubicin as first-line treatment for high-risk metastatic breast cancer. Cancer Chemotherapy and Pharmacology 26 (Suppl.): S74–S77, 1990
Lanza E, Rozza A, Favalli L, Monti E, Poggi P, et al. The rat model in the comparative evaluation of anthracyclines cardiotoxicity. Tumori 75: 533–536, 1989
Laparidou S, Skarlos DV, Polizogopoulos D, Barlas K, Samantas E, et al. Combination chemotherapy of small cell lung cancer (SCLC) with epirubicin (EP), cyclophosphamide (C), etoposide (E) and prophylactic cranial irradiation (PCI). European Journal of Cancer 27 (Suppl. 2): S218, 1991
Latreille J, Blackstein M, Goss P, Guevin R, Sutherland D, et al. Preliminary results of a phase III trial of CMF vs CEF in metastatic breast cancer. A multicentre study. European Journal of Cancer 27 (Suppl. 2): S57, 1991
Lawton PA, Ostrowski MJ, Young T, Spittle MF. Efficacy and toxicity of single agent chemotherapy in advanced breast carcinoma. British Journal of Cancer 61: 177, 1990
Lazzarino G, Viola AR, Mulieri L, Rotilio G, Mavelli I. Prevention by fructose-1,6-bisphosphate of cardiac oxidative damage induced in mice by subchronic doxorubicin treatment. Cancer Research 47: 6511–6516, 1987
Le Bot MA, Bégué JM, Kernaleguen D, Robert J, Ratanasavanh D, et al. Different cytotoxicity and metabolism of doxorubicin, daunorubicin, epirubicin, esorubicin and idarubicin in cultured human and rat hepatocytes. Biochemical Pharmacology 37: 3877–3887, 1988
Lee YC, Byfield JE, Bennett LR, Chan PYM. X-Ray repair replication in L1210 leukemia cells. Cancer Research 34: 2624–2633, 1974
Lefrak EA, Pitha J, Rosenheim S, Gottlieb JA. A clinicopathologic analysis of adriamycin cardiotoxicity. Cancer 32: 302–314, 1973
Lelli G, Melotti B, Martoni A, Rossi AP, Maver P, et al. Cisplatin (P), epirubicin (E) and methotrexate (M) in advanced transitional cell cancer (TCC): a phase II trial. European Journal of Cancer 27 (Suppl. 2): S102, 1991
Lelli G, Melotti B, Martoni A, Strocchi E, Pannuti F, et al. Neoadjuvant cisplatin (P), epirubicin (E) and methotrexate (M) in invasive bladder cancer. A dose intensity and dose response study. Proceedings of the American Society of Clinical Oncology 11: 209, 1992b
Lelli G, Melotti B, Pannuti F, Rossi AP, Maver P, et al. Chemotherapy with cisplatin, epirubicin, methotrexate in the treatment of locally advanced or metastatic transitional cell cancer of the bladder (TCC). Journal of Chemotherapy 4: 239–243, 1992a
Lelli G, Tononi A, Bacchetti P, Maver P, Corrado G, et al. Low dose intermittent 5-fluorouracil, epirubicin and cyclophosphamide (FEC) in poor risk patients with advanced transitional cell bladder cancer: a pilot study. Journal of Chemotherapy 1: 269–271, 1989
Leung WT, Shiu WCT, Leung N, Chan M, Tao M, et al. Treatment of inoperable hepatocellular carcinoma by intra-arterial lipiodol and 4’epidoxorubicin. Cancer Chemotherapy and Pharmacology 29: 401–404, 1992
Levine MN, Bramwell V, Pritchard K, Perrault D, Findlay B, et al. A pilot study of intensive cyclophosphamide epirubicin and fluorouracil in patients with axillary node positive or locally advanced breast cancer. European Journal of Cancer 29A: 37–43, 1993a
Levine MN, Bramwell V, Pritchard K, Perrault D, Findlay B, et al. A pilot study of intensive fluorouracil, epirubicin and cyclophosphamide (FEC) in patients with axillary node-positive or locally advanced breast cancer. Drugs (Suppl.), in press, 1993b
Liang D-C, Jiang C-J. Epirubicin and cytosine arabinoside for the induction therapy of childhood acute nonlymphocytic leukemia. American Journal of Hematology 38: 267–270, 1991
Lind MJ, Cantwell B, Millward MJ, Gumbrell L, Procter M, et al. The use of HGCSF to accelerate the delivery of ifosfamide and epirubicin in women with locally advanced or metastatic carcinoma of the breast. British Journal of Cancer 65 (Suppl. 16): 32, 1992
Lira-Puerto V, Zepeda G, Silva A, Martinez R, Palma F. 5FU, adriamycin and cytoxan (FAC) vs 5 FU, epirubicin and cytoxan (FEC) as neoadjuvant chemotherapy in stage III breast cancer. Proceedings of the American Society of Clinical Oncology 11: 76, 1992
Lira-Puerto V, Zepeda G, Silva A, Palma F, Martinez R. 5FU, adriamycin and cytoxan (FAC) vs 5FU, epirubicin and cytoxan (FEC) — and radiotherapy in stage III breast cancer. European Journal of Cancer 27 (Suppl. 2): S61, 1991
Liso V, Specia G, Pavone V, Capalbo S, Dione R. Continuous infusion chemotherapy with epirubicin and vincristine in relapsed and refractory actue leukemia. Acta Haematologica 83: 116–119, 1990
Lissoni P, Barni S, Rovelli F, Tancini G. Lower survival in metastatic cancer patients with reduced interleukin-2 blood concentrations. Preliminary report. Oncology 48: 125–127, 1991
Lissoni P, Tancini G, Archili C, Rescaldani R, Cattaneo G, et al. Changes in T lymphocyte subsets after single dose epirubicin. European Journal of Cancer 26: 767–768, 1990
Llesuy SF, Milei J, Gonzalez Flecha BS, Boveris A. Myocardial damage induced by doxorubicins: hydroperoxide-initiated chemiluminescence and morphology. Free Radical Biology and Medicine 8: 259–264, 1990
Llesuy SF, Milei J, Molina H, Boveris A, Milei S. Comparison of lipid peroxidation and myocardial damage induced by adriamycin and 4’-epiadriamycin in mice. Tumori 71: 241–249, 1985
Lollini P-L, De Giovanni C, Del Re B, Landuzzi L, Nicoletti G, et al. Myogenic differentiation of human rhabdomyosarcoma cells induced in vitro by antineoplastic drugs. Cancer Research 49: 3631–3636, 1989
Lopez M, Carpano S, Di Lauro L, Vici P, Conti EMS. Epirubicin and DTIC (EDIC) for advanced soft-tissue sarcomas. Oncology 48: 230–233, 1991
Lopez M, Natali M, Di Lauro L, Tonini G, Carpano S, et al. 5-Fluorouracil, epirubicin, and BCNU (FEB) in advanced measurable gastric cancer. American Journal of Clinical Oncology 13: 204–207, 1990
Lopez M, Papaldo P, Di Lauro L, Vici P, Carpano S, et al. 5-Fluorouracil, adriamycin, cyclophosphamide (FAC) vs. 5-fluorouracil, epirubicin, cyclophosphamide (FEC) in metastatic breast cancer. Oncology 46: 1–5, 1989
Lopez M, Perno C-F, Papaldo P, Di Lauro L, Ganzina F, et al. Phase II study of epirubicin in advanced malignant melanoma. Investigational New Drugs 2: 315–317, 1984
Lo Re G, Veronesi A, Dal Bo V, Tumolo S, Talamini R, et al. Combination chemotherapy in non-elderly, patients with hormone refractory prostatic carcinoma. Proceedings of the American Society of Clinical Oncology 11: 211, 1992
Lorusso V, Berardi F, Catino A, De Lera M. Methotrexate, vinblastin, epodoxorubicin and carboplatin (M-VECA) in the treatment of advanced bladder cancer. Proceedings of the American Society of Clinical Oncology 11: 212, 1992
Lown JW, Sim S-K, Majumdar KC, Chang R-Y. Strand scission of DNA by bound adriamycin and daunorubicin in the presence of reducing agents. Biochemical and Biophysical Research Communications 76: 705–710, 1977
Luporini A, Barni S, Verusio C, Arnoldi E, Frontini L, et al. Evaluation of EEP polychemotherapy in advanced gastric and pancreatic carcinomas (GPC). European Journal of Cancer 27 (Suppl. 2): S75, 1991
Macchiarini P, Chella A, Ducci F, Rossi B, Testi C, et al. Neoadjuvant chemotherapy, surgery, and postoperative radiation therapy for invasive thymoma. Cancer 68: 706–713, 1991a
Macchiarini P, Chella A, Riva A, Mengozzi G, Silvano G, et al. Phase II feasibility study of high dose epirubicin-based regimens for untreated patients with small-cell lung cancer. American Journal of Clinical Oncology 13: 495–500, 1990a
Macchiarini P, Danesi R, Hardin M, Del Tacca M, Angeletti CA. Low-toxic combination chemotherapy for limited small-cell lung cancer. Current Therapeutic Research 46: 1203–1212, 1989a
Macchiarini P, Danesi R, Mariotti R, Marchetti A, Fazzi P, et al. Phase II study of high-dose epirubicin in untreated patients with small-cell lung cancer. American Journal of Clinical Oncology 13: 302–307, 1990b
Macchiarini P, Hardin M, Basolo F, Bruno J, Angeletti CA. Adjuvant chemotherapy for T1-2NOMO small cell lung cancer: single-agent or combination chemotherapy? Cancer Investigation 9: 19–25, 1991b
Macchiarini P, Hardin M, Basolo F, Bruno J, Chella A, et al. Surgery plus adjuvant chemotherapy for T1-3NOMO small-cell lung cancer. American Journal of Clinical Oncology 14: 218–224, 1991c
Macchiarini P, Mussi A, Basolo F, Bruno J, Angeletti CA. Optimal treatment for T1-3NOMO small cell lung cancer: surgery plus adjuvant chemotherapy. Anticancer Research 9: 1623–1626, 1989b
Macdonald JS, Schein PS, Woolley PV, Smythe T, Ueno W. 5-Fluorouracil, doxorubicin, and mitomycin (FAM) combination chemotherapy for advanced gastric cancer. Annals of Internal Medicine 93: 533–536, 1980
MacNeil HF, Hall RR, Neal DE, Roberts JT. Systemic chemotherapy for urothelial cancer in patients with ureteric obstruction. British Journal of Urology 67: 169–172, 1991
Magri MD, Foladore S, Veronesi A, Serra C, Nicotra M, et al. Treatment of malignant mesothelioma with epirubicin and ifosfamide: a phase II cooperative study. Annals of Oncology 3: 237–238, 1992
Magri MD, Veronesi A, Foladore S, De Giovanni D, Serra C, et al. Epirubicin in the treatment of malignant mesothelioma: a phase II cooperative study. Tumori 77: 49–51, 1991
Mahjoubi R, Azil N, Bachouchi M, Soulié P, Wibault P, et al. Metastatic (MTS) undifferentiated carcinoma of nasopharyngeal type (UCNT) treated with bleomycin (B), epirubicin (E) and cisplatin (C) (BEC) final report. Proceedings of the American Society of Clinical Oncology 11: 240, 1992
Mahjoubi M, Rougler Ph, Droz JP, Cvitkovic E, Tigaud JM, et al. Phase II trial with 5 FU lederfoline epirubicine and cisplatinum (FLEP) in advanced gastric adenocarcinoma (AGC): Proceedings of the American Association for Cancer Research 32: 185, 1991
Mangioni C, Bolis G, Pecorelli S, Bragman K, Epis A, et al. Randomized trial in advanced ovarian cancer comparing cisplatin and carboplatin. Journal of the National Cancer Institute 81: 1464–1471, 1989
Mansi JL, Smith IE, Selby PJ, Cunningham D. VEEP: a new chemotherapy regimen for Hodgkin’s disease. British Journal of Cancer 63 (Suppl. 8): 26, 1991
Marangolo M, Tienghi A, Fiorentini G, Dazzi C, Graziani G, et al. Treatment of pelvic osteosarcoma. Annals of Oncology 3 (Suppl. 2): S19–S21, 1992
Marschner NW, Kreienberg R, Schütte, Ruffert K, Nagel GA. Doseintensity in the treatment of patients with metastatic breast cancer — high risk subtype. Abstract, p. 13, Deutsch-Italienisches Onkologisches Symposium, Villasimius, May 10, 1991.
Marschner N, Nagel GA, Beyer J-H, Adler M, Ammon A. High-dose epirubicin in combination with cyclophosphamide (HDEC) in advanced breast cancer: final results of a dose finding study and phase II trial. Onkologie 13: 272–278, 1990
Martini A, Moro E, Pacciarini MA, Tamassia V, Natale N, et al. Cross-over study of pharmacokinetics and haematological toxicity of 4’-epi-doxorubicin and doxorubicin in cancer patients. International Journal of Clinical Pharmacology Research IV: 231–238, 1984
Martoni A, Bellucco A, Canova N, Pannuti F. Four-year analysis of platinum and anthracycline combination for ovarian cancer. Oncology 46: 109–116, 1989
Martoni A, Ercolino L, Bellucco A, Canova N, Lelli G, et al. Cisplatinum plus epirubicin alternated with cyclophosphamide plus 5-fluorouracil in ovarian cancer. Journal of Chemotherapy 2: 62–66, 1990
Martoni A, Guaraldi M, Casadio M, Busutti L, Pannuti F. A phase II study of high-dose epirubicin plus cisplatinum in advanced non-small-cell lung cancer (NSCLC). Annals of Oncology 3: 864–866, 1992
Martoni A, Melotti B, Guaraldi M, Pannuti F. Activity of high-dose epirubicin in advanced non small cell lung cancer. European Journal of Cancer 27: 1231–1234, 1991
Martoni A, Tomasi L, Farabegoli G, Fruet F, Pannuti F. 4’-Epidoxorubicin in combination with cisplatin in advanced ovarian cancer. Cancer Treatment Reports 68: 1391–1393, 1984
Marty M. FEC vs CMF: the ICCG experience. In Anthracyclines in the adjuvant treatment of early breast cancer, pp. 13–17, Adis International, Chester, 1992
Mason RC, Derodra J, Hale P, Harper P, Highley M, et al. Significant palliation of advanced gastric/oesophageal adenocarcinoma (G-O/C) with laser endoscopy and combination chemotheray. Gut 33 (Suppl. 1): S33, 1992
Mason RC, Highley M, Bright N, Hill M, Barker S, et al. Treatment of advanced adenocarcinoma of the oesophagus and stomach with epirubicin, cisplatin, and continuous 5-fluorouracil. Gut 32: A1211, 1991
Matsumura Y, Tsushima T, Ozaki Y, Yoshimoto J, Akagi T, et al. Intravesical chemotherapy with 4’-epi-adriamycin in patients with superficial bladder tumors. Cancer Chemotherapy and Pharmacology 16: 176–177, 1986
Mattson K, Giaccone G, Kirkpatrick A, Evrard D, Tammilehto L, et al. Epirubicin in malignant mesothelioma: a phase II study of the European organization for research and treatment of cancer. Lung Cancer Cooperation Group. Journal of Clinical Oncology 10: 824–828, 1992
Matumoto S, Aikawa H, Mori H, Suzuki K, Wakisaka M, et al. Hepatic arterial injection therapy with farmorubicin-PC-lipiodol suspension (FPLS) for hepatocellular carcinoma (HCC). Nippon Gan Chiryo Gakkai-Shi 26: 360, 1991
Mauriac L, Durand M, Chauvergne J, Dilhuydy J-M, Bonichon F. Randomized trial of adjuvant chemotherapy for operable breast cancer comparing i.v. CMF to an epirubicin-containing regimen. Annals of Oncology 3: 439–443, 1992
Mayhew E, Martin FJ, Murray M, Babbar S, Lasic D. In vivo efficacy of doxorubicin or epirubicin entrapped in long circulating liposomes against mouse colon tumor C26. Proceedings of the American Association for Cancer Research 33: 445, 1992
Mazzoni A, Trave F, Russo P, Nicolin A, Rustum YM. Generation and characterization of a low-degree drug-resistant human tumor cell line. Oncology 47: 488–494, 1990
McGuire WP, Rowinsky EK, Rosenhein NB, Grumbine FC, Ettinger DS, et al. Taxol: a unique antineoplastic agent with significant activity in advanced ovarian epithelial neoplasms. Annals of Internal Medicine 111: 273–279, 1989
Melekos MD, Dauaher H, Fokaefs E, Barbalias G. Intravesical instillations of 4’-epi-doxorubicin (epirubicin) in the prophylactic treatment of superficial bladder cancer: results of a controlled prospective study. Journal of Urology 147: 371–375, 1992
Mengozzi G, Palagi C, Petronio AS, Balbarini A, Mariotti R, et al. Evaluation of high-dose 4’-epidoxorubicin cardiotoxicity. Cardiologia 36: 137–142, 1991
Meyers FJ, Cardiff RD, Quadro R, Gribble M, Kohler M, et al. Epirubicin in non-oat cell lung cancer — response rates and the importance of immunopathology: a northern California oncology group study. Cancer Treatment Reports 70: 805–806, 1986
Meyers FJ, Lewis B, Mitchell E, William L, Hannigan Jr JF, et al. Phase II trial of 4’-epi-doxorubicin in advanced Colorectal carcinoma: a Northern California Oncology Group Study. Cancer Treatment Reports 69: 143–144, 1985
Michaelson R, Kemeny N, Young C. Phase II evaluation of 4’-epi-doxorubicin in patients with advanced colorectal carcinoma. Cancer Treatment Reports 66: 1757–1758, 1982
Mimnaugh EG, Trush MA, Bhatnagar M, Gram TE. Enhancement of reactive oxygen-dependent mitochondrial membrane lipid peroxidation by the anticancer drug adriamycin. Biochemical Pharmacology 34: 847–856, 1985
Minow RA, Benjamin RS, Gottlieb JA. Adriamycin (NSC-123127) cardiomyopathy — an overview with determination of risk factors. Cancer Chemotherapy Reports 6: 195–201, 1975
Mitrou PS, Klippstein T, Lautenschläger G, Burckhardt KL. Etoposide and epirubicin containing chemotherapy plus radiotherapy in the treatment of early stages of Hodgkin’s disease (HD). Proceedings of the American Society of Clinical Oncology 11: 328, 1992
Molinaro P, Lafleur F, Blum RH. A phase III randomized trial of epirubicin versus 5-fluorouracil in metastatic rectal/sigmoid adenocarcinoma. American Journal of Clinical Oncology 12: 332–334, 1989
Monti E, Piccinini F, Favalli L, Villani F. Role of the fast-exchanging calcium compartment in the early cardiotoxicity of anthracycline analogs. Biochemical Pharmacology 32: 3303–3306, 1983
Morris RG, Kotasek D, Paltridge G. Disposition of epirubicin and metabolites with repeated courses to cancer patients. European Journal of Clinical Pharmacology 40: 481–487, 1991
Mouridsen HT, Alfthan C, Bastholt L, Bergh J, Dalmark M et al. Current status of epirubicin (farmorubicin) in the treatment of solid tumours. Acta Oncologica 29: 257–285, 1990
Mouridsen HT, Bastholt L, Somers R, Santoro A, Bramwell V, et al. Adriamycin versus epirubicin in advanced soft tissue sarcomas. A randomized phase II/phase III study of the EORTC soft tissue and bone sarcoma group. European Journal of Cancer and Clinical Oncology 23: 1477–1483, 1987
Mross K, Maessen P, van der Vijgh WJF, Bogdanowicz JF, Kurth KH, et al. Absorption of epi-doxorubicin after intravesical administration in patients with in situ transitional cell carcinoma of the bladder. European Journal of Cancer Clinical Oncology 23: 505–508, 1987
Mross K, Maessen P, van der Vijgh WJF, Gall H, Boven E, et al. Pharmacokinetics and metabolism of epidoxorubicin and doxorubicin in humans. Journal of Clinical Oncology 6: 517–526, 1988
Muggia FM, Carter SK. Malignant Diseases. In Speight TM (Ed.) Avery’s drug treatment, 3rd ed., pp. 1023–1077, Adis International, Auckland, 1987
Muhammed H, Ramasarma T, Kurup CKR. Inhibition of mitochondrial oxidative phosphorylation by adriamycin. Biochimica et Biophysica Acta 722: 43–50, 1982
Nagasue N, Yukaya H, Okamura J, Kuroda C, Kubo Y, et al. Intra-arterial administration of epirubicin in the treatment of non-resectable hepatocellular carcinoma. Japanese Journal of Cancer Chemotherapy 13: 2786–2792, 1986
Nagel GA, Beyer J-H, Holtkamp W, Emrich D, Kneba M, et al. High-dose epirubicin + cyclophosphamide (HD-EC) in metastatic breast cancer: a dose-finding study. Onkologie 11: 287–288, 1988
Nara N, Yamashita Y, Murohashi I, Tanikawa S, Imai S, et al. Effects on leukemic clonogenic cells in murine myeloid leukemia of 1-β-D-arabinofuranosylcytosine and the anthracyclines adriamycin, daunomycin, aclacinomycin A, and 4’-epi-doxorubicin. Cancer Research 47: 2376–2379, 1987
Negro A, Morgenfeld E, Rivarola E, Pasccon G, Marantz A, et al. Neoadjuvant chemotherapy (NCT) for locally advanced breast cancer (LABC): clinical response and histopathological changes (HPC) after chemotherapy (CT) with epirubicin combination. Proceedings of the American Society of Clinical Oncology 11: 64, 1992
Neijt JP, ten Bokkel Huinink WW, van der Burg MEL, van Oosterom AT, Willemse PHB, et al. Randomized trial comparing two combination chemotherapy regimens (CHAP-5 v CP) in advanced ovarian carcinoma. Journal of Clinical Oncology 5: 1157–1168, 1987
Nelson RS, de Elizalde R, Howe CD. Clinical aspects of primary carcinoma of the liver. Cancer 19: 533–537, 1966
Nemoto T, Vana J, Bedwani RN, Baker HW, McGregor FH, et al. Management and survival of female breast cancer: results of a national survey by the American College of Surgeons. Cancer 45: 2917–2924, 1980
Neri B, Cini-Neri G, Bandinelli M, Pacini P, Bartalucci S, et al. Doxorubicin and epirubicin cardiotoxicity: experimental and clinical aspects. International Journal of Clinical Pharmacology, Therapy and Toxicology 27: 217–221, 1989
Neri B, Pacini P, Bartalucci S, Moroni F, Menchi I, et al. Epirubicin high-dose therapy in advanced breast cancer: preliminary clinical data. Epirubicin as a single agent in breast cancer. International Journal of Clinical Pharmacology, Therapy and Toxicology 27: 388–391, 1989
Neri B, Cini-Neri G, D’Alterio M. Effect of anthracyclines and mitoxantrone on oxygen uptake and ATP intracellular concentration in rat heart slices. Biochemical and Biophysical Research Communications 125: 954–960, 1984
Neri B, Lottini G, Bruno S, Fabbroni S, Leone V, et al. Epidoxorubicin (EPI) plus high-dose folinic acid (FA) and 5-fluorouracil (5-FU) in advanced gastric cancer. European Journal of Cancer 27 (Suppl. 2): S79, 1991b
Neri GC, Neri B, Bandinelli M, Del Tacca M, Danesi R, et al. Anthracycline cardiotoxicity: in vivo and in vitro effects on biochemical parameters and heart ultrastructure of the rat. Oncology 48: 327–333, 1991a
Nersessian AK, Zilfian VN, Koumkoumadjian VA. Inhibitory effect of rat immunization with tularemia vaccine on the in vivo clastogenicity of 4 anthracycline antibiotics. Mutation Research 260: 215–218, 1991
Newling D. Intravesical therapy in the management of superficial transitional cell carcinoma of the bladder: the experience of the EORTC GU Group. British Journal of Cancer 61: 497–499, 1990
Nicolay K, Sautereau A-M, Tocanne J-F, Brasseur R, Huart P, et al. A comparative model membrane study on structural effects of membrane-active positively charged anti-tumor drugs. Biochimica et Biophysica Acta 940: 197–208, 1988
Nielsen D, Dombernowsky P, Skovsgaard T, Jensen J, Andersen E, et al. Epirubicin or epirubicin and vindesine in advanced breast cancer. A phase-Ill study. Annals of Oncology 1: 275–280, 1990a
Nielsen D, Hansen OP, Dombernowsky. Epirubicin (E) cardiac toxicity in patients with advanced breast cancer. Abstract no. 437. Annals of Oncology 3 (Suppl. 5): 114, 1992
Nielsen D, Jensen JB, Dombernowsky P, Munck O, Fogh J, et al. Epirubicin cardiotoxicity: a study of 135 patients with advanced breast cancer. Journal of Clinical Oncology 8: 1806–1810, 1990b
Niijima T. Cooperative phase II study of epirubicin (EPI) for bladder cancer, renal pelvic and ureteral tumors. Hinyokika Kiyo 32: 1359–1369, 1986
Niiranen A, Kajanti M, Mattson K, Pyrhönen S. Epirubicin in patients with inoperable non-small cell lung cancer — a phase II study. Acta Oncologica 30: 869–870, 1991
Nikkanen TAV, Jakobsson M, Järvinen M, Liippo K, Ojala A, et al. Doxorubicin and 4’-epi-doxorubicin in combination chemotherapy of extensive small cell lung cancer. Acta Oncologica 27: 75–76, 1988
Nobile MT, Vidili MG, Sertoli MR, Venturini M, Simoni G, et al. Intrahepatic epidoxirubicin in metastatic and primary liver tumours: a phase II study. Anticancer Research 11: 861–864, 1991
O’Brien MER, Smith IE, Jones AL, Baum M, Sacks N, et al. Long term continuous infusion 5-FU (F) with epirubicin (E) and cisplatin (P) in the treatment of breast cancer — a very active new regimen. British Journal of Cancer 66 (Suppl. 17): 9, 1992a
O’Brien MER, Pinkerton CR, Kingston J, Mott M, Tait D, et al. ‘VEEP’ in children with Hodgkin’s disease — a regimen to decrease late sequelae. British Journal of Cancer 65: 756–760, 1992b
O’Connell MJ. Current status of chemotherapy for advanced pancreatic and gastric cancer. Journal of Clinical Oncology 3: 1032–1039, 1985
Ohno T, Shimoyama T, Kimura K, Yamada H. Pharmacokinetics of epirubicin in cancer patients. Japanes Journal of Cancer Chemotherapy 13: 1881–1886, 1986
Okunewick JP, Kociban DL, Buffo MJ. Comparative hematopoietic toxicity of doxorubicin and 4’-epirubicin (43124). Proceedings of the Society of Experimental Biology and Medicine 195: 95–99, 1990
Onohara S, Ogita M, Shimada J, Hiraki Y, Abeyama K, et al. Transarterial ‘pile-up’ infusion therapy of cisplatin and lipiodol emulsion for hepatic malignancies without TAE. Japanese Journal of Cancer Chemotherapy 19: 315–321, 1992
Oosterlinck W, Kurth K-H, Schroeder F, Silvester R, Hammond B, et al. TUR only versus epirubicine in single primary or recurrent Ta T1 bladder tumors. EORTC study 30.863. Journal of Urology 147: 272A, 1992
O’Reilly SE, Connors JM. Non-Hodgkin’s lymphoma. I: characterisation and treatment. British Medical Journal 304: 1682–1686, 1992
O’Reilly SE, Hoskins P, Howdle S, Klasa R, Klimo P, et al. POCE chemotherapy — phase II trial in elderly patients with advanced stage diffuse large cell lymphoma. Proceedings of the American Society of Clinical Oncology 11: 327, 1992
Oudard S, Giacchetti S, Extra JM, Mary M. Fluoro-uracile (F), epirubicin (E) and cisplatine (P) for patients (pts).with gastrointestinal cancer (GI). Abstract no. 53. Annals of Oncology 3 (Suppl. 5): 14, 1992
Ozyilkan O, Tekuzman G, Baltali E, Kars A, Barista I, et al. Efficacy of cyclophophamide (C), eipirubicin (E), and etoposide (VP-16) alternating with cisplatin (CP) and VP-16 in the treatment of patients (PTS) with small cell lung cancer (SCLC). Annals of Oncology 3 (Suppl. 5): 42, 1992 (abstract 164)
Painter RB. Inhibition of DNA replicon initiation by 4-nitroquinoline 1 -oxide, adriamycin, and ethyleneimine. Cancer Research 38: 4445–4449, 1978
Palmeri S, Gebbia V, Russo A, Gebbia N, Meli M, et al. Cyclophosphamide plus epidoxorubicin and 5-fluorouracil with folinic acid as a novel treatment in metastatic breast cancer: preliminary results of a phase II study. Journal of Chemotherapy 3: 176–179, 1991
Palva IP, Ahrenberg P, Ala Harja K, Almqvist A, Elonen E, et al. Intensive chemotherapy with combinations containing anthracyclines for refractory and relapsing multiple myeloma. European Journal of Haematology 44: 121–124, 1990
Pannuti F, Camaggi CM, Strocchi E, Comparsi R, Rossi AP, et al. Intrahepatic arterial administration of 4’-epidoxorubicin (epirubicin) in advanced cancer patients. A pharmacokinetic study. European Journal of Cancer Clinical Oncology 22: 1309–1314, 1986
Papacharalambous A, Alexopoulos CG, Papachristodoulou, Xynogalos S, Vaslamatzis M. Continous vs intermittent chemtherapy in advanced breast cancer using two regimens of different intensity. Abstract no. 323. Annals of Oncology 3 (Suppl. 5): 83, 1992
Passalacqua R, Cocconi G, Macaluso G, Potenzoni D, Monica B, et al. M-VAC versus the same combination using epirubicin at double dose (M-VEEC) in locally invasive bladder cancer. A prospective study. Proceedings of the American Society of Clinical Oncology 11: 210, 1992
Passalacqua R, Cocconi G, Macaluso G, Potenzoni D, Monica B, et al. Neoadjuvant chemotherapy for locally invasive bladder cancer. A prospective randomized trial comparing the M-VAC vs MVEEC combinations. European Journal of Cancer 27 (Suppl. 2): S104, 1991
Perez DJ, Harvey VJ, Robinson BA, Atkinson CH, Dady PJ, et al. A randomized comparison of single-agent doxorubicin and epirubicin as first-line cytotoxic therapy in advanced breast cancer. Journal of Clinical Oncology 9: 2148–2152, 1991
Pigram WJ, Fuller W, Hamilton LD. Stereochemistry of intercalation: interaction of daunomycin with DNA. Nature New Biology 235: 17–19, 1972
Periti P, Pannuti F, Robustellidella Cuna G, Mazzei T, Mini E, et al. Combination chemotherapy with cyclophosphamide, fluorouracil, and either epirubicin or mitoxantrone: a comparative randomized multicenter study in metastatic breast carcinoma. Cancer Investigation 9: 249–255, 1991
Phornphutkul K, Handagoon P, Sirivanichai C, Senarat S, Nakamura H. Hepatocellular carcinoma and transcatheter oily chemoembolization in Thailand. Gan to Kagaku Ryoho 19: 1239–1246, 1992
Piazza E, Filipazzi V, Pho B, Cattaneo MT, Zonato S, et al. Cyclophosphamide, epirubicin and cisplatin as first line chemotherapy in N.S.C.L.C. stage III. Abstract no. 134. Annals of Oncology 3 (Suppl. 5): 34, 1992
Pickel H, Petru E, Lahousen M, Stettner H, Lehnert M. Efficacy of adjuvant carboplatinum-epirubicin chemotherapy in advanced ovarian cancer after radical surgery. Oncology 46: 222–225, 1989
Plumbridge TW, Brown JR. Studies on the mode of interaction of 4’-epi-adriamycin and 4-demethoxy-daunomycin with DNA. Biochemical Pharmacology 27: 1881–1882, 1978
Pochanugool L, Subhadharaphandou T, Sirikulchayanonta V, Nontasut S, Hathirat P, et al. Multidisciplinary ‘limb salvage’ treatment of osteosarcoma. Journal of the Medical Association of Thailand 74: 404–411, 1991
Possinger K, Wagner H, Kovacs S, Flath B, Classen S, et al. Lon-idamine in high-risk breast cancer patients. Seminars in Oncology 18 (Suppl. 4): 58–61, 1991
Praga C, Trave F, Petroccione A. Anthracycline-induced cardiotoxicity and its relevance in cancer treatment. In Nimmo et al. (Eds) Clinical measurement in drug evaluation, pp. 131–142, Wolfe Publishing Ltd, London, 1991
Pronzato P, Amoroso D, Bertelli G, Queirolo P, Sertoli MR, et al. Chemotherapy with mitomycin-C, epidoxorubicin and vinblastine in CMF failing breast cancer patients. Anticancer Research 10: 1743–1746, 1990
Pummer K. Epirubicin plus flutamide and orchidectomy in previously untreated advanced prostatic cancer. Seminars in Oncology 18 (Suppl. 6): 26–28, 1991
Pyrhönen SO, Kouri MO. Phase II study of epirubicin sequential methotrexate and 5-fluorouracil for advanced colorectal cancer. European Journal of Cancer 28A, 1828–1832, 1992
Pyrhönen SO, Kuitunen T, Kouri M. A randomized, phase III trial comparing fluorouracil, epidoxorubicin and methotrexate (FEMTX) with best supportive care in non-resectable gastric cancer. Abstract no. 46. Annals of Oncology 3 (Suppl. 5): 12, 1992
Queisser W, Flechtner H, Heim ME, Kabelitz K, Massner B, et al. Dose escalation and split course of 4’-epidoxorubicin in combination chemotherapy (FEM II) of advanced gastric carcinoma. Onkologie 12: 202–206, 1989
Quoix EA, Giaccone G, Jassem J, Karnicka H, Wiatr E, et al. Epirubicin in previously untreated patients with small cell lung cancer: a phase II study by the EORTC lung cancer cooperation group. European Journal of Cancer 28A: 1667–1670, 1992
Rahal M, Filali T, Jemaa AD, Ferdi N, Hamani MT. MVEC: an effective and well tolerated second line regimen for refractory breast cancer. European Journal of Cancer 27 (Suppl. 2): S62, 1991
Recchia F, Serafini F, Rea S, Frati L. Phase II study of 5-fluorouracil (5FU), folinic acid (FA), epirubicine (E), mitomycin-c (MMC), beta Interferon (IFN) and retinole palmitate (R) in patients (PTS) with unresectable pancreatic carcinoma. Proceedings of the American Association of Cancer Research 33: 216, 1992
Redman C, Lawton F, Stuart N, O’Brien M, Buxton J, et al. Phase II study of combination 4’-epidoxorubicin and mitomycin C in recurrent epithelial ovarian cancer. Cancer Chemotherapy and Pharmacology 23: 51–53, 1989
Richards M. In Anthracyclines in the adjuvant treatment of early breast cancer, Adis International, Chester, pp. 31–35, 1992
Ries F, Meyer S, Le Moine F, Dicato M, Lobelle JP, et al. High dose (HD) epirubicin (E) followed by GM-CSF in advanced cancer. A dose finding study. 17th Congress of the European Society for Medical Oncology, Lyon, p. 2351, Nov 6–10, 1992
Ries F, Focan C, Closon MT, Andrien JM, Dicato M, et al. Dose intensity for epirubicin is highly correlated to tumor response in advanced breast cancer. British Journal of Cancer 62 (Suppl. 12): 30, 1990
Robert J, Bui NB. Pharmacokinetics and metabolism of epirubicin administered as i.v. bolus and 48-h infusion in patients with advanced soft-tissue sarcoma. Annals of Oncology 3: 651–656, 1992
Robert J, Hoerni B, Vrignaud P, Lagarde C. Early-phase pharmacokinetics of doxorubicin in non-Hodgkin lymphoma patients. Cancer Chemotherapy and Pharmacology 10: 115–119, 1983
Robert J, Vrignaud P, Nguyen-Ngoc T, Iliadis A, Mauriac L, et al. Comparative pharmacokinetics and metabolism of doxorubicin and epirubicin in patients with metastatic breast cancer. Cancer Treatment Reports 69: 633–640, 1985
Robustelli della Cuna G. 5-FU, CTX plus epirubicin (FEC) or novantrone (FNC) in metastatic breast cancer: 3 years results of a multicentric clinical trial. European Journal of Cancer 27 (Suppl. 2): S63, 1991
Rocchi P, Ferreri AM, Simone G, Prodi G. Epirubicin-induced differentiation of human neuroblastoma cells in vitro Anticancer Research 7: 247–250, 1987
Rocchi P, Simone G, Lorenzetti A, Granchi D, Paolucci P, et al. Effectiveness of differentiation inducers in combination on human neuroblastoma cells in vitro. Anticancer Research 9: 1605–1610, 1989
Rogan AM, Hamilton TC, Young RC, Klecker Jr RW, Oxols RF. Reversal of adriamycin resistance by verapamil in human ovarian cancer. Science 224: 994–996, 1984
Romito S, Giotta F, Maggi V, Naglieri E, Colucci G. 120 hour continuous infusion (C.I.) of 4-epidoxorubicin (4-EPI) in untreated patients with advanced breast cancer. A pilot study: preliminary data. Abstract no. 336. Annals of Oncology 3 (Suppl. 5): 87, 1992
Rosell R, Abad-Esteve A, Morera J, Monras P, Moreno I, et al. A randomized study comparing platinum, doxorubicin, and VP-16 with platinum, 4’-epidoxorubicin, and VP-16 in patients with non-small-cell lung cancer. American Journal of Clinical Oncology 10: 245–248, 1987
Rosell R, Carles J, Abad A, Jimeno JM, Moreno I, et al. Phase II feasibility study of high dose epirubicin plus etoposide. and cisplatin (HDEEC) regimen in small cell lung cancer. Investigation New Drugs 10: 123–128, 1992
Rosenthal M, Kefford R, Raghavan D, Stuart-Harris R. Epirubicin: a phase II study in recurrent small-cell lung cancer. Cancer Chemotherapy and Pharmacology 28: 220–222, 1991
Ross WE. DNA topoisomerases as targets for cancer therapy. Biochemical Pharmacology 34: 4191–4195, 1985
Rosso R, Salvati F, Rubagotti A, Ardizzoni A, Pennucci MC, et al. Potentiation of eisplatin containing chemotherapy by recombinant alfa interferon in non small cell lung cancer (NSCLC): a randomized F.O.N.I.CA.P trial. European Journal of Cancer 27 (Suppl. 2): S167, 1991
Roth A, Kolaric K. High doses of epirubicin and 5-fluorouracile in the treatment of metastatic gastric cancer — a phase II study. European Journal of Cancer 27 (Suppl. 2): S80, 1991
Roth A, Zupanc D, Luetic J, Kolaric K. Open phase II with 5-fluorouracil, 4’-epi-doxorubicin and mitomycin C (FEM) in advanced gastric cancer. Tumori 76: 51–53, 1990
Rozencweig M, ten Bokkel Huinink W, Cavalli F, Bruntsch U, Dombernowsky P, et al. Randomized phase II trial of carminomycin versus 4’-epidoxorubicin in advanced breast cancer. Journal of Clinical Oncology 2: 275–281, 1984
Ruiz A, Camps C, Godes C, Quillem V, Munoz MA, et al. Phase II high dose epirubicin (HD-4EPI) plus ciclophosphamide (CTX) in metastatic breast cancer (MBC). Abstract no. 349. Annals of Oncology 3 (Suppl. 5): 90, 1992
Ryoji O, Nakazawa H, Toma H. Effect of prophylactic intravesical instillation therapy with epi-adriamycin in patients with superficial bladder cancer. Nip-Chir-Gakk Nippon Gan Chiryo Gakkai-Shai 27: 437, 1992.
Salmon SE, Liu RM, Casazza AM. Evaluation of new anthracycline analogs with the human tumor stem cell assay. Cancer Chemotherapy and Pharmacology 6: 103–110, 1981
Sampi K, Masaoka T, Shirakawa S, Shirai T, Abe T, et al. A phase II study of epirubicin in acute leukemia: a cooperative group study. Anticancer Research 7: 29–32, 1987
Sarcina R, Lorusso V, Berardi F, Guida M, Cataldi AM, et al. Updated results of a randomized trial comparing CHOP-B vs CEOP-B in non Hodgkin’s lymphomas (NHL). European Journal of Cancer 27 (Suppl. 2): S244, 1991
Satterwhite B, Zimm S. The use of scalp hypothermia in the prevention of doxorubicin-induced hair loss. Cancer 54: 34–37, 1984
Sauer H, Mair W, Fink U, Ruckle H, Mjaaland I, et al. Polychemotherapie fortgeschrittener Weichteilsarkome mit 4’-epidoxorubicin (4’-epi-DX) und eisplatin (DDP). Onkologie 9 (Suppl. 1): 24–28, 1986
Scarffe JH, Kenny JB, Johnson RJ, Owens SE, Giles GR, et al. Phase II trial of epirubicin in gastric cancer. Cancer Treatment Reports 69: 1275–1277, 1985
Schmid H, Kaufmann M, Grischke E-M, Bastert G. Ifosfamide combination chemotherapy in advanced breast cancer. Cancer Chemotherapy and Pharmacology 26 (Suppl.): S71–S73, 1990
Schmid M, Samonigg H, Stöger H, Kasparek AK, Bauernhofer T, et al. Treatment of metastatic breast cancer with the combination of 5-fluorouracil (5-FU), high dose folinic acid (HDFA) and epirubicin (EPI) in a weekly schedule as first line chemotherapy. A phase II study. Onkologie 14 (Suppl. 2): 143, 1991
Schott B, Robert J. Comparative cytotoxicity, DNA synthesis inhibition and drug incorporation of eight anthracyclines in a model of doxorubicin-sensitive and -resistant rat glioblastoma cells. Biochemical Pharmacology 38: 167–172, 1989
Schütte J, Niederle N, Eberhardt W, Seeber S, Alberti W, et al. Sequentielle induktionschemotherapie und Strahlenbehandlung inoperabler kleinzelliger bronchialkarzinome. Klinische Wochesnschrift 67: 1182–1193, 1989
Schwartz HS. DNA breaks in P-288 tumor cells in mice after treatment with daunorubicin and adriamycin. Research Communications in Chemical Pathology and Pharmacology 10: 51–64, 1975
Scinto AF, Campioi N, Cavaliere R, Tonacnella R, La Ferla G, et al. Phase II trial of high-dose epirubicin (HD Epi) and cyclophosphamide (CTX) in advanced breast cancer. Abstract no. 316. Annals of Oncology 3 (Suppl. 5): 82, 1992
Scott WW, Menon M, Walsh PC. Hormonal therapy of prostatic cancer. Cancer 45: 1929–1936, 1980
Serretta V, Pavone C, Corselli G, Pavone-Macaluso M. A pilot study comparing different dose levels and administration schedules of interferon-α2b combined with epirubicin for prevention of recurrence in bladder cancer. Anti-Cancer Drugs 3 (Suppl. 1): 19–23, 1992
Sertoli M, Pronzato P, Queirolo P, Canavese G, Rosso R. Perioperative and postoperative chemotherapy in primary breast cancer. In Anthracyclines in the adjuvant treatment of early breast cancer, Adis International, pp. 25–29, 1992b
Sertoli MR, Pronzato P, Queirolo P, Gardin G, Miglietta L, et al. Perioperative polichemotherapy for primary breast cancer. 4th International Conference on Adjuvant Therapy of Primary Breast Cancer, St Gallen, p. 57, Feb 26–29 1992a
Shinagawa K, Ogawa M, Horikoshi N, Inoue K, Mukaiyama T, et al. Phase II study of combination chemotherapy with epirubicin, cyclophosphamide and ftorafur for metastatic breast cancer. Gan to Kagaku Ryoho 15: 3277–3281, 1988
Shiu W, Leung N, Li M, Leung WT. The efficacy of high-dose 4’-epidoxorubicin in hepatocellular carcinoma. Japanese Journal of Clinical Oncology 18: 235–237, 1988
Shiu W, Mok SD, Tsao SKOSY, Woo KS, Li A, et al. Phase II trial of epirubicin in hepatoma. Cancer Treatment Reports 70: 1035–1036, 1986a
Shiu W, ‘O’ SK, Tsao SY, Woo KS, Martin C. Phase II trial of 4’-epidoxorubicin in advanced gastrointestinal tumor. Oncology 43: 341–343, 1986b
Shiu W, Tao M, Leung T. Phase II study of combination vincristine, epirubicin and cyclophosphamide in advanced breast cancer in Chinese patients. European Journal of Cancer 27: 1710, 1991
Silva RR, Pilone A, Rossini S, Manocchi P, Mattioli R, et al. FEC + folinic acid (FA) in advanced breast cancer: preliminary results. European Journal of Cancer 27 (Suppl. 2): S57, 1991
Skarlos DV, Aravantinos G, Fountzilas G, Kosmidis P, Pavlidis N. Carboplatinum (CBDCA) alone vs carboplatinum combination in advanced ovarian cancer (O.C.) European Journal of Cancer 27 (Suppl. 2): S135, 1991
Sledge Jr GW, Antman KH. Progress in chemotherapy for metastatic breast cancer. Seminars in Oncology 19: 317–332, 1992
Sledge GW, Roth BJ, Munshi N, Baughman C, Einhorn LH, et al. Abstract no. 166 High-dose epirubicin (EPI) as initial therapy for advanced breast cancer. Proceedings of ASCO 11: 85, 1992
Smit EF, Berendsen HH, Piers DA, Smeets J, Riva A, et al. A phase II study of high dose epirubicin in unresectable non small cell lung cancer. British Journal of Cancer 65: 405–408, 1992a
Smit EF, Piers DA, Postmus PE. Phase II study of high-dose epirubicin and etoposide in advanced non-small cell lung cancer. European Journal of Cancer 28A: 1965–1967, 1992b
Smith HS, Zoli W, Volpi A, Hiller A, Lippman M, et al. Preliminary correlations of clinical outcome with in vitro chemosensitivity of second passage human breast cancer cells. Cancer Research 50: 2943–2948, 1990
Sobrero A, Muzzulini C, D’Amore F, Bogliolo G, Massa G, et al. Activity of 4’-epi-doxorubicin on normal hematopoietic precursor cells in mice. Cancer Treatment Reports 66: 2061–2070, 1982
Sola C, Vinolas N, Palombo H, Daniels M, Grau JJ, et al. Methotrexate (M), vinblastine (V), epirubicin (E) and carboplatin (CBDCA) for bladder cancer (BC). European Journal of Cancer 27 (Suppl. 2): S107, 1991
Soloway MS. Rationale for intensive intravesical chemotherapy for superficial bladder cancer. Journal of Urology 123: 461–466, 1980
Spadari S, Pedrali-Noy G, Focher F, Montecucco A, Bordoni T, et al. DNA polymerases and DNA topoisomerases as targets for the development of anticancer drugs. Anticancer Reseach 6: 935–940, 1986
Sternberg CN, Sordillo PP, Cheng E, Chuang YJ, Niedzwiecki D. Evaluation of new anticancer agents against human pancreatic carcinomas in nude mice. American Journal of Clinical Oncology 10: 219–221, 1987
Stöckle M, Meyenburg W, Wellek S, Voges G, Gertenbach U, et al. Advanced bladder cancer (stages pT3b, pT4a, pN 1 and pN2): improved survival after radical cystectomy and 3 adjuvant cycles of chemotherapy. Results of a controlled prospective study. Journal of Urology 148: 302–307, 1992
Stöger H, Samonigg H, Kasparek AK, Schmid M, Bauernhofer T, et al. Treatment of advanced breast cancer with weekly epirubicin (EPI), 5-fluorouracil (5 FU) and high dose folinic acid (HDFA). Breast Disease 4: 79, 1991
Strocchi E, Camaggi CM, Rossi AP, Angelelli B, Comparsi R, et al. Epirubicin pharmacokinetics after intrahepatic arterial and intraperitoneal administration. Drugs Under Experimental and Clinical Research 11 (4): 295–301, 1985
Sun IL, Crane FL, Chou JY, Low H, Grebing C. Transformed liver cells have modified transplasma membrane redox activity which is sensitive to adriamycin. Biochemical and Biophysical Research Communications 116: 210–216, 1983
Taal BG, ten Bokkel Huinink WW, Simonetti G, Franklin H, McVie JG. A phase II trial of sequential MTx and 5-FU alternated with 4’-epidoxorubicin and cisplatin in advanced gastric cancer. Cancer Investigation 8: 501–504, 1990
Taetle R, Howell SB, Giuliani FC, Koziol J, Koessler A. Comparison of the activity of doxorubicin analogues using colonyforming assays and human xenografts. Cancer 50: 1455–1461, 1982
Taguchi T, Ogawa M, Izuo M, Terasawa T, Yoshida M, et al. A prospective randomized trial comparing epirubicin and doxorubicin in advanced or recurrent breast cancer. Japanese Journal of Cancer Chemotherapy 13: 3498–3507, 1986
Takatsuka Y, Nakayama H, Kawahara T. Combination chemotherapy using low-dose epirubicin and vindesine in patients with metastatic breast cancer. Japan Journal of Cancer Chemotherapy 15: 667–671, 1988
Tan YO, Lim F. 4’-Epidoxorubicin (epirubicin) as a single agent in advanced primary hepatocellular carcinoma — a preliminary experience. Annals of the Academy of Medicine in Singapore 15: 169–171, 1986
Tanaka M, Yoshida S. Mechanism of the inhibition of the calf thymus DNA polymerases α and β by daunomycin and adriamycin. Journal of Biochemistry 87: 911–918, 1980
Tanaka M, Yoshida S, Kimura K. Mechanism of inhibition of DNA polymerases by 4’-epiadriamycin and 4’-O-tetrahydropyranyladriamycin. Gann 74: 829–836, 1983
Tannock IF, Boyd NF, Deboer G, et al. A randomised trial of two dose levels of cyclophosphamide, methotrexate, and fluorouracil chemotherapy for patients with metastatic breast cancer. Journal of Clinical Oncology 6: 1377–1387, 1988
Terzoli E, Rasi G, Izzo F, Nisticò C, Garufi C, et al. Combined treatment of advanced breast cancer (ABC) with epirubicin (E), al-thymosin (Tal) and alfa-interferon (IFN). Proceedings of the American Society of Clinical Oncology 11: 69, 1992
Tewey KM, Rowe TC, Yang L, Halligan BD, Liu LF. Adriamycininduced DNA damage mediated by mammalian DNA topoisomerase II. Science 226: 466–468, 1984
Thongprasert S. Analysis of the four combination chemotherapies in non-small cell lung cancer treated at Maharaj Nakorn Chiang Mai Hospital. Gan to Kagaku Ryoh 19: 1197–1201, 1992
Tidefelt U, Sundman-Engberg B, Paul C. Intracellular uptake and cytotoxic effect in vitro of doxorubicin and epirubicin in human leukemic and normal hematopoietic cells. Cancer Chemotherapy and Pharmacology 29: 7–12, 1991
Tidefelt U. Intracellular pharmacokinetics and in vitro effects of anthracyclines on human leukemic cells — relation to treatment results in acute non-lymphoblastic leukemia. Acta Oncologica 30: 75, 1991
Tokes ZA, Rogers KE, Rembaum A. Synthesis of adriamycin-coupled polyglutaraldehyde microspheres and evaluation of their cytostatic activity. Proceedings of the National Academy of Science 7.9: 2026–2030, 1982
Toma S, Coialbu T, Biassoni L, Folco U, Gatti C, et al. Epidoxorubicin plus ifosfamide in advanced and/or metastatic soft-tissue sarcomas. Cancer Chemotherapy and Pharmacology 26: 453–456, 1990
Tominaga T, Kitamura M, Nomura Y, Abe O, Enomoto K, et al. Phase II study of epirubicin on breast cancer: a cooperative group study. Japanese Journal of Cancer Chemotherapy 13: 2187–2192, 1986
Toner GC, Bishop JF, Fennessy A, Woollett A, Olver IN, et al. Escalation of dose intensity of single agent epirubicin with granulocyte-macrophage colony stimulating factor (GM-CSF). Proceedings of the American Society of Clinical Oncology 11: 415, 1992
Topham C, Glees J, Rawson NSB, Woods EM, Coombes RC. Randomised trial of epirubicin alone versus 5-fluorouracil, epirubicin and mitomycin C in locally advanced and metastatic carcinoma of the pancreas. British Journal of Cancer 64: 179–181, 1991
Torti FM, Bristow MM, Lum BL, Carter SK, Howes AE, et al. Cardiotoxicity of epirubicin and doxorubicin: assessment by endomyocardial biopsy. Cancer Research 46: 3722–3727, 1986
Torti FM, Lum BL. The biology and treatment of superficial bladder cancer. Journal of Clinical Oncology 2: 505–531, 1984
Torti FM, Porzig KJ, Gandara DR, Volberding P, Mitchell E, et al. Phase II trial of 4’-epi-doxorubicin in metastatic melanoma. Cancer Treatment Reports 68: 1509–1510, 1984
Treat J, Falchuk SC, Woolley PV, Ahlgren JD, Neefe JR, et al. Therapy of advanced gastric carcinoma. The Georgetown-Lombardi Cancer Center Experience. American Journal of Clinical Oncology 12: 162–168, 1989
Tritton TR, Yee G. The anticancer agent adriamycin can be actively cytotoxic without entering cells. Science 217: 248–250, 1982
Tsuruo T, Iida H, Yamashiro M, Tsukagoshi S, Sakurai Y. Enhancement of vincristine- and adriamycin-induced cytotoxicity by verapamil in P388 leukemia and its sublines resistant to vincristine and adriamycin. Biochemical Pharmacology 31: 3138–3140, 1982
Tsushima T, Matsumura Y, Ozaki Y, Akagi T, Obama T, et al. Intravesical chemotherapy with 4’-epi-adriamycin in patients with superficial bladder tumors (randomized study). Hinyokika Kiyo31: 2215–2218, 1985
Tsushima K, Sakata Y, Suzuki H, Saitoh S, Sugimoto N, et al. A randomized controlled study of 5-fluorouracil/doxorubicin/mitomycin C/OK-432 (FAM-OK) therapy and 5-fluorouracil/epirubicin/mitomycin C/OK-432 (FEM-OK) therapy in advanced gastric cancer. Nippon Gan Chiryo Gakka Shi 26: 1317–1324, 1991
Tucci E, Algeri R, Guarnieri A, Pepi F, Sapio L, et al. Weekly epirubicin in advanced breast cancer. Tumori 74: 689–692, 1988
Tveter KJ, Hagen S, Holme I, Klepp O, Kloster SE, et al. A randomized study on hormone-resistant prostatic cancer: estramustine phosphate versus low dose epirubicin with or without medroxyprogesterone acetate. Scandinavian Journal of Urology and Nephrology 24: 243–247, 1990
Twelves C, Dobbs N, Summers L, Harper P, Richard M, et al. Pharmacokinetics of epirubicin (EPI) in patients with abnormal liver biochemistry. British Journal of Cancer 62: 528–529, 1990
Twelves CJ, Dobbs NA, Michael Y, Summers LA, Gregory W, et al. Clinical pharmacokinetics of epirubicin: the importance of liver biochemistry tests. British Journal of Cancer 66: 765–769, 1992
Twelves CJ, O’Reilly SM, Coleman RE, Richards MA, Rubens RD. Weekly epirubicin for breast cancer with liver metastases and abnormal liver biochemistry. British Journal of Cancer 60: 938–941, 1989
Twelves CJ, Richards MA, Smith P, Rubens RD. Epirubicin in breast cancer patients with liver metastases and abnormal liver biochemistry: initial weekly treatment followed by rescheduling and intensification. Annals of Oncology 2: 663–666, 1991
Urba WJ, Longo DL. Hodgkin’s disease. New England Journal of Medicine 326: 678–687, 1992
Valenza R, Gebbia V, Testa A, Latteri MA, Cipolla C, et al. Levo folinic acid (1-FA) and 5-fluorouracil (5-FU) plus high dose epidoxorubicin (EDXR) in metastatic breast carcinoma (MBC): preliminary data. Abstract no. 340. Annals of Oncology 3 (Suppl. 5): 88, 1992
van der Meijden APM, Kurth K-H, Oosterlinck W, Debruyne FMJ, Members of the EORTC Genito-urinary Group. Intravesical therapy with adriamycin and 4-epirubicin for superficial bladder cancer: the experience of the EORTC GU Group. Cancer Chemotherapy and Pharmacology 30 (Suppl.): S95–S98, 1992
van de Velde. Preoperative chemotherapy in operable breast cancer (POCOB) — the influence of timing FEC in relation to surgery. Drugs (Suppl.), in press, 1993
Villani F, Favalli L, Piccinini F. Relationship between the effect on calcium turnover and early cardiotoxicity of doxorubicin and 4’-epi-doxorubicin in guinea pig heart muscle. Tumori 66: 689–697, 1980
Venturini M, Sertoli MR, Ardizzoni A, Danova M, Brema F, et al. Prospective randomized trial of accelerated FEC chemotherapy (CT) with or without GM-CSF in advanced breast can cer (ABC). Proceedings of the American Society of Clinical Oncologists 11: 52, 1992
Verhees S, Flechtner H, Queisser W, Heim ME, Fritz M, et al. Behandlung des fortgeschrittenen Pankreaskarzinoms mit 5-Fluorouracil, 4-Epidoxorubicin und Mitomycin C (FEM II). Aus der chemotherapeigruppe gastrointesinaler tumoren (CGT). Onkologie 13: 50–52, 1990
Vermorken JB. High-dose intensity regimens with epirubicin in ovarian cancer. Pathologie Biologie 39: 833, 1991
Vermorken JB, ten Bokkel Huinink WW, Kobierska A, Forni M, van der Burg MEL, et al. Antitumor activity of high-dose epirubicin (HDE) in patients with ovarian cancer (OC) failing platinum (P)-based regimens. Abstract no. 1117. Proceedings of the American Association for Cancer Research 31: 188, 1990
Vermorken JB, Zanaboni F, Kobierska A, Chevallier B, van der Burg MEL, et al. Phase II study of high-dose epirubicin (HDE) in patients with ovarian carcinoma (OC) previously treated with cisplatin (P). Annals of Oncology 3 (Suppl. 5): 104, 1992 (abstract 402)
Verweij J, Herweijer H, Oosterom R, van der Burg MEL, Planting ASTh, et al. A phase II study of epidoxorubicin in colorectal cancer and the use of cyclosporin-A in an attempt to reverse multidrug resistance/British Journal of Cancer 64: 361–364, 1991
Villalon AH, Guancia AA, Machica MEG. Comparison of epirubicin and doxorubicin combination therapy for intermediate and high grade non-Hodgkin’s lymphoma. Philippine Journal of Internal Medicine 29: 109–113, 1991
Villar Aj, Anton A, Aranda E, Belón J, Balana C, et al. High dose epirubicin (HDE) in lung adenocarcinoma (LAD). European Journal of Cancer 27 (Suppl. 2): S174, 1991
Voce S, Montanari F, Arnone S, Dal Pozzo C, Suprani G, et al. Phase I-II pilot study on the efficacy and tolerability of neoadjuvant chemotherapy (rescue M-VEC) and preoperative radiation therapy for infiltrating bladder cancer: results of an 18-month follow-up. Archivos Espanoles de Urologia 45: 491–498, 1992
Vogl SE, Pagano M, Kaplan BH, Greenwald E, Arseneau J, et al. Cis-platin based combination chemotherapy for advanced ovarian cancer. High overall response rate with curative potential only in women with small tumor burdens. Cancer 51: 2024–2030, 1983
Von Hoff DD, Layard MW, Basa P, Davis Jr HL, Von Hoff AL, et al. Risk factors for doxorubicin-induced congestive heart failure. Annals of Internal Medicine 91: 710–717, 1979
Vorobiof DA, Falkson G. Phase II study of high-dose 4’-epidoxorubicin in the treatment of advanced gastrointestinal cancer. European Journal of Cancer Clinical Oncology 25: 563–564, 1989
Vrignaud P, Eghbali H, Hoerni B, Iliadis A, Robert J. Pharmacokinetics and metabolism of epirubicin during repetitive courses of administration in Hodgkin’s patients. European Journal Cancer Clinical Oncology 21: 1307–1313, 1985
Wade JR, Kelman AW, Kerr DJ, Robert J, Whiting B. Variability in the pharmacokinetics of epirubicin: a population analysis. Cancer Chemotherapy and Pharmacology 29: 391–395, 1992
Wadler S, Fuks JZ, Wiernik PH. Phase I and II agents in cancer therapy: I. anthracyclines and related compounds. Journal of Clinical Pharmacology 26: 491–509, 1986
Wagener D, Wils J, Conroy Th, Burghouts J, Fickers M, et al. Phase II trial with FEMTX-P in advanced gastric cancer. Proceedings of the American Society of Clinical Oncology 11: 189, 1992
Wassermann K, Mølgaard K, Steiness E. Similar changes in cardiac morphology and DNA synthesis induced by doxorubicin and 4’-epi-doxorubicin. Cancer Chemotherapy and Pharmacology 15: 244–252, 1985
Watanabe T, Miya T, Fukutomi T, Okamoto R, Adachi I. Phase I study of high-dose epirubicin (EPI) and cyclophosphamide (CPA) with rG-CSF for metastatic breast cancer. Proceedings of the American Society of Clinical Oncology 11: 88, 1992
Weenen H, Lankelma J, Penders PGM, McVie JG, ten Bokkel Huinink WW, et al. Pharmacokinetics of 4’-epi-doxorubicin in man. Investigational New Drugs 1: 59–64, 1983
Whelan P, Cumming JA, Garvie WHH, Hargreave TB, Kirk D, et al. Multi-centre phase II study of low dose intravesical epirubicin in the treatment of superficial bladder cancer. British Journal of Urology 67: 600–602, 1991.
Wickerham DL, Wolmark N. Diseases of the breast. In Robert ER (Ed.) Latest approved methods of treatment for the practicing physician. WB Saunders Company, Philadelphia, 986–989, 1991
Wils J, Bleiberg H, Blijham G, Dalesio O, Duez N, et al. Phase II study of epirubicin in advanced adenocarcinoma of the pancreas. European Journal of Cancer Clinical Oncology 21: 191–194, 1985
Wils J, Bleiberg H, Buyse M, Wagener DTh, Splinter T, et al. An EORTC gastrointestinal group phase II evaluation of epirubicin combined with ifosfamide in advanced adenocarcinoma of the pancreas. European Journal of Cancer Clinical Oncology 25: 1119–1120, 1989
Wils J, Bleiberg H, Dalesio O, Duez N, Blijham G, et al. An EORTC Gastrointestinal group phase II evaluation of epirubicin combined with 5-fluorouracil in advanced adenocarcinoma of the pancreas. European Journal of Cancer Clinical Oncology 24: 1017–1018, 1987
Wils J, Smeets J. Phase II study of cyclophosphamide (C) epirubicin (E), oncovin (O), prednisone (P), methotrexate (M) + leucovorin and bleomycin (B) (CEOP-MB) in intermediate and high grade non-Hodgkin lymphomas. Journal of Chemotherapy 3: 260–263, 1991
Wils J, Utama I, Smeets J. Chemotherapy consisting of epirubicin, cisplatin, and etoposide followed by irradiation in stage III non-small cell lung cancer and chemotherapy alone in stage IV non-small cell lung cancer. Oncology 45: 413–416, 1988
Wils J, Utama I, Sala L, Smeets J, Riva A. Phase II study of high-dose epirubicin in non-small cell lung cancer: European Journal of Cancer 26: 1140–1141, 1990
Wilson RG, Kalonaros V, King M, Lockwood R, McNeill M. Comparative inhibition of nuclear RNA synthesis in cultured mouse leukemia L1210 cells by adriamycin and 4’-epi-adriamycin. Chemico-Biological Interactions 37: 351–363, 1981
Wiltshaw E, Evans B, Rustin G, Gilbey E, Baker J, et al. A prospective randomized trial comparing high-dose cisplatin with low-dose cisplatin and chlorambucil in advanced ovarian carcinoma. Journal of Clinical Oncology 4: 722–729, 1986
Wishart GC, Plumb JA, Kaye SB. The effect of quinidine on intracellular distribution of epirubicin in an animal model of multidrug resistance. British Journal of Cancer 65 (Suppl. 16): 63, 1992
Wong L-C, Choy DTK, Ngan HYS, Sham JST, Ma H-K. 4-Epidoxorubicin in recurrent cervical cancer. Cancer 63: 1279–1282, 1989
Yamamoto M, Furuse K, Fukuoka M, Kawahara M, Arai R, et al. Phase II studies of single agent and cisplatinum-based two drug combination in non-small cell lung cancer patients. Japanese Journal of Cancer Chemotherapy 15: 487–492, 1988
Yeung TK, Simmonds RH, Hopewell JW. A functional assessment of the relative cardiotoxicity of adriamycin and epirubicin in the rat. Radiotherapy and Oncology 15: 275–284, 1989a
Yeung TK, Simmonds RH, Hopewell JW. The relative toxicity of intravenous and intraperitoneal doses of epirubicin. Cancer Chemotherapy and Pharmacology 24: 211–218, 1989b
Young CW, Raymond V. Clinical assessment of the structure-activity relationship of anthracyclines and related synthetic derivatives. Cancer Treatment Reports 70: 51–63, 1986
Young RC, Ozols RF, Myers CE. The anthracycline antineoplastic drugs. New England Journal of Medicine 305: 139–153, 1981
Zamagni C, Martoni A, Ercolino L, Baroni M, Tanneberger S, et al. 5-Fluorouracil, epirubicin and chyclophosphamide (FEC combination) in advanced breast cancer. Journal of Chemotherapy 3: 126–129, 1991
Zanaboni F, Scarfone G, Presti M, Maggi R, Borello C, et al. Salvage chemotherapy for ovarian cancer recurrence: weekly cisplatin in combination with epirubicin or etoposide. Gynecologic Oncology 43: 24–28, 1991
Zaniboni A, Marpicati P, Simoncini E, Gorni F, Ferrari V, et al. Cyclophosphamide, epirubicin and cisplatin (CEP) in advanced breast cancer: preliminary results. Anticancer Research 7: 813–816, 1987
Zaniboni A, Simoncini E, Marpicati P, Montini E, Ferrari V, et al. Cyclophosphamide, epirubicin, high-dose folinic acid and 5-fluorouracil (super-FEC) as first-line chemotherapy for advanced breast cancer: preliminary results. European Journal of Cancer Clinical Oncology 25: 1151–1155, 1989
Ziegler LD, Buzdar AU. Current status of adjuvant therapy of early breast cancer. American Journal of Clinical Oncology 14: 101–110, 1991
Zinzani PL, Mazza P, Gherlinzoni F, Zanchini R, Bocchia M, et al. CEOP regimen in the treatment of advanced low-grade non-Hodgkin’s lymphomas: preliminary report. Tumori 76: 533–536, 1990
Zittoun R, Eghbali H, Audebert A, Rojouan J, David B, et al. Association d’epirubicine, bleomycine, vinblastine et prednisone (EBVP) avant radiotherapie dans les stades localises de la maladie de Hodgkin. Essai de phase II. Bulletin du Cancer 74: 151–157, 1987
Zoli W, Volpi A, Bonaguri C, Riccobon A, Savini S, Brizio R, et al. An efficient method for culturing human breast carcinoma to evaluate antiblastic drug activity in vitro: experience on 136 primary cancers and on 116 recurrences. Breast Cancer Research and Treatment 17: 231–238, 1990
Author information
Authors and Affiliations
Additional information
Various sections of the manuscript reviewed by: D.J. Booser, University of Texas, MD Anderson Cancer Center, Houston, Texas, USA; O. Cantoni, Istituto di Farmacologia e Farmacognosia, Università degli Studi di Urbino, Urbino, Italy; C. Gridelli, Cattedra di Oncologia Medica, Università di Napoli, Napoli, Italy; T. Habeshaw, Beatson Oncology Centre, Western Infirmary, Glasgow, Scotland; H.D. Homesley, Bowman Gray School of Medicine, Wake Forest University, Winston-Salem, North Carolina, USA; G.N. Hortobagyi, University of Texas, MD Anderson Cancer Center, Houston, Texas, USA; M. Iigo, Chemotherapy Division, National Cancer Center Research Institute, Tokyo, Japan; M. Kajanti, Department of Radiotherapy and Oncology, Helsinki University Central Hospital, Helsinki, Finland; S. Kohnoe, Cancer Center of Kyushu University Hospital, Faculty of Medicine, Kyushu University, Fukuoka, Japan; R.M. Lowenthal, Clinical Haematology and Medical Oncology Unit, Royal Hobart Hospital, Hobart, Tasmania, Australia; G. Mathé, Institut de Cancérologie et d’Immunologie, Villejuif, France; H.T. Mouridsen, Department of Oncology, Rigshospitalet, Copenhagen, Denmark; J. Wils, St. Laurentius Hospital, Roermond, The Netherlands.
Rights and permissions
About this article
Cite this article
Plosker, G.L., Faulds, D. Epirubicin. Drugs 45, 788–856 (1993). https://doi.org/10.2165/00003495-199345050-00011
Published:
Issue Date:
DOI: https://doi.org/10.2165/00003495-199345050-00011