Summary
Synopsis
Ceforanide1 is a ‘second generation’ cephalosporin administered intravenously or intramuscularly. It is similar to cefamandole and cefonicid in its in vitro superiority to ‘first generation’ cephalosporins against several species of Enterobacteriaceae as well as its activity against Haemophilus influenzae, including β- lactamase- producing strains. Its activity against Staphylococcus aureus is less than that of cefamandole, cefuroxime and first generation cephalosporins. The in vitro activity against Neisseria gonorrhoeae is excellent. Pseudomonas, Acinetobacter and Serratia species, and Bacteroides fragilis are resistant, as are many strains of Proteus and Providencia species.
The elimination half- life is relatively long, although shorter than that of cefonicid, and in most clinical trials ceforanide has been administered twice daily. It appeared to be comparable in therapeutic efficacy to procaine penicillin and cephazolin in the treatment of patients with community- acquired pneumonia, to cephazolin in the treatment of skin and soft tissue infections due to S. aureus or β- haemolytic streptococci and to cefapirin in S. aureus endocarditis in parenteral drug abusers. Also, it was comparable in efficacy to cephalothin in the prophylaxis of infection in patients undergoing open heart surgery or vaginal hysterectomy, and to cephazolin in patients undergoing cholecystectomy.
Thus, ceforanide is an alternative to first and certain other second generation cephalosporins in several important therapeutic and prophylactic situations. It has no advantage over other cephalosporins with regard to spectrum of antibacterial activity, but has a longer half- life than other second generation cephalosporins, except cefonicid, and can be administered according to a twice daily dosage schedule.
Antibacterial Activity
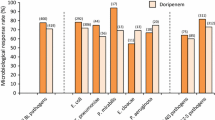
Ceforanide has a spectrum of in vitro antibacterial activity which most closely resembles those of cefamandole and cefonicid. Most isolates of Staphylococcus aureus (except methicillin-resistant strains) are susceptible or moderately susceptible to ceforanide, but ceforanide is a less potent inhibitor of staphylococci than cephazolin, cephalothin, cefamandole or cefuroxime. In common with other cephalosporins, ceforanide is a potent inhibitor of most species of streptococci, excluding enterococci. The activity of ceforanide against Escherichia coli is similar to that of cefamandole and cefoxitin and greater than that of cephazolin and cephalothin. Citrobacter diversus is very susceptible to ceforanide but other Citrobacter species, including C. freundii, are generally resistant. The comparative inhibitory activity of ceforanide, cefamandole and cefoxitin against Klebsiella species and K. pneumoniae varied between studies, with ceforanide generally being the most potent. Salmonella species are very susceptible to ceforanide while Shigella species are moderately susceptible. In general, Enterobacter, Proteus, Providencia and Serratia species are resistant to ceforanide. Neisseria gonorrhoeae and Neisseria meningitidis, including penicillinase-producing strains, and Haemophilus influenzae, including β-lactamase-producing strains are susceptible to ceforanide. However, Pseudomonas and Acinetobacter species are resistant. Ceforanide is less active than cefoxitin against anaerobic bacteria; Bacteroides fragilis is resistant but Clostridium species and Gram-positive anaerobic cocci are susceptible. The activity of ceforanide against Clostridium difficile has not been reported.
In general, increases in the inoculum size up to 105 colony-forming units (cfu) has little effect on the minimum inhibitory concentration (MIC) of ceforanide. However, when the size of the inoculum is increased to 106 cfu or greater considerable increases occur in the MIC, particularly with the Enterobacteriaceae. This may be the result of hydrolysis of the drug by β-lactamases with such large inocula. Ceforanide MICs generally are not affected by use of different growth media, the presence of human serum or pH changes.
Minimum bactericidal concentrations (MBC) of ceforanide are equivalent to or no more than 2-fold higher than MICs for most bacterial species. However, bactericidal concentrations against strains of Enterobacter species, indole-positive Proteus species, staphylococci and streptococci may exceed inhibitory concentrations by several-fold.
No significant synergism has been reported for ceforanide and other antibacterial agents.
Ceforanide is resistant to hydrolysis by the type IIa β-lactamase and the penicillinase of S. aureus. In addition, ceforanide is similar in stability to cephradine against the RTEM enzyme (type IIIa) and type IVa β-lactamase. The selection of resistant isolates occurred more frequently in ceforanide-treated mice than in mice treated with other cephalo-sporins; cross-resistance was complete between ceforanide and cefamandole, partial between ceforanide and cephalothin, but rare with cefoxitin.
Pharmacokinetics
Following intravenous administration the plasma concentration profile of ceforanide is best characterised by a first-order, 2-compartment open model. Average peak plasma concentrations in healthy adults generally ranged from 135 to 140 mg/L following a single 30-minute intravenous infusion or rapid intravenous bolus injection of 1g, and increased approximately linearly with dose. Ceforanide is 100% bioavailable following intramuscular administration, producing a peak plasma concentration, after 1 hour, of 50 to 60% of the peak concentration after intravenous infusion. Results of multiple-dose studies indicate no significant drug accumulation after up to 11 days’ administration of ceforanide.
The mean apparent volume of distribution in healthy adult volunteers is 8 to 13L. Ceforanide concentrations likely to afford sufficient activity against susceptible organisms are achieved in most tissues and fluids, including bone, cardiac tissue, female pelvic organ tissues, abscess fluid, gallbladder tissue and bile and urine. However, penetration into synovial fluid and saliva is poor. Ceforanide is approximately 80% bound to plasma proteins, but the significance of this to its clinical potential is unclear since its antibacterial activity in vitro is influenced little by human serum.
Ceforanide is primarily excreted by glomerular filtration as unchanged drug, with an average urinary recovery of 83 to 95% over the first 24 hours. Total body clearance approximates 3.0 L/h with a renal clearance of approximately 2.1 to 2.7 L/h. The mean plasma elimination half-life of ceforanide is approximately 2.5 to 3.0 hours in healthy adults.
The plasma and renal clearances of ceforanide decrease linearly as the creatinine clearance decreases. The elimination half-life increased to 25 hours in patients with severe renal insufficiency (creatinine clearance < 5 ml/min). Ceforanide is removed from the body by haemodialysis.
The elimination half-life of ceforanide is somewhat shorter in infants and children than adults, and the extent of protein binding may be reduced in children. The mean peak plasma concentrations attained in women with suspected endometritis after caesarean section were lower than those reported in healthy volunteers and may be due to an increased volume of distribution in this population.
The pharmacokinetics of intramuscularly administered ceforanide were unaltered by concomitant administration of probenecid.
Therapeutic Trials
Ceforanide 0.5 to 2g 12-hourly has proven to be therapeutically effective in several types of infection. Clinical and bacteriological cures are obtained in nearly 100% of patients with pneumonia due to S. pneumonia, while H. influenzae infections may persist after therapy. Randomised comparative trials reveal it is similar in efficacy to procaine penicillin 600,000U 12-hourly and cephazolin 0.5 to lg 8-hourly. In skin and soft tissue infections (mostly cellulitis) due to S. aureus or β-haemolytic streptococci, ceforanide clinical and bacteriological efficacy rates are approximately 95%; randomised clinical trials comparing ceforanide and cephazolin reveal similar efficacy rates for the 2 cephalosporins in these types of infections. Equally high clinical and bacteriological efficacy rates have also been reported for ceforanide in S. aureus endocarditis in parenteral drug abusers; ceforanide appeared to be similar in clinical and bacteriological efficacy to cefapirin 2g 4-hourly. In uncomplicated lower urinary tract infection there was no statistically significant difference in clinical efficacy between a single 1g intramuscular dose of ceforanide and 7 days’ treatment with ampicillin 250mg 4 times daily, although there was a trend towards a higher failure rate with ceforanide (33% and 15%, respectively). Ceforanide has also shown promising therapeutic results in a few patients with osteomyelitis and in paediatric patients with infections of various body sites (excluding the central nervous system).
Several randomised trials have assessed the comparative efficacy of ceforanide and cephalothin in the prophylaxis of infection following open heart surgery or vaginal hysterectomy. In patients undergoing open heart surgery, a single preoperative dose of ceforanide 0.5 to lg, followed by 2 to 4 doses administered 12-hourly after surgery was, in general, similar in efficacy to 6 to 10 doses of cephalothin 1g 6-hourly (beginning with a single preoperative dose). However, in a study which analysed the patients who underwent cardiac artery bypass grafting independently of the patients who underwent grafting plus additional procedures (e.g. valve replacement), the patients in the latter subgroup had a significantly (p=0.001) lower overall infection rate with ceforanide than cephalothin prophylaxis. In patients undergoing vaginal hysterectomy with or without repair there were no statistically significant differences in the rate of infection, or other indices of prophylactic efficacy, between a preoperative dose of ceforanide 0.5g followed by a single postoperative dose 12 hours later and a preoperative (dose of cephalothin followed by 5 postoperative doses 6-hourly. Intrauterine administration of a single 2g dose of ceforanide was not significantly different in efficacy from intravenous administration for the prophylaxis of infection in patients undergoing non-elective caesarean section after rupture of membranes for 3 hours or longer.
There were no postoperative infections among any of the patients undergoing cholecystectomy (with or without additional procedures) who were included in a randomised comparison of prophylaxis with ceforanide 1g (a single preoperative dose alone or followed by a single postoperative dose 12 hours later) or cephazolin 1g (a single preoperative dose followed by 3 postoperative doses 6-hourly). In addition, in noncomparative studies, ceforanide has demonstrated a high rate of prophylactic efficacy in patients undergoing elective gastric bypass surgery, surgery for acute non-perforated appendicitis, and total joint arthroplasty.
Adverse Effects
Significant adverse effects from ceforanide are uncommon. Discomfort or pain associated with intramuscular injection, mild gastrointestinal effects, and skin rash or other hypersensitivity reactions occur occasionally but rarely necessitate discontinuation of therapy. Other untoward effects that have been reported rarely (incidence less than 1%) are lethargy, confusion, headache and hypotension. Mild, transient alterations in laboratory values, similar to those seen with other cephalosporins (eosinophilia, thrombocytosis, elevated liver function test values, serum creatinine and blood urea nitrogen) also occur with ceforanide. The development of positive direct and indirect Coombs’ tests (without haemolysis) occurred in 3 of 52 patients in 1 study.
Dosage and Administration
The recommended adult dosage of ceforanide is 0.5 to 1g administered every 12 hours by intravenous injection over 3 to 5 minutes, infusion over 30 minutes or by deep intramuscular injection. The perioperative prophylactic dose for adults undergoing surgery, where there is presumed contamination or the potential for contamination, is 0.5 to 1g administered 1 hour prior to the initiation of surgery; prophylactic ceforanide may be continued for up to 2 days postoperatively when significant risk of postoperative infection exists, e.g., prosthetic arthroplasty or open heart surgery.
For children the recommended dose of ceforanide is 20 to 40 mg/kg/day in equally divided doses.
The dosage of ceforanide should be reduced in patients with impaired renal function.
Similar content being viewed by others
References
Aswapokee N, Aswapokee P, Kwung P Fu, Neu HC. In vitro activity and beta-lactamase stability of BL-S786 compared with those of other cephalosporins. Antimicrobial Agents and Chemotherapy 14: 1–5, 1978
Barry AL, Jones RN, Packer RR. Antistaphylococcal activity of ceforanide and cefonicid in the presence of human serum. Antimicrobial Agents and Chemotherapy 29: 147–149, 1986
Bell RM, Record KE, Bivins B, Rapp RP. The prophylactic use of ceforanide in gastric bypass surgery. Advances in Therapy 1: 329–335, 1984
Bennett JV, Brodie JL, Benner EJ, Kirby WMM. Simplified, accurate method for antibiotic assay of clinical specimens. Applied Microbiology 14: 170, 1966
Burch KH, Pohlod D, Saravolatz LD, Madhavan T, Kiani D, et al. Ceforanide: in vitro and clinical evaluation. Antimicrobial Agents and Chemotherapy 16: 386–391, 1979
Cady WJ, Bradley GW, O’Neil MT, Burney Jr DW, Hamsa Jr WR. Ceforanide vs. cephalothin concentrations in total joint arthroplasty. Drug Intelligence and Clinical Pharmacy 17: 645–648, 1983
Cardenas J, Burch K, Salo S, Saravolatz L, Madhavan T, et al. Comparative clinical trial of ceforanide and cefazolin in skin and soft tissue infections. 20th Interscience Conference on Antimicrobial Agents and Chemotherapy, New Orleans, Sept. 22–24, 1980. Abstract no. 629, 1980
Carrizosa J, Kobasa WB, Kaye D. Comparison of ceforanide, cefazolin, methicillin and nafcillin in Staphylococcus aureus endocarditis therapy in rabbits. Antimicrobial Agents and Chemotherapy 18: 562–565, 1980
Cooper RH, Savitch CB, Joseph WP, Mills J. Evaluation of ceforanide as treatment for staphylococcal and streptococcal endocarditis. Antimicrobial Agents and Chemotherapy 19: 256–259, 1981
Counts GW, Turck M. Antibacterial activity of a new parenteral cephalosporin — HR 756: comparison with cefamandole and ceforanide. Antimicrobial Agents and Chemotherapy 16: 64–68, 1979
Dajani AS, Thirumoorthi MC, Bawdon RE, Buckley JA, Pfeffer M, et al. Pharmacokinetics of intramuscular ceforanide in infants, children, and adolescents. Antimicrobial Agents and Chemotherapy 21: 282–287, 1982
DiPiro JT, Bayoumi SM, Vallner JJ, Nesbit RR, Gokhale R, et al. Intraoperative ceforanide pharmacokinetics and protein binding. Antimicrobial Agents and Chemotherapy 27: 487–490, 1985
Done AK, Cohen SN, Strebel L. Pediatric clinical pharmacology and the ‘therapeutic orphan’. Annual Reviews of Pharmacological Toxicology 17: 561–573, 1977
Dornbusch K, Nord CE, Olsson-Liljeqvist B. Antibiotic susceptibility of anaerobic bacteria with special reference to Bacteroides fragilis. Scandinavian Journal of Infectious Diseases (Suppl. 19): 17-25, 1979
Dudley MN, Nightingale CH, Quintiliani R, Tilton RC. In vitro activity of cefonicid, ceforanide, and cefazolin against Staphylococcus aureus and Staphylococcus epidermidis and the effect of human serum. Journal of Infectious Diseases 148: 178, 1983
Dudley MN, Quintiliani R, Nightingale CH. Review of cefonicid, a long-acting cephalosporin. Clinical Pharmacy 3: 23–32, 1984
Estey EH, Weaver SS, LeBlanc BM, Brown N, Ho DH, et al. Ceforanide kinetics. Clinical Pharmacology and Therapeutics 30: 398–403, 1981
Goering RV, Sanders CC, Sanders Jr WE. Comparison of BL-S786 with cephalothin, cefamandole and cefoxitin in vitro and in treatment of experimental infections in mice. Journal of Antibiotics 31: 363–372, 1978
Gottstein WJ, Kaplan MA, Cooper JA, Silver VH, Nachfolger SJ, et al. 7-(2-Aminomethylphenylacetamido)-3-(1-carboxymethyltetrazol-5-ylthiomethyl)-3-cephem-4-carboxylic acid. Journal of Antibiotics 29: 1226–1229, 1976
Greenberg RN, Bollinger M. Comparison of Escherichia coli susceptibility to cephalothin, cefazolin, ceforanide, and cefamandole. Clinical Therapeutics 7: 151–153, 1985
Greenman RL, Arcey SM, Gutterman DA, Zweig RM. Twice-daily intramuscular ceforanide therapy of staphylococcus aureus endocarditis in parenteral drug abusers. Antimicrobial Agents and Chemotherapy 25: 16–19, 1984
Grove DC, Randall WA. Assay methods of antibiotics: a laboratory manual. In New York Medical Encyclopedia, Inc., New York, 1955
Hanna CB. Single-dose ceforanide prophylaxis in biliary tract surgery — comparison with multiple-dose regimens. Advances in Therapy 1: 321–328, 1984
Hawkins SS, Alford RH, Stone WJ, Smyth RD, Pfeffer M. Ceforanide kinetics in renal insufficiency. Clinical Pharmacology and Therapeutics 30: 468–474, 1981
Hess JR, Berman SJ, Boughton WH, Sugihara JG, Musgrave JE, et al. Pharmacokinetics of ceforanide in patients with end stage renal disease on hemodialysis. Antimicrobial Agents and Chemotherapy 17: 251–253, 1980a
Hess JR, Berman SJ, Boughton WH, Sugihara JG, Wong EGC, et al. Ceforanide treatment of infections in patients undergoing hemodialysis. 20th Interscience Conference on Antimicrobial Agents and Chemotherapy, New Orleans, Sept. 22–24, 1980. Abstract no. 654, 1980b
Hooton TM, Blair AD, Turck M, Counts GW. Synergism at clinically attainable concentrations of aminoglycoside and beta-lactam antibiotics. Antimicrobial Agents and Chemotherapy 26: 535–538, 1984
Jacobson JA, Hebertson R, Kasworm E. Comparison of ceforanide and cephalothin prophylaxis for vaginal hysterectomies. Antimicrobial Agents and Chemotherapy 22: 643–647, 1982
Jones RN, Fuchs PC, Gavan TL, Gerlach EH, Barry A, et al. BL-S786, a new parenteral cephalosporin. I. A collaborative in vitro susceptibility comparison to cephalothin against 5,762 clinical bacterial isolates. Journal of Antibiotics 30: 576–582, 1977a
Jones RN, Thornsberry C, Barry AL, Fuchs PC, Gavan TL, et al. BL-S786, new parenteral cephalosporin. IL In vitro antimicrobial activity comparison with six related ccphalosporins. Journal of Antibiotics 30: 583–592, 1977b
Jovanovich JF, Saravolatz LD, Burch K, Pohlod DJ. Failure of probenecid to alter the pharmacokinetics of ceforanide. Antimicrobial Agents and Chemotherapy 20: 530–532, 1981
Karney W, Correa-Coronas R, Zajtchuk R, Schwartz J, Smith LP, et al. Comparison of cephalothin and ceforanide prophylaxis in cardiac surgery with cardiopulmonary bypass. Antimicrobial Agents and Chemotherapy 24: 85–88, 1983
Kenady DE, Ram MD. Biliary levels of ceforanide. Antimicrobial Agents and Chemotherapy 23: 706–709, 1983
Kerbs SB, Stone JR, Berg SW, Harrison WO. In vitro antimicrobial activity of eight new beta-lactam antibiotics against penicillin-resistant neisseria gonorrhoeae. Antimicrobial Agents and Chemotherapy 23: 541–544, 1983
Khurana CM, Deddish PA. In vitro susceptibility of 1035 gramnegative enteric pathogens to moxalactam and five other cephalosporins. Current Therapeutic Research 34: 136–141, 1983
Krause LK, Guyer W. Penetration of ceforanide into bone. Abstract no. A5. 84th Meeting of the American Society for Microbiology, St Louis, Missouri, 4–9 Mar, 1984
Lanao JM, de Prada CR, Dominguez-Gil A, Tabernero JM, Martin J, et al. Ceforanide pharmacokinetics in haemodialysis: the effect of ultrafiltration. Biopharmaceutics and Drug Disposition 7: 335–346, 1986
Laverdiere M, Welter D, Sabath LD. Use of a heavy inoculum in the in vitro evaluation of the anti-staphylococcal activity of 19 cephalosporins. Antimicrobial Agents and Chemotherapy 13: 669–675, 1978
Lee FH, Pfeffer M, Van Harken DR, Smyth RD, Hottendorf GH. Comparative pharmacokinetics of ceforanide (BL-S786R) and cefazolin in laboratory animals and humans. Antimicrobial Agents and Chemotherapy 17: 188–192, 1980
Lefrock JL, Holloway W, Carr BB, Schell RF. In vitro and clinical evaluation of ceforanide. American Journal of the Medical Sciences 287: 21–25, 1984
Leitner F, Misiek M, Pursiano TA, Buck RE, Chisholm DR, et al. Laboratory evaluation of BL-S786, a cephalosporin with broad-spectrum antibacterial activity. Antimicrobial Agents and Chemotherapy 10: 426–435, 1976
Lentnek A, Kidd L. Single dose ceforanide versus conventional dose ampicillin in the treatment of uncomplicated lower urinary tract infections. Abstract. Clinical Research 28: 373A, 1980
McMillan JA, Rodgers Jr GC, Weiner LB, Van Harken DR. Efficacy and pharmacokinetics of ceforanide in pediatric patients. Abstract. Pediatric Research 14: 469, 1980
Meyer RD. In vitro susceptibility of cephalothin-resistant enterobacteriaceae to cefoxitin and BL-S786. Journal of Antibiotics 30: 326–329, 1977
Miller KW, McCoy HG, Chan KKH, Fischer RP, Lindsay WG, et al. Effect of cardiopulmonary bypass on cefazolin disposition. Clinical Pharmacology and Therapeutics 27: 550–556, 1980
Mullany LD, French MA, Nightingale CH, Low HBC, Ellison LH, et al. Penetration of ceforanide and cefamandole into the right atrial appendage, pericardial fluid, sternum, and intercostal muscle of patients undergoing open heart surgery. Antimicrobial Agents and Chemotherapy 21: 416–420, 1982
Musher DM, Fainstein V, Young EJ. Treatment of cellulitis with ceforanide. Antimicrobial Agents and Chemotherapy 17: 254–257, 1980
Muytjens HL, van der Ros-van de Repe J. Comparative activities of 13 β-lactam antibiotics. Antimicrobial Agents and Chemotherapy 21: 925–934, 1982
Muytjens HL, van der Ros-van de Repe J. Comparative in vitro susceptibilities of eight enterobacter species, with special reference to enterobacter sakazakii. Antimicrobial Agents and Chemotherapy 29: 367–370, 1986
Nardella PA, Coyle C, Sutton F, Pfeffer M, Van Harken DR. Comparison of a microbiologic and HPLC assay for ceforanide, a new cephalosporin, in blood plasma. The American Chemical Society Northeast Regional Meeting, Potsdam, New York, 1980
National Committee for Clinical Laboratory Standards. Methods for dilution antimicrobial susceptibility tests for bacteria that grow aerobically. NCCLS Publication M7-A, 5(22), 1985
Parish LC, Witkowski JA. Ceforanide compared with cefazolin in skin and soft tissue infections. Therapeutics for the Clinician 33: 313–319, 1984
Perlino CA, Jurado R. Double-blind comparison of ceforanide vs. procaine penicillin treatment of pneumococcal pneumonia. Current Therapeutic Research 30: 271–276, 1981
Pfeffer M, Gaver RC, Van Harken DR. Human pharmacokinetics of a new broad-spectrum parenteral cephalosporin antibiotic, ceforanide. Journal of Pharmaceutical Sciences 69: 398–403, 1980
Platt R, Munoz A, Stella J, Van Devanter S, Koster Jr JK. Antibiotic prophylaxis for cardiovascular surgery. Annals of Internal Medicine 101: 770–774, 1984
Platt R, Rosner B. Effect of hydrogen ion and protein concentrations on the activity of beta-lactam antibiotics. Reviews of Infectious Diseases 6 (Suppl. 4): S829–834, 1984
Raab TA, Balderman S, Bhayana J, Bingham K, Mylotte J, et al. A comparison of the safety, efficacy, and distribution of ceforanide and cephalothin in coronary artery bypass graft surgery. Annals of Thoracic Surgery 33: 340–344, 1982
Randolph JA, Buck RE, Price KE, Leitner F. Comparative bactericidal effect of ceforanide (BL-S786) and five other cephalosporins in an in vitro pharmacokinetic model. Journal of Antibiotics 32: 727–733, 1979
Rawson D, Jones DS, Crain D, Perlino CA. Comparison of ceforanide and cefazolin treatment of bacterial pneumonia. In Nelson JD & Grass C (Eds) Current chemotherapy and infectious disease, Proceedings of the 11th International Congress of Chemotherapy and the 19th Interscience Conference on Antimicrobial Agents and Chemotherapy, Boston, 1–5 Oct, 1979. American Society for Microbiology, Washington DC, 1980
Richmond MH, Sykes RB. β-Lactamases of Gram-negative bacteria and their possible physiological role. Advances in Microbial Physiology 9: 31–88, 1973
Ripa S, La Rosa F, Ghezzi A, Prenna A, Pfeffer M. Pharmacokinetics of ceforanide. Antimicrobial Agents and Chemotherapy 21: 323–326, 1982a
Ripa S, La Rosa F, Prenna M, Ghezzi A. Ceforanide: human pharmacokinetics after i.v. injection. Drugs Under Experimental and Clinical Research 8: 269–273, 1982b
Ripa S, Prenna M, Bonomo A, Ghezzi A, Calio R. Laboratory evaluation of BL-S786 a new semisynthetic cephalosporin. Drugs Under Experimental and Clinical Research 5: 11–18, 1979
Sanders WE, Schneider N, Hartwig C, Cacciatore R, Valdez H. Comparative activities of cephalosporins against mycobacteria. In Nelson & Grassi (Eds) Current chemotherapy and infectious disease, Proceedings of the 11th International Congress of Chemotherapy and the 19th Interscience Conference on Antimicrobial Agents and Chemotherapy, Boston, Oct 1–5, 1979, Vol. II, pp. 1075-1077, American Society for Microbiology, Washington, D.C., 1980
Saravolatz LD, Lee C, Drukker B. Comparison of intravenous administration with intrauterine irrigation with ceforanide for nonelective cesarean section. Obstetrics and Gynecology 66: 513–516, 1985
Scheid WM, Van Ness MM, Sande MA. Therapy of experimental Staphylococcus aureus endocarditis: relative importance of serum bacterial activity. In Nelson JD & Grassi C (Eds) Current chemotherapy and infectious disease, Proceedings of the 11th International Congress of Chemotherapy and the 19th Interscience Conference on Antimicrobial Agents and Chemotherapy, Boston, 1–5 Oct, 1979. American Society for Microbiology, Washington DC, 1980
Shadomy S, Wagner G, Carver M. In vitro and in vivo studies with BL-S786, cefoxitin, and cefamandole. Antimicrobial Agents and Chemotherapy 13: 412–415, 1978
Sirinek KR, Dunkle L, Yamauchi T, Congeni B, Holcroft J. Single-dose preoperative antibiotic prophylaxis for acute nonperforated appendicitis. Advances in Therapy 3: 340–346, 1986
Smyth RD, Pfeffer M, Glick A, Van Harken DR, Hottendorf GH. Clinical pharmacokinetics and safety of high doses of ceforanide (BL-S786R) and cefazolin. Antimicrobial Agents and Chemotherapy 16: 615–621, 1979
Soave R, Hirsch JC, Salvati EA, Brause BD, Roberts RB. Comparison of ceforanide and cephalothin prophylaxis in patients undergoing total joint arthroplasty. Orthopedics 9: 1657–1660, 1986
Souney PF, Elboim S, Tuomala MD, Polk BF. Comparative uterine tissue levels of ceforanide and cefazolin: Departments of Pharmacy and Medicine, Brigham and Women’s Hospital, Boston. Drug Intelligence and Clinical Pharmacy 16: 481–482, 1982
Tartaglione TA, Polk RE. Review of the new second-generation cephalosporins: cefonicid, ceforanide, and cefuroxime. Drug Intelligence and Clinical Pharmacy 19: 188–198, 1985
Thirumoorthi MC, Buckley JA, Vincent CV, Kobos D, Dajani AS. Efficacy and safety of ceforanide in the treatment of childhood infections. Pediatric Infectious Disease 2: 377–380, 1983
Thrupp LD, Miller RD, Elliott J, Welch WD. Ceforanide dosage in the puerperium. Current Therapeutic Research 34: 130–135, 1983
Van Harken DR, Pfeffer M, McMillan JA, Rodgers Jr GC, Weiner LB. Pediatric pharmacokinetics of ceforanide following intravenous administration. 20th Interscience Conference on Antimicrobial Agents and Chemotherapy, New Orleans, Sept. 22–24, 1980. Abstract no. 646, 1980
Wallace RJ, Martin RR, Greenberg SB. Ceforanide (BL-S786) in the treatment of community-acquired bacterial pneumonia. Infection 7: 176–179, 1979
Wallace RJ, Martin RR, Quinones FJ, Greenberg SB. Ceforanide and cefazolin therapy of pneumonia: comparative clinical trial. Antimicrobial Agents and Chemotherapy 20: 648–652, 1981
Washington II JA. Discrepancies between in vitro activity of and in vivo response to antimicrobial agents. Diagnostic Microbiology and Infectious Disease 1: 25–31, 1983
Wasilauskas BL. Comparative in vitro activity of 5 cephalosporins with other antibiotics tested against 887 recent clinical isolates. Chemotherapy 27: 416–422, 1981
Weaver SS, LeBlanc BM, Bodey GP. BL-S786 (ceforanide), a new parenteral cephalosporin: in vitro studies. Antimicrobial Agents and Chemotherapy 15: 318–322, 1979
Williams DN, Gustilo RB, Beverly R, Kind AC. Bone and serum concentrations of five cephalosporin drugs. Clinical Orthopaedics and Related Research 179: 253–265, 1983
Williams RJ, Williams JD. The cephalosporin group of antibiotics. In Grineberg (Ed.) Antibiotics and chemotherapy: current topics, pp. 63-111, MTP Press Ltd, 1980
Author information
Authors and Affiliations
Additional information
Various sections of the manuscript reviewed by: A.L. Barry, The Clinical Microbiology Institute, Tualatin, Oregon, USA; R.L. Greenman, Department of Medicine, Division of Infectious Diseases, University of Miami, Miami, Florida, USA; D. Greenwood, Department of Microbiology, University Hospital, Queen’s Medical Centre, Nottingham, England; M. Inoue, Laboratory of Drug Resistance in Bacteria, Gunma University, School of Medicine, Maebashi, Gunma-ken, Japan; R. Janknegt, Medisch Centrum Alkmaar, Department of Clinical Pharmacy, Alkmaar, The Netherlands; T. Kesado, Institute of Anaerobic Bacteriology, Gifu University School of Medicine, Tsukasa-Machi, Gifu, Japan; R.W. Lacey, Department of Microbiology, University of Leeds, Leeds, England; L.A. Mandell, Division of Infectious Disease, McMaster University, McMaster Medical Unit, Henderson General Hospital, Hamilton, Ontario, Canada; N.E. Moller, Ronneboervej 10, Holte, Denmark; S.R. Norrby, Department of Infectious Diseases, University of Umeå, Umeå University Hospital, Umeå, Sweden; D. Pastel, Department of Pharmacy, Cedars-Sinai Medical Center, Los Angeles, California, USA; R.P. Rapp, College of Pharmacy, University of Kentucky, Lexington, Kentucky, USA; D.J. Winston, Los Angeles, California, USA.
‘Precef’ (Bristol Laboratories).
Rights and permissions
About this article
Cite this article
Campoli-Richards, D.M., Lackner, T.E. & Monk, J.P. Ceforanide. Drugs 34, 411–437 (1987). https://doi.org/10.2165/00003495-198734040-00001
Published:
Issue Date:
DOI: https://doi.org/10.2165/00003495-198734040-00001