Summary
Synopsis: Piretanide1 is a potent ‘loop’ diuretic whose principal site of action is in the thick ascending limb of the loop of Henle. When administered orally or intravenously to healthy volunteers it rapidly increases diuresis and electrolyte excretion, and the effects are short-lived. In comparative studies, piretanide has generally been found to be 5 to 7 times more potent than frusemide (furosemide) but only one-tenth as potent as bumetanide, on a weight-for-weight basis. Piretanide 6 to 12 mg/day, in conventional or sustained release formulations, has been shown to significantly lower elevated blood pressure in a large proportion of patients with mild to moderate hypertension. Comparative trials of up to 3 months duration indicate that at this dosage piretanide is of comparable antihypertensive efficacy as hydrochlorothiazide 50 to 100 mg/day, but has significantly less effect on serum potassium levels. Short term studies in patients with oedema caused by renal, hepatic or cardiac failure demonstrated that piretanide 6 to 9mg is of similar diuretic potency as frusemide 40mg and bumetanide lmg. In medium term trials in patients with congestive heart failure piretanide 6 mg/day produced equivalent symptomatic improvement as frusemide 40 mg/day. When used to treat oedema caused by liver disease, piretanide 12 to 24 mg/day was successful in only about 50% of patients, but spironolactone added to the treatment regimen greatly increased the response rate. Generally, piretanide has been well-tolerated in clinical trials, although the conventional tablet formulation has caused a relatively high incidence of acute adverse effects — these were greatly reduced with the introduction of the sustained release formulation. Serum concentrations of most electrolytes have not shown any consistent adverse trends and hyperuricaemia and hypokalaemia have been encountered infrequently.
Thus, piretanide appears to offer an effective alternative to other ‘loop’ diuretics for the treatment of oedematous diseases and to hydrochlorothiazide for the management of mild to moderate hypertension. However, its relative place in therapy remains to be clarified with wider clinical experience.
Pharmacodynamic Studies: Piretanide is a potent ‘loop’ diuretic with qualitative properties characteristic of this group of drugs and it has a weight-for-weight potency intermediate between bumetanide (the most potent) and frusemide. In healthy volunteers piretanide produces a rapid diuresis, natriuresis, chloruresis and kaliuresis (1 to 2 hours after oral administration and 0.25 to 1 hour after an intravenous injection), the effects of which are short-lived (oral: 4 to 6 hours; intravenous: 2 to 3 hours). At peak saluresis, piretanide reduced fractional free water clearance during hydration by almost 20% and reduced fractional free water reabsorption during hydropenia by approximately 70%. Piretanide has produced no consistent changes in renal plasma flow or glomerular filtration rate in studies in healthy volunteers or in patients with renal insufficiency.
Mean fractional sodium and chloride excretion were increased following piretanide administration by greater than 10-fold, while the fractional excretion of potassium was increased only 2- to 3-fold. Additionally, after single doses of piretanide 3, 6 and 12mg the diuretic and natriuretic effects were found to be dose-dependent whereas the differences in potassium excretion between these doses were not statistically significant.
Following 7 days of high dose piretanide administration (18 mg/day) no clinically significant changes in the plasma or tissue concentrations of sodium or chloride were documented, while plasma potassium concentrations were significantly reduced. However, this reduction was relatively small (from 3.76 to 3.41 mmol/L) and there was no significant change in the tissue concentration of potassium. Administration of a sustained release formulation of piretanide reduced peak diuresis and electrolyte excretion but produced a more prolonged duration of action — overall net salidiuretic effects were comparable with those of a conventional formulation of piretanide.
The urinary excretion of calcium and magnesium, as for sodium and chloride, were greatly increased by piretanide, but changes with respect to phosphate excretion have been conflicting, with both increases and decreases being reported. Similarly, no consistent changes in uric acid excretion have been observed in studies to date. Piretanide has generally had little influence on urinary pH, although in a small number of studies significant decreases have been observed. As a result of its diuretic activity piretanide lowered blood volume by approximately 5 to 10%, but the plasma concentration of antidiuretic hormone was not significantly affected.
In healthy volunteers piretanide 6mg once or twice daily was largely unaccompanied by counter-regulatory changes in the renin-angiotensin-aldosterone system. There is some evidence that plasma renin activity may be slightly increased in elderly hypertensive patients administered piretanide, but plasma aldosterone and catecholamine concentrations were not affected. In a clinical trial in patients with liver cirrhosis and ascites, piretanide produced a positive therapeutic response only in those patients with normal or slightly raised levels of plasma renin activity or plasma aldosterone.
Apart from its diuretic properties, piretanide has been shown to have extrarenal relaxant effects on vascular smooth muscle, which may contribute to its blood pressure lowering effects, and also fibrinolytic and antiplatelet properties similar to those of frusemide.However, the relative contribution of the extrarenal vascular effects of piretanide to its antihypertensive efficacy requires further evaluation.
Piretanide and other ‘loop’ diuretics have been shown to have ototoxic potential in animal experiments, but in the case of piretanide the clinical relevance of these findings awaits clarification, since audiometric testing has rarely been performed.
Pharmacokinetic Studies: When administered in a conventional tablet formulation piretanide 6mg produced peak plasma concentrations of 0.2 to 0.3 mg/L within 1.1 to 1.8 hours. Following 5 days’ treatment with piretanide 6 mg/day no evidence for drug accumulation was observed. Administration of sustained release piretanide 6mg resulted in a 2-fold increase in the time to achieve maximum plasma concentrations, which were reduced by about 50%. The volume of distribution following intravenous administration of piretanide (12mg) was approximately 0.3 L/kg, which indicates limited tissue distribution and is consistent with the high level of protein binding (> 90%) reported for piretanide. Renal clearance accounts for almost half of the total body clearance of 11 to 15 L/h in healthy subjects. Cumulative recovery of unchanged piretanide in the urine has been reported to be between 40 and 75%, with maximal recovery occurring in the first 2 hours. Five metabolites of piretanide have been isolated from the urine but only 1 of these has been identified to date. The elimination half-life of piretanide in healthy volunteers is approximately 1 to 1.7 hours.
In patients with chronic renal insufficiency, large oral doses of piretanide (18 to 96mg) resulted in peak plasma concentrations of 0.5 to 4.2 mg/L within 1 to 2 hours. There was no evidence of drug accumulation following repeat administration of a high dose of piretanide (18mg) 6 hours after the first dose. The volume of distribution of piretanide appears to be slightly increased (0.4 to 0.5 L/kg) and the extent of protein binding decreased (88%). Both the plasma and renal clearances of piretanide were markedly reduced in patients with renal failure, and the elimination half-life was increased to between 1.7 and 9.0 hours. In patients with congestive heart failure, piretanide pharmacokinetics were similar to those in healthy volunteers.
Therapeutic Trials: Clinical trials with piretanide have been performed in oedematous conditions caused by cardiac, renal or liver disease, and also in hypertension and angina. When used as a diuretic to relieve oedema the majority of trials have studied the effects of only a single dose of piretanide.
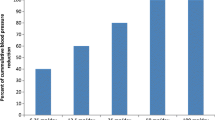
Dose-finding studies in patients with mild to moderate hypertension have shown that piretanide 6 to 12 mg/day (as either conventional tablets or sustained release capsules) effectively lowers blood pressure (10 to 30%) in a large proportion of patients (60 to 90%). Comparative trials of up to 3 months duration indicate that piretanide 6 to 12 mg/day is of comparable antihypertensive efficacy as hydrochlorothiazide 25mg or 50mg twice daily. In one study piretanide 6 or 12 mg/day reduced blood pressure by a significantly greater amount than a fixed combination of hydrochlorothiazide and amiloride (50mg + 5mg) twice daily. Other medium term comparative trials in patients with mild to moderate hypertension have shown that conventional piretanide and sustained release piretanide produce similar reductions in blood pressure, and that a combination of low dose piretanide (3mg) combined with penbutolol (20mg) is at least as effective as penbutolol 40mg and elicited a better global assessment both from patients and investigators.
In patients with congestive heart failure, piretanide 6 to 9mg administered orally has been found to be of comparable efficacy as bumetanide lmg, frusemide 40mg and hydrochlorothiazide 50mg, while doses of 6 to 12 mg/day were superior to chlorothiazide 1000 to 2000 mg/day. Generally, piretanide tended to produce a more intense acute diuresis but had equivalent 24-hour diuretic activity as frusemide and hydrochlorothiazide (alone or combined with amiloride). Following intravenous administration, piretanide 6 to 12mg produced similar changes to frusemide 20 to 40mg and both drugs at the higher doses were approximately 30% more effective than bumetanide lmg. Short term haemodynamic studies in patients with congestive heart failure demonstrated that the major changes produced by piretanide were reductions in pulmonary wedge, pulmonary arterial and right atrial pressures; however, small reductions in cardiac output have also been noted. The longest of these trials (3 weeks) found a good correlation between haemodynamic and echocardiographic changes and overall clinical response, although haemodynamic improvement usually exceeded diuretic activity by at least 15 hours. During medium term studies, piretanide 6 mg/day produced a similar reduction of the symptoms of congestive heart failure as frusemide 40 mg/day and piretanide plus triamterene (6mg + 50mg) once daily. Long term (up to 3 years) administration of piretanide (up to 24 mg/day) was generally well tolerated and controlled congestive heart failure in 15 patients with moderate to severe disease.
In patients with renal insufficiency the salidiuretic effects of piretanide seem to be less pronounced but generally more prolonged than in healthy subjects, the extent of the difference depending on the severity of renal impairment. Increased doses of piretanide are therefore required to obtain a clinically significant response in this type of patient. Raising the piretanide dosage 8-fold (48mg) only partially restored salidiuretic efficacy in patients with renal failure, to approximately 30% of its maximal effects at a dose of 6mg in patients with normal or slightly reduced glomerular filtration rates. Piretanide 18mg twice daily and bumetanide 3mg twice daily equally increased salidiuresis in patients with renal insufficiency, while in a single-dose study piretanide 12mg was marginally less effective than frusemide 80mg. Similarly, in a 3-day study, piretanide and frusemide, administered at a dose ratio of 6mg to 40mg (the dose administered being dependent upon the severity of renal failure), produced equivalent changes for most parameters, although urinary volume was increased fractionally more by frusemide. Piretanide has also been used in patients with stable renal transplants and a dose of 9mg was of similar efficacy as lmg of bumetanide over a period of 3 days. In a single-dose study in 6 patients with nephrotic syndrome, piretanide 12mg was more effective than frusemide 80mg during maximal water diuresis, but these dosages produced comparable changes during hydropenia.
Clinical trials with piretanide in patients with liver disease have been of short duration. Intravenous piretanide 6mg was found to be as effective as frusemide 20mg over a 3-day period in the largest study to date, and in a single-dose study in 6 patients with liver cirrhosis, intravenous piretanide 9mg induced diuretic effects comparable to bumetanide 0.5mg. However, in another trial, piretanide 12 to 24 mg/day administered orally to patients with liver cirrhosis and associated ascites produced a response rate of only about 50%. The number of patients responding to diuretic therapy was greatly increased by the addition of spironolactone 150 mg/day to their drug regimen. These data suggest that piretanide may not be a suitable first-line choice for patients with liver cirrhosis and ascites but may be useful in combination with spironolactone.
Side Effects: Short term trials indicate that piretanide has generally been well tolerated. Using a conventional tablet formulation in comparative studies the incidence of side effects has been the same or less than the other diuretics employed. However, in a 12-week open trial of 210 patients with mild to moderate hypertension, the incidence of some of the more common acute reactions such as pronounced diuresis (10%), nausea (5.2%), and thirst (4.8%) were more frequent than previously reported for other ‘loop’ diuretics such as bumetanide and frusemide. Recent data using a newer sustained release formulation of piretanide have shown a greatly decreased incidence of these acute side effects. There have been no consistent adverse changes in laboratory values. Serum levels of most important electrolytes have not significantly altered during clinical trials with piretanide and a low incidence of hypokalaemia has been recorded. In comparative trials in patients with congestive heart failure, renal or liver disease, piretanide has had no greater effect on potassium homeostasis than other diuretics such as bumetanide, frusemide, hydrochlorothiazide (alone or combined with amiloride), chlorothiazide, and triamterene. There is evidence that it has lesser kaliuretic potential than frusemide or chlorothiazide in patients with congestive heart failure, and than hydrochlorothiazide in patients with hypertension. Changes in blood uric acid concentrations have been variable, but isolated cases of hyperuricaemia have been noted. The effects of piretanide on carbohydrate metabolism appear to be minimal, with most studies reporting little change in blood glucose concentrations.
In a 12-week study in patients with mild to moderate hypertension, piretanide 12 mg/day was found to alter a number of indices of lipid metabolism and its effects were generally greater than those of hydrochlorothiazide (100 mg/day) despite having a lesser effect on potassium balance. However, in therapeutic trials published to date no clinically important changes in serum cholesterol or serum triglycerides have been documented.
Dosage and Administration: For the treatment of oedema in adults the recommended starting dose using conventional tablets is 6 mg/day. If necessary, this dosage may be increased. Results from patients with renal disease suggest that much greater dosages may be necessary (30 to 60mg) when glomerular filtration rates are severely reduced. Piretanide is also available as sustained release capsules (6mg); the recommended dosage with this formulation is 1 capsule daily, but further doses may be administered if necessary to achieve a desired level of diuresis. For the clinical management of mild to moderate hypertension in adults, the recommended starting dosage is 9 mg/day using conventional tablets or 1 to 2 sustained release capsules (6mg) daily. The maintenance dosage in adults is usually 1 capsule daily. At present insufficient data are available for dosage recommendations in children. The usual precautions for other ‘loop’ diuretics and for drugs which lower blood pressure apply to piretanide.
Similar content being viewed by others
References
Aiton, J.F.; Chipperfield, A.R.; Lamb, J.F.; Ogden, P. and Simmons, N.L.: Occurrence of passive furosemide-sensitive transmembrane potassium transport in cultured cells. Biochimica et Biophysica Acta 646: 389–398 (1981).
Aiton, J.F. and Simmons, N.L.: An effect of piretanide on the intracellular cations of cultured cells subjected to partial, chronic Na-K pump blockade. (Abstract.) 1st International Conference of Diuretics, Miami Beach, Florida, March 5–9, p. 27 (1984a).
Aiton, J.F. and Simmons, N.L.: An effect of piretanide upon the intracellular cation contents of cells subjected to partial chronic (Na-K) pump blockade by ouabain. Biochemical Pharmacology 33: 3425–3431 (1984b).
Alvestrand, A. and Bergström, J.: The effect of HOE 118 (Piretanide) on muscle electrolytes in normal subjects. Current Therapeutic Research 25: 786–790 (1979).
Arroyo, V.; Bosch, J.; Casamitjana, R.; Cabrera, J.; Rivera, F. and Rodés, J.: Use of piretanide, a new loop diuretic, in cirrhosis with ascites. Relationship between the diuretic response and the plasma aldosterone level. Gut 21: 855–859 (1980).
Bailey, R.R.: Open comparison of diuretic effects of piretanide and bumetanide in patients with stable renal transplants. New Zealand Medical Journal 95: 44–45 (1982).
Bauer, U.; Frick, G.; Mouselimis, N.; Cernoch, K. and Fehske, K.H.: Akuter und chronischer effekt von piretanid auf die hä-modynamik herzinsuffizienter patienten mit koronarer herzerkrankung. Herz Kreislauf Zeitschrift für Kardiologie und Angiologie in Klinik und Praxis 15: 493–500 (1983).
Baylis, P.H. and De Beer, F.C.: Human plasma vasopressin response to potent loop-diuretic drugs. European Journal of Clinical Pharmacology 20: 343–346 (1981).
Berg, K.J.; Walstad, R.A. and Bergh, K.: The pharmacokinetics and diuretic effects of piretanide in chronic renal insufficiency. British Journal of Clinical Pharmacology 15: 347–353 (1983).
Biamino, G.: Vergleichende Untersuchungen über die Wirkung der diuretika furosemid (F), bumetanide (B) and Piretanide (P) (HOE 118) auf die isolierte gefäßmuskulatur von ratten. Zeitschrift für Kardiologie 67: 195 (1978).
Brater, D.C.; Anderson, S.; Baird, B. and Kaojarern, S.: Effects of piretanide in normal subjects. Clinical Pharmacology and Therapeutics 34: 324–330 (1983).
Brazier, J.L.; Pozet, N.; Faucon, G.; Traeger, J. and Hadj-Haissa, A.: Kinetics of a high dose of piretanide in renal failure. European Journal of Clinical Pharmacology 21: 307–310 (1982).
Brooks, B.A. and Lant, A.F.: The use of the human erythrocyte as a model for studying the action of diuretics on sodium and chloride transport. Clinical Science and Molecular Medicine 54: 679–683 (1978).
Brown, C.D.A. and Simmons, N.L.: A comparison of the actions of certain loop diuretics upon passive potassium fluxes and chloride secretion in a cultured epithelium of renal origin. Journal of Physiology 322: 25P (1981).
Brown, R.D.: Comparisons of the acute effects of intravenous furosemide and bumetanide on the cochlear action potential (N,) and on the AC cochlear potential (CM) at 6kHz in cats, dogs and guinea pigs. Scandinavian Audiology (Suppl. 14): 71–83 (1981a).
Brown, R.D.: Comparative acute cochlear toxicity of intravenous bumetanide and furosemide in the purebred Beagle. Journal of Clinical Pharmacology 21: 620–627 (1981b).
Brunkhorst, R.; Müller-Ott, K.; Gutsche, H.U. and Niedermayer, W.: Effect of furosemide, bumetanide and piretanide on the sensor of the tubuloglomerular feedback mechanism. Proceedings of the European Dialysis Transplantation Association 15: 613–616 (1978).
Buckert, C; Muhlhausler, W.; Fratzer, U. and Verho, M.: A double-blind multicentre study of piretanide and hydrochlorothiazide in patients with essential hypertension. Journal of International Medical Research 12: 81–86 (1984).
Chaffman, M.; Heel, R.C.; Brogden, R.N.; Speight, T.M. and Avery, G.S.: Indapamide: A review of its pharmacodynamic properties and therapeutic efficacy in hypertension. Drugs 28: 189–235 (1984).
Chohan, I.S.: Piretanide: A new synthetic fibrinolytic and antiplatelet agent. Haemostasis II: 87 (1982).
Comis, S.D.; Leng, G. and Pratt, S.R.: The effects of frusemide, bumetanide and piretanide on the guinea pig cochlea and auditory nerve. Scandinavian Audiology (Suppl. 14): 85–93 (1981a).
Comis, S.D. and Pratt, S.R.: The effect of sulfamyl loop diuretics on crossed olivo-cochlear bundle. Neuropharmacology 18: 739–741 (1979).
Comis, S.D.; Pratt, S.R. and Hayward, T.L.: The effect of frusemide, piretanide and bumetanide on cochlear succinic dehydrogenase. Neuropharmacology 20: 405–407 (1981b).
Cook, J.J.; Buchan, S.; Rooke, T. and Bailey, R.R.: Piretanide (HOE 118): A new potent diuretic: Preliminary Communication. New Zealand Medical Journal 89: 87 (1979).
Daneshmend, T.K.; Homeida, M.; Roberts, C.J.C. and Dombey, S.: The effects of a combination of piretanide and triamterene in healthy subjects. British Journal of Clinical Pharmacology 11: 106–108 (1981).
Deetjen, P.; Greger, R.; Lang, F.; Oberleithner, H. and Silbernagl, S.: Piretanide, a new high ceiling diuretic. Seventh International Congress of Nephrology p. R–l (1978).
De La Paz, A.G.; Rosario, N.U. and Garduno, M.D.: Piretanide in the treatment of hypertension. Asian-Pacific Congress of Cardiology VIII: 257 (1983).
Diehm, C. and Mörl, H.: Long term study of piretanide in man. (Abstract.) Presented at the IX European Congress of Cardiology, Düsseldorf, 11 July (1984).
Doglio, G.R.; Pusajo, J.F.; Bonfigli, G.; Hualde, R.; Lafourcade, E.S. and Maldonado, H.G.O.: Effecto de un diuretico de asa, la piretanida, en la insuficiencia cardiaca grave. Medicina (Buenos Aires) 44: 53–58 (1984).
Dois, W.; Freude, J.; Verho, M. and Rangoonwala, B.: A double-blind study of piretanide in the treatment of hypertension. Current Medical Research and Opinion 8: 649–658 (1983).
Dois, W.; Verho, M.; Rangoonwala, B. and Jaeger, S.: Piretanide — a potassium stable diuretic — in the treatment of essential hypertension: A double blind comparison of two formulations. Journal of International Medical Research 13: 31–39 (1985).
Dorian, B.; Larrue, J.; Defeudis, F.V.; Salari, H.; Borgeat, P. and Braquet, P.: Activation of prostacyclin synthesis in cultured aortic smooth muscle cells by diuretic-antihypertensive drugs. Biochemical Pharmacology 33: 2265–2269 (1984).
Drebinger, K.; Fehske, K.J. and Wörz, K.: Piretanide drug surveillance study in 5000 patients with essential hypertension. (Abstract.) Presented at the IX European Congress of Cardiology, Düsseldorf 11 July (1984).
Elliott, H.L.; Ansari, A.F.; Campbell, B.C. and Lawrence, J.R.: Protein binding of piretanide in normal and uraemic serum. European Journal of Clinical Pharmacology 21: 311–313 (1982).
Ellory, J.C. and Stewart, G.W.: The human erythrocyte Cl-dependent Na-K cotransport system as a possible model for studying the action of loop diuretics. British Journal of Pharmacology 75: 183–188 (1982).
Farmer, J.A.; O’Meara, M.E.; Fennell, W.H.; Luck, J.C; Waggoner, P.C. and Murray, M.: Piretanide: A potent new diuretic for heart failure. Clinical Research 30: 838A (1982).
Fehske, K.J. and Drebinger, K.: Bedürfen Diuretika in der Hypertonie — therapie immer einer Kombination mit einem ‘Kaliumsparer’? Therapiewoche 33: 6165–6176 (1983).
Frölich, J.C.; Filep, J.; Kroemer, H. and Fejes-Toth, G.: Extrarenal effects of diuretics — Are prostanoids involved? (Abstract.) 1st International Conference of Diuretics, Miami Beach, Florida, March 5–9 (1984).
Geck, P.: Sodium-potassium-chloride cotransport: A target system for inhibition by high-ceiling diuretics. Kidney International 17: 405 (1980).
George, C.R.P.: Piretanide. Effective diuretic in renal failure. Medical Journal of Australia 1: 83–84 (1981).
Gillies, A.; Morgan, T. and Myers, J.: Comparison of piretanide and chlorothiazide in the treatment of cardiac failure. Medical Journal of Australia 1: 170–172 (1980).
Glasson, Ph.; Dayer, P.; Favre, H. and Leski, M.: Piretanide-kinetics and therapeutic effect in end stage renal disease patients. World Conference on Clinical Pharmacology and Therapeutics II: 42 (1983).
Graml, E.; Verho, M; Rangoonwala, B.; Englert, R.; Häringer, E. and Pahnke, K.: Efficacy of penbutolol and a combination of a low dose of penbutolol with piretanide in the treatment of mild to moderate hypertension. Pharmatherapeutica (In press, 1985).
Greger, R. and Schlatter, E.: Presence of luminal K+, a prerequisite for active NaCl transport in the cortical thick ascending limb of Henle’s loop of rabbit kidney. Pflügers Archiv 392: 92–94 (1981).
Greger, R. and Schlatter, E.: Cellular mechanism of the action of loop diuretics on the thick ascending limb of Henle’s loop. Klinische Wochenschrift 61: 1019–1027 (1983).
Grigoleit, H.-G.; Meyer, B.H. and Müller, F.O.: Electrolyte clearance studies in healthy subjects with piretanide and triamterene. World Conference on Clinical Pharmacology and Therapeutics II: 42 (1983).
Gupta, P.S. and Dave, M.L.: Piretanide (HOE 118): A new diuretic in essential hypertension. Current Therapeutic Research 30: 468–476 (1981).
Gutsche, H.-U.; Müller-Ott, K.; Brunkhorst, R. and Niedermayer, W.: Dose-related effects of furosemide, bumetanide, and piretanide on the thick ascending limb function in the rat. Canadian Journal of Physiology and Pharmacology 61: 159–165 (1983).
Hadj-Aissa, A.H.; Pozet, N.; Labeeuw, M.; Pellet, M. and Traeger, J.: Acute effect of a high dose (48mg) of piretanide in advanced renal insufficiency. British Journal of Clinical Pharmacology 1: 63–68 (1981).
Halstenson, C.E. and Matzke, G.R.: Bumetanide: A new loop diuretic. Drug Intelligence and Clinical Pharmacy 17: 786–797 (1983).
Handa, M.; Kondo, K. and Saruta, T.: Effects of diuretics on the vasoconstrictor responses to norepinephrine and potassium ions in the rat mesenteric artery. Archives Internationales de Pharmacodynamie et de Therapie 262: 124–131 (1983).
Harno, K.; Välimäki, M. and Verho, M.: Effects of a new diuretic piretanide on glucose tolerance, insulin secretion and 125I-insulin binding. European Journal of Clinical Pharmacology 27: 697–700 (1985).
Henderson, I.S.; Beattie, T.J.; Kennedy, A.C. and Dombey, S.L.: Oral piretanide in chronic renal failure. British Journal of Clinical Pharmacology 14: 857–860 (1982).
Heptner, W.; Baudner, S.; Dagrosa, E.E.; Hellstern, C; Irmisch, R.; Strecker, H. and Wissman, H.: A radioimmunoassay to measure piretanide in human serum and urine. Journal of Immunoassay 5: 13–27 (1984).
Heuer, V.L.J.; Machnik, H.; Studt, J. and Fink, R.: Klinische vergleichsprüfung von piretanid und furosemid bei patienten mit fortgeschrittener niereninsuffizienz. Wirkstoffspiegel und diuretische potenz einer oralen einzldosis. Arzneimittel-Forschung 33: 1318–1321 (1983).
Hoffmann, J.; Day, U.-H.; Fehske, K.J.; Garanin, G. and Lindner, U.: Ansprechquote der essentiellen Hypertonie gegenüber einem vasoaktiven Diuretikum. Medizinische Welt 35: 835–841 (1984).
Homeida, M.; Roberts, C.J.C. and Dombey, S.L.: A single-dose comparison of piretanide and bumetanide in congestive cardiac failure. British Journal of Clinical Pharmacology 8: 173–178 (1979).
Hropot, M.; Fowler, N. and Giebisch, G.: Effects of several diuretics and their combinations upon distal tubular H2O, Na, K, NH and titratable acid (TA) transport. Kidney International 14: 762 (1978).
Hropot, M.; Muschaweck, R.; Klaus, E.; Roller, B. and Schrader, R.: Effect of indomethacin on diuretic-induced natriuresis and renin release in dogs. Naunyn-Schmiedeberg’s Archives of Pharmacology 324 (Suppl.): 217 (1983).
Hropot, M.; Sörgel, F.; Muschaweck, R. and Giebisch, G.: Pharmacodynamic and pharmacokinetic studies with several diuretics: Micropuncture and clearance experiments in rats. Naunyn-Schmiedeberg’s Archives of Pharmacology 308 (Suppl.): R26 (1979).
Imbs, J.L.; Schmidt, M.; Spriet, A. and Schwartz, J.: Variance comparison as a tool in clinical pharmacology: An example with two loop diuretics. European Journal of Clinical Pharmacology 23: 221–224 (1982).
Klaus, E.; Alpermann, H.G.; Caspritz, G.; Linz, W. and Schölkens, B.: Vascular effects of piretanide. Studies on extrarenal action in several animal models. Arzneimittel-Forschung 33: 1273–1276 (1983).
Klinke, R.; Göttl, K.-H. and Roesch, A.: Ototoxic side effect of loop diuretics: Furosemide, piretanide, bumetanide, azosemide and ozolinone in the cat. Naunyn-Schmiedeberg’s Archives of Pharmacology 322: R117 (1983).
Kondo, T. and Ishikawa, Y.: Diuretic activity of piretanide — intravenous dose equivalence of piretanide injection to bumetanide injection in liver cirrhosis patients. Japanese Journal of Clinical Pharmacology 11: 141–151 (1980).
Kondo, T.; Nagashima, H. and Nakashima, M.: The diuretic effects and safety of piretanide in liver cirrhosis — a double-blind intergroup comparison study. Rinsho Yakuri 12: 73–97 (1981).
Kostis, J.B.; Cosgrove, N.; Krieger, S.; Moreyra, A.E.; Baumgardner, S. and Weinstein, J.: Comparison of piretanide and hydrochlorothiazide in congestive heart failure. Clinical Research 30: 198A (1982).
Krüger, B.; Sörgel, F. and Koob, R.: Intraindividual comparison of three loop diuretics in patients with congestive heart failure (CHF). Naunyn-Schmiedeberg’s Archives of Pharmacology (Suppl.): R130 (1983).
Kume, S.; Yamanaka, M.; Kariya, T.; Tanabe, A.; Iwamoto, A.; Ozaki, Y. et al.: Effect of the new diuretic piretanide on platelet function. Presented at the 43rd Congress of the Japan Hematological Society held in Nagoya, April 9–11 (1983).
Lant, A.F.: Modern diuretics and the kidney. Journal of Clinical Pathology 34: 1267–1275 (1981).
Lant, A.F.: Diuretics: Clinical pharmacology and therapeutic use (Part 1). Drugs 29: 57–87 (1985a).
Lant, A.F.: Diuretics: Clinical pharmacology and therapeutic use (Part II). Drugs 29: 162–188 (1985b).
Lawrence, J.R.; Ansari, A.F.; Elliott, H.L.; Sumner, D.J.; Brunton, G.F.; Whiting, B. and Whitesmith, R.: Kinetic and dynamic comparison of piretanide and furosemide. Clinical Pharmacology and Therapeutics 23: 558–565 (1978).
Leary, W.P. and Reyes, A.J.: Piretanide in the treatment of hypertension. Effects on arterial blood pressure and several blood variables. South African Medical Journal 60: 925–928 (1981).
Leary, W.P.; Reyes, A.J.; Venter, C.P. and Blake, G.T.W.: Mathematical description of diuresis after oral administration of piretanide, furosemide and placebo to healthy adults. Current Therapeutic Research 29: 249–252 (1981).
Marone, C. and Reubi, F.C.: Effects of a new diuretic (piretanide) compared with furosemide on renal diluting and concentrating mechanisms in patients with the nephrotic syndrome. European Journal of Clinical Pharmacology 17: 165–171 (1980).
Marone, C; Reubi, F.C. and Lahn, W.: Comparison of the short-term effects of the loop diuretic piretanide and furosemide in patients with renal insufficiency. European Journal of Clinical Pharmacology 26: 413–418 (1984a).
Marone, C; Reubi, F.C; Perisic, M. and Lahn, W.: Pharmacokinetics of high doses of piretanide in moderate to severe renal failure. European Journal of Clinical Pharmacology 27: 589–593 (1984b).
Marsh, J.D.; Nesto, R.; Glynn, M.A. and Smith, T.W.: Comparison of intravenous piretanide and furosemide in patients with congestive heart failure. Journal of Cardiovascular Pharmacology 4: 949–954 (1982).
Mautalen, C; Casco, C; Massironi, C. and Ghiringhelli, G.: The effect of piretanide upon the urinary excretion of calcium and magnesium in normal subjects and hypoparathyroid patients. Urological Research 12: 65 (1984).
McNabb, W.R.; Noormohamed, F.H.; Brooks, B.A. and Lant, A.F.: Renal actions of piretanide and three other ‘loop’ diuretics. Clinical Pharmacology and Therapeutics 35: 328–337 (1984a).
McNabb, W.R.; Noormohamed, F.H. and Lant, A.F.: Acute and chronic effects of piretanide in congestive cardiac failure; in Puschett, J.R. and Greenberg, A. (Eds) Diuretics, Chemistry, Pharmacology and Clinical Applications, pp. 355–357 (Elsevier, New York 1984b).
Merkel, W.; Bormann, D.; Mania, D.; Muschawek, R. and Hropot, M.: Piretanide (HOE 118) a new high ceiling salidiuretic. European Journal of Medicinal Chemistry-Chimica Therapeutica 11: 399–406 (1976).
Meyer, B.H.; Müller, F.O.; Grigoleit, H.-G. and Dagrosa, E.E.: Pharmacodynamics and urine pharmacokinetics of three doses of piretanide. European Journal of Clinical Pharmacology 25: 783–785 (1983).
Meyer, B.H.; Müller, F.O.; Grigoleit, H.-G. and Heptner, W.: Endoscopie placement in volunteers to investigate site of absorption of piretanide: Preliminary report. IRCS Medical Science: Clinical Pharmacology and Therapeutics 1: 14 (1982).
Meyer, B.H.; Müller, F.O.; Werkman, I.M.; Luus, H.G.; Lötter, M.G. and Grigoleit, H.-G.: Effects of piretanide on potassium balance in hypertension. South African Medical Journal 67: 206–208 (1985).
Miyamoto, M.; Nakanishi, Y.; Amamiya, H.; Takahashi, F.; Yoshizumi, K. and Arai, J.: Clinical effect and mechanism of a new diuretic: Piretanide. Japanese Journal of Clinical Pharmacology 10: 13–19 (1979).
Morgant, C; Contrepois, A.; Chau, N.P.; Romaru, A.; Fourtillan, J.-B. and Carbon, C: Effects of furosemide, piretanide and water loading on urinary excretion of cefazolin in humans. Antimicrobial Agents and Chemotherapy 25: 618–621 (1984).
Müller, F.O.; Meyer, B.H.; de Waal, A.; van Reenen, O.R. and Grigoleit, H.-G.: Potassium balance in piretanide and digoxin treatment. Clinical Pharmacology and Therapeutics 31: 339–342 (1982).
Müller, F.O.; Meyer, B.H.; Grigoleit, H.-G.; Heptner, W.; Malerczyk, V. and Merkel, W.: Multiple dose comparison of two oral formulations of the salidiuretic piretanide. Japanese Journal of Pharmacology 15: 463–468 (1984).
Muller-Ott, K; Brunkhorst, R.; Gutsche, H.U. and Niedermayer, W.: Effect of piretanide (HOE 118) on the electrolyte transport of thick ascending limbs of rat nephrons. Seventh International Congress of Nephrology p. R–ll (1978).
Nechwatal, W.; Stange, A.; Siegel, H.; Kress, P.; Resch, M. and Stauch, M.: Der Einfluss von Piretanid auf die zentrale Hä-modynamik und Belastungstoleranz von Patienten mit Angina pectoris. Herz Kreislauf 2: 91–96 (1982).
Odlind, B.: Determinants of access of diuretics to their site of action. Federation Proceedings 42: 1703–1706 (1983).
Odlind, B.: Relation between renal tubular secretion and effects of five loop diuretics. Journal of Pharmacology and Experimental Therapeutics 211: 238–244 (1979).
Odlind, B.; Beermann, B.; Selén, G. and Persson, A.E.G.: Renal tubular secretion of piretanide and its effects on electrolyte reabsorption and tubuloglomerular feedback mechanism. Journal of Pharmacology and Experimental Therapeutics 225: 742–746 (1983).
Omosu, M.; Fujimoto, K.; Sakaguchi, T.; Yamamoto, K.; Iwao, H. and Abe, Y.: Renal effects of piretanide in dogs. Arzneimittel-Forschung 33: 1277–1280 (1983).
Palfrey, H.C.; Feit, P.W. and Greengard, P.: cAMP-stimulated cation cotransport in avian erythrocytes: Inhibition by ‘loop’ diuretics. American Journal of Physiology: Cell Physiology 238: C139–C148 (1980).
Pellet, M.; Hadj-Aissa, A.; Pozet, N. and Rupp, W.: Activity of a new high ceiling diuretic in man. Seventh International Congress of Nephrology p. R–10 (1978).
Peterson, J.; Whiting, P.H. and Simpson, J.G.: Protective effect of diuretics in gentamicin induced nephrotoxicity. Kidney International 16: 235 (1979).
Pozet, N.; Hadj-Aissa, A. and Pellet, M.: Activity of a new high efficiency diuretic in man: Piretanide (HOE 118). British Journal of Clinical Pharmacology 9: 577–583 (1980).
Reyes, A.J.; Leary, W.P. and Venter, C.P.: Mathematical evaluation of the effects of piretanide, furosemide and placebo in healthy adults. Current Therapeutic Research 29: 253–259 (1981).
Roberts, C.J.C.; Homeida, M.; Roberts, F. and Bogie, W.: Effects of piretanide, bumetanide and frusemide on electrolyte and urate excretion in normal subjects. British Journal of Clinical Pharmacology 6: 129–133 (1978).
Rudofsky, G.: Venenverschlußplethysmographische Untersuchungen mit Vasodilatantien und Diuretika. Medizinische Klinik 1: 31–32 (1983).
Rudofsky, G.: Venenerweiternde Wirkung von Vasodilatantien und Diuretika. Medizinische Klinik 79: 607–609 (1984).
Saruta, T. and Kato, E.: Diuretic effect of piretanide (HOE 118) — An examination of its dose equivalent to furosemide. Japanese Journal of Clinical Pharmacology 10: 395–405 (1979).
Saruta, T. and Kato, E.: The diuretic effect of piretanide in man. Arzneimittel-Forschung 30: 1807–1812 (1980).
Schlatter, E.; Greger, R. and Weidtke, C: Effect of ‘high ceiling’ diuretics on active salt transport in the cortical thick ascending limb of Henle’s loop of rabbit kidney. Correlation of chemical structure and inhibitory potency. Pflügers Archiv 396: 210–217 (1983).
Schuster, P.; Wiechmann, H.W. and Trieb, G.: Use of piretanid in hypertensive patients with left ventricular compliance disorder — A hemodynamically controlled trial. (Abstract.) Presented at the IX European Congress of Cardiology, Düsseldorf, 11 July (1984).
Sharma, S.D.; Mehra, A.D. and Vakil, B.J.: Piretanide (HOE 118): Single dose studies in cardiac and hepatic oedema. World Conference on Clinical Pharmacology and Therapeutics (1980).
Stremme, D.; Leps, L. and Bruhn, H.D.: Einfluss von Piretanid (Arelix®) auf das Fibrinolyse- und Plättchensystem. Therapiewoche 34: 4155–4158 (1984).
Teredesai, P. and Puschett, J.B.: Acute effects of piretanide in normal subjects. Clinical Pharmacology and Therapeutics 25: 331–339 (1979).
Unger, T.; Fleck, T.; Ganten, D. and Lang, R.E.: Antihypertensive action of chronic treatment with piretanide in spontaneously hypertensive rats. (Abstract.) 1st International Conference of Diuretics, Miami Beach, Florida, March 5–9 (1984).
Valette, H.; Hébert, J.L. and Apoil, E.: Acute haemodynamic effects of a single intravenous dose of piretanide in congestive heart failure. European Journal of Clinical Pharmacology 24: 163–167 (1983).
Valette, H.; Hébert, J.L.; Raffestin, B.; Lockhart, A. and Apoil, E.: Comparison of haemodynamic effects of furosemide and piretanide in normovolemic patients. Journal of Cardiovascular Pharmacology 2: 103–111 (1980).
Valette, H.; Duhaze, P. and Apoil, E.: Plethysmographic study with frusemide and piretanide in healthy volunteers. British Journal of Clinical Pharmacology 7: 219 (1979).
Valette, H. and Apoil, E.: Interaction between salicylate and two loop diuretics. British Journal of Clinical Pharmacology 8: 592–594 (1979).
Välimäki, M.; Harno, K. and Nikkilä, E.A.: Serum lipoproteins and indices of glucose tolerance during diuretic therapy: A comparison between hydrochlorothiazide and piretanide. Journal of Cardiovascular Pharmacology 5: 525–530 (1983).
Verho, M.; Bückert, G; Freude, J.; Jaeger, S. and Pahnke, K.: The effects of piretanide on serum electrolytes in patients with congestive heart failure. A double-blind comparison with piretanide alone and in combination with triamterene. (Abstract.) 5th International Meeting of Pharmaceutical Physicians in Munich, October 14–17 (1984a).
Verho, M.; Heintz, B.; Nelson, K. and Kirsten, R.: The effects of piretanide on catecholamine metabolism, plasma renin activity and plasma aldosterone: A double-blind study against furosemide in healthy volunteers. Current Medical Research and Opinion (In press, 1985a).
Verho, M.; Kirsten, R. and Bückert, C: The effects of piretanide on catecholamine metabolism, plasma renin activity and serum aldosterone: A double-blind pilot comparison against hydrochlorothiazide in patients with essential hypertension. Pharmatherapeutica 3: 595–606 (1984b).
Verho, M.; Rangoonwala, B. and Dols, W.: Single and divided daily dose piretanide in the treatment of uncomplicated essential hypertension: A double-blind comparison with a combination of hydrochlorothiazide and amiloride. (Submitted for publication 1985b).
Verho, M.; Rangoonwala, B.; Dols, W.; Fratzer, U.; Kaczynski, J. and Mühlhäusler, W.: Piretanide, a potassium stable diuretic, in the treatment of essential hypertension. European Journal of Clinical Pharmacology 27: 407–414 (1984c).
Ward, A. and Heel, R.C.: Bumetanide: A review of its pharmacodynamic and pharmacokinetic properties and therapeutic use. Drugs 28: 426–464 (1984).
Whiting, P.H.; Barber, H.E. and Peterssen, J.: The effect of frusemide and piretanide on the renal clearance of gentamicin in man. British Journal of Clinical Pharmacology 12: 795–799 (1981).
Widdicombe, J.H.; Nathanson, I.T. and Highland, E.: Effect of ‘loop’ diuretics on ion transport by dog trachealepithelium. American Journal of Physiology 245: C388–C396 (1983).
Winaver, J.; Sylk, D.B.; Teredesai, P.R.; Robertson, J.S. and Puschett, J.B.: Dissociative effects of piretanide on proximal tubular PO4 and HCO3 transport. American Journal of Physiology 238: F60–F68 (1980).
Zidek, W.; Losse, H.; Schmidt, W.; Fehske, K.J. and Vetter, H.: Intracellular electrolytes during antihypertensive treatment with a loop diuretic. Journal of Hypertension 2: 393–395 (1984).
Author information
Authors and Affiliations
Additional information
‘Arelix’ (Cassella-Riedel/Hoechst).
Various sections of the manuscript reviewed by: R.R. Bailey, Department of Nephrology, Christchurch Hospital, Christchurch, New Zealand; D.C. Brater, Department of Pharmacology, University of Texas Health Center, Dallas, USA; W. Flamenbaum, Division of Nephrology and Hypertension, Beth Israel Medical Center, New York, USA; C.R.P. George, Concord Hospital Renal Unit, Concord, Australia; R. Greger, Max-Planck-Institut Für Biophysik, Frankfurt, West Germany; D.E. Hutcheon, Department of Pharmacology, University of Medicine and Dentistry of New Jersey, Newark, New Jersey, USA; A.C. Kennedy, Department of Medicine, University of Glasgow, Glasgow, Scotland; A.T. Lambie, Department of Medicine, University of Edinburgh, Edinburgh, Scotland; A.F. Lant, Department of Therapeutics, Charing Cross and Westminster Medical School, London, England; W.P. Leary, Department of Clinical and Experimental Pharmacology, University of Natal Medical School, Durban, South Africa; B.H. Meyer, Department of Pharmacology, University of the Orange Free State, Bloemfontein, South Africa; T.O. Morgan, Department of Physiology, University of Melbourne, Parkville, Australia; J.S. Robson, Department of Medicine, University of Edinburgh, Edinburgh, Scotland; H. Valette, Laboratoire de Physiologie et d’Explorations cardio-pulmonaires, Université Paris-Sud, Hôpital de Bicêtre, Kremlin-Bicêtre, France.
Rights and permissions
About this article
Cite this article
Clissold, S.P., Brogden, R.N. Piretanide. Drugs 29, 489–530 (1985). https://doi.org/10.2165/00003495-198529060-00002
Published:
Issue Date:
DOI: https://doi.org/10.2165/00003495-198529060-00002