Summary
We report the use of cefoperazone in 62 cases of serious infection, most of which occurred in patients with renal impairment. 43 severe or complicated urinary tract infections, 11 cases of pneumonia and 8 with other severe sepsis were treated with cefoperazone 1 to 2g twice daily usually for 5 to 10 days.
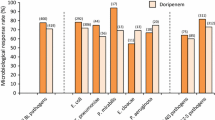
Of the patients with urinary tract infection, all who were symptomatic showed a rapid clinical response; 26 (61%) were cured including 11 of 16 with chronic renal failure; 12 relapsed and 5 were reinfected with a different pathogen. All of these patients were infected by organisms sensitive to cefoperazone by disc testing but in 5 of those who relapsed the cefoperazone MIC was in fact ⩾50µg/ml.
Ten of 11 cases with radiologically confirmed pneumonia were cured with cefoperazone. 7 episodes of pneumonia were in patients with end-stage chronic renal failure (6 were on dialysis) and 1 was in a patient with acute renal failure.
Seven of 8 cases with severe sepsis were cured with cefoperazone. 1 patient was withdrawn from the study when acute bronchospasm followed a 2g intravenous dose. 2 of the successfully treated patients had functioning renal transplants, 2 of 3 with severe chronic renal failure were on dialysis and 1 had acute renal failure.
Side effects included minor disturbances of liver function in 6 patients (11%), diarrhoea in 7 (13 % ) and marked alcohol intolerance in one. 4 patients with chronic renal failure developed a coagulation disorder which was corrected with vitamin K. None of the patients showed deterioration in renal function while receiving cefoperazone.
Cefoperazone promises to be an effective drug for the treatment of a wide spectrum of severe infections in hospitalised patients including those with impaired renal function.
Similar content being viewed by others
References
Bailey, R.R.; Peddie, B. and Blake, E.: Serum and urine levels of cefoperazone in severe chronic renal failure: Single and multiple dose studies. Second International Symposium on Cefoperazone Sodium; Hamburg, August (1980).
Hooper, C.A.; Haney, B.B. and Stone, H.H.: Gastrointestinal bleeding due to vitamin K deficiency in patients on parenteral cefamandole. Lancet 1: 39–40 (1980).
Jones, R.N.; Fuchs, P.C.; Barry, A.L.; Gavan, T.L.; Sommers, H.M. and Gerlach, E.H.: Cefoperazone (T-1551), a new semi-synthetic cephalosporin: Comparison with cephalothin and gentamicin. Antimicrobial Agents and Chemotherapy 17: 743–749 (1980).
Lahon, H.: A review of cefoperazone in clinical use. Second International Symposium on Cefoperazone Sodium; Hamburg, August (1980).
Mashimo, K. and Kunii, O.: Clinical trials with cefoperazone in the field of internal medicine in Japan. Clinical Therapeutics 3: 159–172 (Special Issue, 1980).
Matsubara, N.; Minami, S.; Muraoka, T.; Saikawa, I. and Mitsuhashi, S.: In vitro antibacterial activity of cefoperazone (T-1551), a new semisynthetic cephalosporin. Antimicrobial Agents and Chemotherapy 16: 731–735 (1979).
Neu, H.C. and Prince, A.S.: Interaction between moxalactam and alcohol. Lancet 1: 1422 (1980).
Reddy, J. and Bailey, R.R.: Vitamin K deficiency developing in patients with renal failure treated with cephalosporin antibiotics. New Zealand Medical Journal 92: 378–379 (1980).
Reeves, D.S. and Davies, A.J.: Antabuse effect with cephalosporins. Lancet 2: 540 (1980).
Shibata, K.: Clinical trials with cefoperazone in the field of surgery in Japan. Clinical Therapeutics 3: 173–189 (Special Issue, 1980).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Bailey, R.R., Peddie, B., Blake, E. et al. Cefoperazone in the Treatment of Severe or Complicated Infections. Drugs 22 (Suppl 1), 76–86 (1981). https://doi.org/10.2165/00003495-198100221-00017
Published:
Issue Date:
DOI: https://doi.org/10.2165/00003495-198100221-00017