Abstract
Objective
To develop a model that relates the probability of occurrence of nephrotoxicity to the cumulative area under the curve (AUC) of amikacin serum concentration.
Design and patients
This was a retrospective study of two groups of patients in whom nephrotoxicity was observed after administration of amikacin. The first group consisted of patients treated with once-daily administration (ODA) [n = 13]. The second group consisted of patients treated with twice-daily administration (TDA) [n = 22].
Main outcome measure
The probability of nephrotoxicity occurrence.
Results
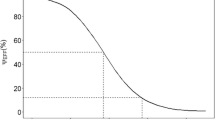
The model is a powerful tool to represent and describe the influence of the dosage regimen on aminoglycoside nephrotoxicity. The onset of nephrotoxicity is delayed in the ODA group (p = 0.01) for the same total daily dose among the two groups. The cumulative serum AUC values at onset of nephrotoxicity were greater for the ODA group (p = 0.029). In addition, for the same probability of nephrotoxicity occurrence (50%), the cumulative AUC for the ODA dosage regimen is 2 613 mg · h/L versus only 1 521 mg · h/L for the TDA dosage regimen. The difference in nephrotoxicity between ODA and TDA is greatest for a cumulative AUC of 2 495 mg · h/L, which corresponds to standard therapy with amikacin 900 mg/day during a 7-day period, i.e. 15 mg/kg/day for a 60kg patient with normal renal function (initial creatinine clearance >80 mL/min). For an AUC above 2 495 mg · h/L, the difference in nephrotoxicity decreases slowly to zero. This result means that ODA is especially justified when the treatment is administered over a short duration, i.e. less than 7 days.
Conclusions
The utility of selecting ODA in order to obtain less nephrotoxicity in comparison with TDA is therefore not established when the treatment is prolonged. In clinical use, the choice of the dosage regimen is not clear-cut, and both expected efficacy and expected toxicity must be taken into account in order to obtain an overall optimisation of each patient’s therapy.




Similar content being viewed by others
References
Maire PH, Corvaisier S, Rougier F, et al. Pharmacocinétique/ pharmacodynamie clinique des antibiotiques. In: Freney J, Renaud F, Hansen W, Bollet C, editors. Précis de bactériologie clinique. Paris: Elsevier, 2000: 715–31
Murry KR, McKinnon PS, Mitrzyk B, et al. Pharmacodynamic characterization of nephrotoxicity associated with once-daily aminoglycoside. Pharmacotherapy 1999; 19: 1252–60
Rybak MJ, Abate BJ, Kang SL, et al. Prospective evaluation of the effect of an aminoglycoside dosing regimen on rates of observed nephrotoxicity and ototoxicity. Antimicrob Agents Chemother 1999; 43: 1549–55
De Broe ME, Verpooten GA. Influence of dosage schedules on renal accumulation of amikacin and tobramycin in man. J Antimicrob Chemother 1991; 27 Suppl. C: 41–7
Verpooten GA, Guiliano RA, Verbist L, et al. Once daily dosing decreases renal accumulation of gentamicin and netilmicin. Clin Pharmacol Ther 1989; 45: 22–7
Schmitz C, Hilpert J, Jacobsen C, et al. Megalin deficiency offers protection from renal aminoglycoside accumulation. J Biol Chem 2002; 277(1): 618–22
Lerner SA, Schmitt BA, Seligsohn R, et al. Comparative study of ototoxicity and nephrotoxicity in patients randomly assigned to treatment with amikacin or gentamicin. Am J Med 1986; 80 Suppl. 6B: 98–104
USC*PACK [computer program]. Version 10.7. Los Angeles, CA: Laboratory of Applied Pharmacokinetics, University of Southern California. School of Medicine; 1995. USC*PACK PC Collection Clinical Research Programs
Jelliffe RW, Jelliffe S. A computer program for estimation of creatinine clearance from unstable serum creatinine levels. Math Biosci 1972; 14: 17–24
Schumitzky A. Non-parametric EM algorithms for estimating prior distributions. Los Angeles: Laboratory of Applied Pharmacokinetics, University of Southern California, School of Medicine; 1990. Technical Report: 90-2
Schentag JJ. Renal clearance and tissue accumulation of gentamicin. Clin Pharmacol Ther 1977; 22: 364–70
Jelliffe RW. The USC*PACK PC programs for population pharmacokinetic modeling of large kinetic/dynamic systems, and adaptive control of drug dosage regimens. Proc Annu Symp Comput Appl Med Care 1991, 922-4
Maurin M, Rougier F, Maire P. Note de calcul sur les lois de Hill: aspects probabiliste, déterministe et épistémologique 2000. Edition INRETS-LTE 2026. ISRN: INRETS/NST/00-548-FR
Hill AV. The possible effects of the aggregation of hemoglobin on its dissociation curve. J Physiol 1910; 40: iv–vii
Michaelis L, Menten ML. Die kinetik der Invertinwirkung. Biochem Z 1913; 49: 333–69
Clark AJ. The reaction between acetylcholine and muscle cells. J Physiol 1926; 61: 530–46
McCullagh P, Neider JA. Generalized linear models. London: Chapman and Hall, 1983
Holford HG, Sheiner LB. Understanding the dose-effect relationship: clinical application of pharmacokinetic-pharmacodynamic models. Clin Pharm 1981; 6: 429–53
Guiliano RA, Verpooten GA, Verbist L, et al. In vivo uptake kinetics of aminoglycosides in the kidney cortex of rats. J Pharmacol Exp Ther 1986; 236: 470–5
Kosek JC, Mazze RI, Cousins J. Nephrotoxicity of gentamicin. Lab Invest 1974; 30: 48–57
Tulkens PM. Nephrotoxicity of aminoglycosides. Toxicol Lett 1989; 46: 107–23
De Broe ME, Paulus GJ, Verpooten G, et al. Early effects of gentamicin, tobramycin and amikacin on the human kidney. Kidney Int 1984; 25: 643–52
Schnermann J, Häberle DA, Davis JM, et al. Tubuloglomerular feedback control of renal vascular resistance. In: Windhager E, editor. Handbook of renal physiology. Oxford: Oxford University Press, 1992: 1675–705
Van der Auwera P. Pharmacokinetic evaluation of single daily dose amikacin. J Antimicrob Chemother 1991; 27 Suppl. C: 63–71
Freeman CD, Nicolau DP, Belliveau PP, et al. Once-daily dosing of aminoglycosides: review and recommendations for clinical practice. J Antimicrob Chemother 1997; 39: 677–86
Gonzales LS, Spencer JP. Aminoglycosides: a practical review. Am Fam Physician 1998; 58: 1811–20
Acknowledgements
This study was funded by Hospices Civils de Lyon, without any grant from Bristol-Myers Squibb; however ADCAPT was funded by this company several years ago.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Rougier, F., Ducher, M., Maurin, M. et al. Aminoglycoside Dosages and Nephrotoxicity. Clin Pharmacokinet 42, 493–500 (2003). https://doi.org/10.2165/00003088-200342050-00007
Published:
Issue Date:
DOI: https://doi.org/10.2165/00003088-200342050-00007