Summary
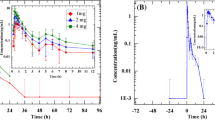
The pharmacokinetics of doxazosin were determined in hypertensive subjects after a single dose of 1mg, and at steady-state while receiving doses of 1, 2, 4 and 8mg of the drug daily. Chlorothiazide 500mg once daily was administered as additional therapy throughout the study. After a single dose doxazosin was rapidly absorbed, with peak plasma drug concentrations (Cmax) occurring after 2.1 ± 0.4 hours. The elimination half-life in plasma was 10.7 ± 1.2 hours. These parameters remained essentially unchanged during maintenance administration of doxazosin at each of the dose levels. Calculations of Cmax and area under the concentration-time curve (A UC0→∞) indicated that the pharmacokinetic disposition of the drug remained linear over the dose range 1 to 8mg.
Similar content being viewed by others
References
Cox DA, Leader JP, Milson JA, Singleton W. The antihypertensive effects of doxazosin: a clinical overview. British Journal of Clinical Pharmacology 21: 83S–90S, 1986
Frick MH, Halttunen P, Himanen P, Huttunen M, Porsti P, et al. A long-term double-blind comparison of doxazosin and atenolol in patients with mild to moderate essential hypertension. British Journal of Clinical Pharmacology 1: 55S–62S, 1986
Lund-Johansen P. Hemodynamic changes at rest and during exercise in long-term prazosin therapy for essential hypertension. Postgraduate Medical Journal 58 (Suppl.): 45–52, 1975
Lund-Johansen P, Omvik P, Haugland H. Acute and chronic haemodynamic effects of doxazosin in hypertension at rest and during exercise. British Journal of Clinical Pharmacology 21: 45S–54S, 1986
McNeil JJ, Drummer OH, Conway EL, Workman BS, Louis WJ. Effects of age on pharmacokinetics and blood pressure responses to prazosin and terazosin. Journal of Cardiovascular Pharmacology 10: 168–175, 1987
Rubin PC, Scott PJW, Reid JL. Prazosin disposition in young and elderly subjects. British Journal of Clinical Pharmacology 12: 401–404, 1981
Sachs FM, Dzau VJ. Adrenergic effects on plasma lipoprotein metabolism. Speculation on mechanisms of action. American Journal of Medicine 80 (Suppl. 2A): 71–81, 1986
Stokes GS. Prazosin. In Doyle AE (Ed.) Clinical pharmacology of antihypertensive drugs, Handbook of Hypertension, Vol. 5, pp. 350–375, Elsevier, Amsterdam, 1984
Vincent J, Elliot HL, Meredith PA, Reid JL. Doxazosin, an alpha-1-adrenoceptor antagonist: pharmacokinetics and concentration effect relationships in man. British Journal of Clinical Pharmacology 15: 719–725, 1983
Vincent J, Meredith PA, Elliott HL, Reid JL. The pharmacokinetics of doxazosin in elderly normotensives. British Journal of Clinical Pharmacology 21: 521–524, 1986
Wagner JG. Biopharmaceutics and relevant pharmacokinetics. In Drug Intelligence Publications, pp. 242–246, Hamilton Press, Hamilton, Illinois, 1971
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Conway, E.L., McNeil, J.J., Meng, L. et al. Single-Dose and Steady-State Pharmacokinetics of Doxazosin Given in Combination with Chlorothiazide to Hypertensive Subjects. Clin-Pharmacokinet 16, 387–391 (1989). https://doi.org/10.2165/00003088-198916060-00004
Published:
Issue Date:
DOI: https://doi.org/10.2165/00003088-198916060-00004