Abstract
▲ Brinzolamide 1%/timolol 0.5% fixed combination (brinzolamide/timolol) is a twice-daily eyedrops suspension comprising the carbonic anhydrase-II inhibitor brinzolamide and the β-adrenergic receptor antagonist timolol.
▲ Brinzolamide/timolol produced clinically relevant reductions in mean intraocular pressure (IOP) from baseline and was more effective than brinzolamide or timolol monotherapy in lowering IOP in a 6-month, randomized, phase III trial in patients with open-angle glaucoma or ocular hypertension (n = 523). The proportion of patients achieving a mean IOP of <18 mmHg was significantly greater in recipients of brinzolamide/timolol than in recipients of brinzolamide or timolol monotherapy.
▲ The IOP-lowering efficacy of brinzolamide/timolol was maintained for up to 12 months, and was no less effective than dorzolamide 2%/timolol 0.5% solution (dorzolamide/timolol) in a randomized, phase III, noninferiority trial (n = 437).
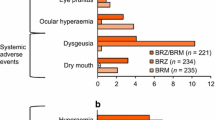
▲ Brinzolamide/timolol was generally well tolerated and was associated with significantly lower ocular discomfort scores than dorzolamide/timolol. Moreover, a significantly greater number of patients expressed a preference for brinzolamide/timolol over dorzolamide/timolol. The main ocular adverse event was blurred vision, and was not considered to be a safety issue.




Similar content being viewed by others
References
Resnikoff S, Pascolini D, Etya’ale D, et al. Global data on visual impairment in the year 2002. Bull World Health Organ 2004; 82(11): 844–51
Glaucoma Research Foundation. African-Americans and glaucoma [online]. Available from URL: http://www.glaucoma.org/learn/africanamerican.php [Accessed 2009 Feb 12]
Muñoz B, West S, Rubin G, et al. Causes of blindness and visual impairment in a population of older Americans. Arch Ophthalmol 2000; 118(6): 819–25
Goldberg L. Clinical guidelines for the treatment of glaucoma. Manag Care 2002; 11(11): 16–24
Fechtner RD, Khouri AS. Evolving global risk assessment of ocular hypertension to glaucoma. Curr Opin Ophthalmol 2007; 18(2): 104–9
Glaucoma Research Foundation. Are you at risk for glaucoma? [online]. Available from URL: http://www.glaucoma.org/learn/are_you_at_risk.php [Accessed 2009 Feb 12]
The Eye Digest: University of Illinois Eye & Ear Infirmary. Glaucoma causes optic nerve cupping (atrophy) and vision loss [online]. Available from URL: http://www.agingeye.net/glaucoma/glaucomainformation.php [Accessed 2009 Feb 13]
Foster A, Johnson G. Magnitude and causes of blindness in the developing world. Int Ophthalmol 1990; 14(3): 135–40
Klaver C, Wolfs R, Vingerling J, et al. Age-specific prevalence and causes of blindness in an older population. Arch Ophthalmol 1998; 116(5): 653–8
Forsman E, Kivela T, Vesti E. Lifetime visual disability in open-angle glaucoma and ocular hypertension. J Glaucoma 2007; 16(3): 313–9
The Royal College of Ophthalmologists. A national research strategy for ophthalmology (March 2002) [online]. Available from URL: http://www.rcophth.ac.uk/docs/publications/published-guidelines/EyeResearchFinal.pdf [Accessed 2009 Feb 5]
European Glaucoma Society. Terminology and guidelines for glaucoma. 3rd ed. Savona, Italy: Dogma, 2008
Japan Glaucoma Society. Guidelines for glaucoma [online]. Available from URL: http://www.ryokunaisho.jp/english/Guidelines_for_Glaucoma.pdf [Accessed 2009 Feb 13]
Kass M, Heuer D, Higginbotham E, et al. The ocular hypertension treatment study: a randomized trial determines that topical ocular hypotensive medication delays prevents the onset of open-angle glaucoma. Arch Ophthalmol 2002; 120(6): 701–13
Geringer C, Imami N. Medical management of glaucoma. Int Ophthamol Clin 2008; 48(4): 115–41
International Council of Ophthalmology. Primary open-angle glaucoma (initial evaluation) [online]. Available from URL: http://www.icoph.org/guide/guidepri.html [Accessed 2009 Feb 13]
Fechtner R, Realini T. Fixed combinations of topical glaucoma medications. Curr Opin Ophtahalmol 2004; 15(2): 132–5
Cvetkovic RS, Perry CM. Brinzolamide: a review of its use in the management of primary open-angle glaucoma and ocular hypertension. Drugs Aging 2003; 20(12): 919–47
Iester M. Brinzolamide. Expert Opin. Pharmacother 2008; 9(4): 653–62
DeSantis L. Preclinical overview of brinzolamide. Surv Opthalmol 2000; 44(Suppl. 2): S119–29
Heel R, Brogden R, Speight T, et al. Timolol: a review of its therapeutic efficacy in the topical treatment of glaucoma. Drugs 1979; 17(1): 38–55
Han JA, Frishman WH, Wu Sun S, et al. Cardiovascular and respiratory considerations with pharmacotherapy of glaucoma and ocular hypertension. Cardiol Rev 2008; 16(2): 95–108
March W, Ochsner K. The long-term safety and efficacy of brinzolamide 1.0% (Azopt) in patients with primary open-angle glaucoma or ocular hypertension. Brinzolamide Long-Term Therapy Study Group. Am J Ophthalmol 2000; 129(2): 136–43
European Agency for the Evaluation of Medicinal Products. Brinzolamide/timolol: summary of product characteristics [online]. Available from URL: http://www.emea.europa.eu/humandocs/PDFs/EPAR/azarga/H-960-PI-en.pdf [Accessed 2009 Feb 2]
Kaup M, Plange N, Niegel M, et al. Effects of brinzolamide on ocular haemodynamics in healthy volunteers. Br J Ophthalmol 2004; 88(2): 257–62
Izzotti A, Saccà S, Di Marco B, et al. Antioxidant activity of timolol on endothelial cells and its relevance for glaucoma course. Eye 2008; 22(3): 445–53
Kiland J, Gabelt B, Kaufman P. Studies on the mechanism of action of timolol and the effects and redirection of aqueous flow on outflow facility. Exp Eye Res 2004; 78(3): 639–51
Korte J, Kaila T, Saari K, et al. Systemic bioavailability and cardiopulmonary effects of 0.5% timolol eyedrops. Graefes Arch Clin Exp Ophthalmol 2002; 240(6): 430–5
Manni G, Denis P, Chew P, et al. The safety and efficacy of brinzolamide 1%/timolol 0.5% fixed combination versus dorzolamide 2%/timolol 0.5% in patients with open-angle glaucoma or ocular hypertension. J Glaucoma 2009; 18(4): 293–300
Kaback M, Scoper SV, Arzeno G, et al. Intraocular pressure-lowering efficacy of brinzolamide 1%/timolol 0.5% fixed combination compared with brinzolamide 1 % and timolol 0.5%. Ophthalmology 2008; 115(10): 1728–34
Vold SD, Evans RM, Stewart RH, et al. A one-week comfort study of BID-dosed brinzolamide 1%/timolol 0.5% ophthalmic suspension fixed combination compared to BID-dosed dorzolamide 2%/timolol 0.5% ophthalmic in patients with open-angle glaucoma or ocular hypertension. J Ocul Pharmacol Ther 2008; 24(6): 601–5
Mundorf TK, Rauchman SH, Williams RD, et al. A patient preference comparison of Azarga™ (brinzolamide/timolol fixed combination) vs Cosopt® (dorzolamide/timolol fixed combination) in patients with open-angle glaucoma or ocular hypertension. Clin Ophthalmol 2008; 2(3): 623–8
Acknowledgements and Disclosures
This manuscript was reviewed by: G. Manni, Ophthalmology Department, University of Roma, Roma, Italy; F. Ziemssen, University Eye Hospital, Centre of Ophthalmology, Eberhard Karl University, Tuebingen, Germany
The preparation of this review was not supported by any external funding. During the peer review process, the manufacturer of the agent under review was offered an opportunity to comment on this article. Changes resulting from comments received were made on the basis of scientific and editorial merit.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Croxtall, J.D., Scott, L.J. Brinzolamide/Timolol. Drugs Aging 26, 437–446 (2009). https://doi.org/10.2165/00002512-200926050-00007
Published:
Issue Date:
DOI: https://doi.org/10.2165/00002512-200926050-00007