Abstract
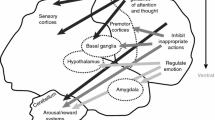
Although originally developed for the treatment of hypertension, α2-agonists have been used to treat Tourette’s syndrome, attention-deficit hyperactivity disorder (ADHD), developmental disorders and substance abuse for nearly three decades. Based on studies of clonidine, α2-agonists were presumed to reduce arousal by decreasing the firing of noradrenaline neurons in locus ceruleus. Accumulated preclinical evidence indicates that guanfacine has features in common with clonidine, in addition to other pharmacological effects. Clonidine binds to the three subtypes of α2-receptors, A, B and C, whereas guanfacine binds more selectively to α2A-receptors, which appears to enhance prefrontal function. Several reports on the use of the α2-agonists show improvements in children with ADHD and improvements in hyperactivity, impulsiveness and inattention in children with tic disorders and pervasive developmental disorders. Both clonidine and guanfacine are associated with sedation, fatigue and somnolence. Reductions in heart rate and blood pressure are modest and rarely lead to discontinuation of treatment across these trials.

Similar content being viewed by others
References
Cohen DJ, Young JG, Nathanson JA, et al. Clonidine in Tourette’s syndrome. Lancet 1979; 2(8142): 551–3
Gold MS, Redmond Jr DE, Kleber HD. Clonidine blocks acute opiate-withdrawal symptoms. Lancet 1978; 2(8090): 599–602
Riddle MA, Bernstein GA, Cook EH, et al. Anxiolytics, adrenergic agents, and naltrexone. J Am Acad Child Adolesc Psychiatry 1999; 38(5): 546–56
Arnsten AF, Steere JC, Hunt RD. The contribution of alpha 2-noradrenergic mechanisms of prefrontal cortical cognitive function. Potential significance for attention-deficit hyperactivity disorder. Arch Gen Psychiatry 1996; 53(5): 448–55
Arnsten AFT. Alpha-2 agonists in the treatment of ADHD. Medscape Psychiatry & Mental Health 2007 [online]. Available from URL: http://www.medscape.com/viewarticle/560074 [Accessed 2008 Aug 24]
Wang M, Ramos BP, Paspalas CD, et al. Alpha2A-adrenoceptors strengthen working memory networks by inhibiting cAMP-HCN channel signaling in prefrontal cortex. Cell 2007; 129(2): 397–410
Arnsten AF, Cai JX, Goldman-Rakic PS. The alpha-2 adrenergic agonist guanfacine improves memory in aged monkeys without sedative or hypotensive side effects: evidence for alpha-2 receptor subtypes. J Neurosci 1988; 8(11): 4287–98
Hunt RD, Minderaa RB, Cohen DJ. Clonidine benefits children with attention deficit disorder and hyperactivity: report of a double-blind placebo-crossover therapeutic trial. J Am Acad Child Psychiatry 1985; 24(5): 617–29
MacDonald E, Kobilka BK, Scheinin M. Gene targeting-homing in on alpha 2-adrenoceptor-subtype function. Trends Pharmacol Sci 1997; 18(6): 211–9
Rubia K, Smith AB, Brammer MJ, et al. Right inferior prefrontal cortex mediates response inhibition while mesial prefrontal cortex is responsible for error detection. Neuroimage 2003; 20(1): 351–8
Jakala P, Riekkinen M, Sirvio J, et al. Guanfacine, but not clonidine, improves planning and working memory performance in humans. Neuropsychopharmacology 1999; 20(5): 460–70
Arnsten AFT. Toward a new understanding of attention-deficit hyperactivity disorder pathophysiology: an important role for prefrontal cortex dysfunction. CNS Drugs 2009; 23Suppl. 1: 33–41
Balldin J, Berggren U, Eriksson E, et al. Guanfacine as an alpha-2-agonist inducer of growth hormone secretion— a comparison with clonidine. Psychoneuroendocrinology 1993; 18(1): 45–55
Sica DA. Centrally acting antihypertensive agents: an update. J Clin Hypertens 2007;9(5): 399–405
Sarlis NJ, Caticha O, Anderson JL, et al. Hyperadrenergic state following acute withdrawal from clonidine used at supra therapeutic doses. Clin Auton Res 1996; 6(2): 115–7
Westfall T, Westfall D. Adrenergic agonists and antagonists. In: Brunton L, Lazo J, Parker K, editors. Goodman & Gilman’s the pharmaceutical basis of therapeutics. New York (NY): McGraw-Hill, 2008: 237–96
Connor DF, Fletcher KE, Swanson JM. A meta-analysis of clonidine for symptoms of attention-deficit hyperactivity disorder. J Am Acad Child Adolesc Psychiatry 1999; 38(12): 1551–9
Scahill L, Chappell PB, Kim YS, et al. A placebo-controlled study of guanfacine in the treatment of children with tic disorders and attention deficit hyperactivity disorder. Am J Psychiatry 2001; 158(7): 1067–74
Tourette syndrome (TS) Study Group. Treatment of ADHD in children with tics: a randomized controlled trial. Neurology 2002; 58(4): 527–36
Cummings DD, Singer HS, Krieger M, et al. Neuropsychiatric effects of guanfacine in children with mild Tourette syndrome: a pilot study. Clin Neuropharmacol 2002; 25(6): 325–32
Palumbo DR, Sallee FR, Pelham Jr WE, et al. Clonidine for attention-deficit/hyperactivity disorder: I. Efficacy and tolerability outcomes. J Am Acad Child Adolesc Psychiatry 2008; 47(2): 180–8
Handen BL, Sahl R, Hardan AY. Guanfacine in children with autism and/or intellectual disabilities. J Dev Behav Pediatr 2008; 29(4): 303–8
Biederman J, Melmed RD, Patel A, et al. A randomized, double-blind, placebo-controlled study of guanfacine extended release in children and adolescents with attention-deficit/hyperactivity disorder. Pediatrics 2008; 121(1): e73–84
Sallee FR, McGough J, Wigal T, et al., for the SPD503 Study Group. Guanfacine extended release in children and adolescents with attention-deficit/hyperactivity disorder: a placebo-controlled trial. J Am Acad Child Adolesc Psychiatry 2009; 48(2): 155–65
Scahill L, Erenberg G, Berlin CM, et al. Contemporary assessment and pharmacotherapy of Tourette syndrome. NeuroRx 2006; 3(2): 192–206
Multimodal Treatment Study of Children with Attention-Deficit/Hyperactivity Disorder (MTA) Cooperative Group. Moderators and mediators of treatment response for children with attention-deficit/hyperactivity disorder: the Multi-modal Treatment Study of children with Attention-deficit/ hyperactivity disorder. Arch Gen Psychiatry 1999; 56: 1088–96
Daviss WB, Patel NC, Robb AS, et al. Clonidine for attention-deficit/hyperactivity disorder: II. ECG changes and adverse events analysis. J Am Acad Child Adolesc Psychiatry 2008; 47(2): 189–98
Hunt RD, Arnsten AFT, Asbell MD. An open trial of guanfacine in the treatment of attention-deficit hyperactivity disorder. J Am Acad Child Adolesc Psychiatry 1995; 34: 50–4
Chappell PB, Riddle MA, Scahill L, et al. Guanfacine treatment of comorbid attention-deficit hyperactivity disorder and Tourette’s syndrome: preliminary clinical experience. J Am Acad Child Adolesc Psychiatry 1995; 34: 1140–6
Horrigan JP, Barnhill LJ. Guanfacine for treatment of attention-deficit hyperactivity disorder in boys. J Child Adolesc Psychopharmacol 1995; 5: 215–23
DuPaul GJ, Ervin RA, Hook CL, et al. Peer tutoring for children with attention deficit hyperactivity disorder: effects on classroom behavior and academic performance. J Appl Behav Anal 1998; 31: 579–92
Aman MG, Singh NN, Stewart AW, et al. The Aberrant Behavior Checklist: a behavior rating scale for the assessment of treatment effects. Am J Ment Defic 1985; 89(5): 485–91
Scahill L, Aman MG, McDougle CJ, et al. A prospective open trial of guanfacine in children with pervasive developmental disorders. J Child Adolesc Psychopharmacol 2006; 16(5): 589–98
Boreman CD, Arnold LE. Hallucinations associated with initiation of guanfacine. J Am Acad Child Adolesc Psychiatry 2003; 42(12): 1387
Luthra V, Markov D, Ambrosini P. Does guanfacine cause hallucinations in children? J Child Adolesc Psychopharmacol 1999; 9(4): 313–4
Horrigan JP, Barnhill LJ. Guanfacine and secondary mania in children. J Affect Disord 1999; 54(3): 309–14
Acknowledgements
Lawrence Scahill, MSN, PhD, has been a consultant for Bristol-Myers Squibb, Janssen Pharmaceuticals Inc., Neuropharm, Shire Pharmaceuticals Inc. and Supernus Pharmaceuticals Inc.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Scahill, L. Alpha-2 Adrenergic Agonists in Children with Inattention, Hyperactivity and Impulsiveness. CNS Drugs 23 (Suppl 1), 43–49 (2009). https://doi.org/10.2165/00023210-200923000-00006
Published:
Issue Date:
DOI: https://doi.org/10.2165/00023210-200923000-00006