Abstract
Background
Calcitonin gene-related peptide (CGRP) is a vasodilatory neuropeptide that plays an important role in the blood vessels of heart and peripheral circulation, a lack of which may cause vasculopathies.
Objective
In this study, the clinical course of disease, as well as the efficacy, side effects, and patient satisfaction of systemic calcitonin therapy in patients with systemic sclerosis (SSc), was evaluated.
Methods
Forty-nine patients received repetitive intravenous calcitonin infusions as first-line treatment. The average number of cycles was 12.2 ± 10.3 over a period of 30 months (each cycle: 100 U/day over 10 days). Clinical examinations, laboratory tests, and organ imaging were performed before the start of, and at regular intervals during therapy in order to evaluate organ manifestations and the clinical course of the disease. In addition, patients’ own experiences of the therapy, side effects, and therapy success were evaluated with standardized questionnaires.
Results
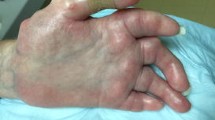
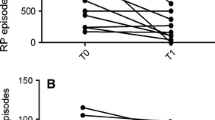
Over the course of the treatment, seven patients experienced improvements in their condition with a considerable reduction in digital ulceration and improved movement (14.3%). Pulmonary function in seven patients improved during the therapy (14.3%). With regards to side effects, nausea (41.7%), headaches (33.3%), fluctuations in blood pressure (29.2%), and flushing (29.2%) were observed. Overall, 45.8% of patients evaluated the therapy as good and 58.3% would undergo further courses of therapy with calcitonin.
Conclusions
Systemic calcitonin treatment seems to have positive clinical effects on SSc and contributes to relieving symptoms, especially in patients with cutaneous manifestations. No severe side effects were reported during this study.
Similar content being viewed by others
References
Sticherling M. Systemic sclerosis–dermatological aspects. Part 1:Pathogenesis, epidemiology, clinical findings. J Dtsch Dermatol Ges 2012; 10: 705–18.
Klein–Weigel P, Opitz C, Riemekasten G. Systemic sclerosis–a systematic overview:part 1–disease characteristics and classification, pathophysiologic concepts, and recommendations for diagnosis and surveillance. Vasa 2011; 40: 6–19.
Sticherling M. Systemic sclerosis–focus on dermatological aspects. Part 2:diagnostics, therapy. J Dtsch Dermatol Ges 2012; 10: 783–91.
Young A, Khanna D. Systemic sclerosis:a systematic review on therapeutic management from 2011 to 2014. Curr Opin Rheumatol 2015; 27: 241–8.
Ciechomska M, van Laar J, O’Reilly S. Current frontiers in systemic sclerosis pathogenesis. Exp Dermatol 2015; 24: 401–6.
Chizzolini C. Update on pathophysiology of scleroderma with special reference to immunoinflammatory events. Ann Med 2007; 39: 42–53.
Savarino E, Mei F, Parodi A, et al. Gastrointestinal motility disorder assessment in systemic sclerosis. Rheumatology (Oxford) 2013; 52: 1095–100.
McNearney TA, Reveille JD, Fischbach M, et al. Pulmonary involvement in systemic sclerosis:associations with genetic, serologic, sociodemographic, and behavioral factors. Arthritis Rheum 2007; 57: 318–26.
Trang G, Steele R, Baron M, Hudson M. Corticosteroids and the risk of scleroderma renal crisis:a systematic review. Rheumatol Int 2012; 32: 645–53.
Walker UA, Tyndall A, Czirják L, et al. Clinical risk assessment of organ manifestations in systemic sclerosis:a report from the EULAR Scleroderma Trials And Research group database. Ann Rheum Dis 2007; 66: 754–63.
Turk M, Pope EP. The frequency of scleroderma renal crisis over time:a metaanalysis. J Rheumatol 2016; 43: 1350–5.
Knockaert DC. Cardiac involvement in systemic inflammatory diseases. Eur Heart J 2007; 28: 1797–804.
Avouac J, Sordet C, Depinay C, et al. Systemic sclerosisassociated Sjögren’s syndrome and relationship to the limited cutaneous subtype:results of a prospective study of sicca syndrome in 133 consecutive patients. Arthritis Rheum 2006; 54: 2243–9.
Riemekasten G, Sunderkötter C. Vasoactive therapies in systemic sclerosis. Rheumatology (Oxford) 2008; 47: 234–5.
Brueckner CS, Becker MO, Kroencke T, et al. Effect of Sildenafil on digital ulcers in systemic sclerosis–analysis from a single centre pilot study. Ann Rheum Dis 2010; 69: 1475–8.
García de la Pe˜na–Lefebvre P, Rodríguez Rubio S, Valero Expósito M, et al. Long–term experience of bosentan for treating ulcers and healed ulcers in systemic sclerosis patients. Rheumatology 2008; 47: 464–6.
Herrick AL. Pathogenesis of Raynaud’s phenomenon. Rheumatology 2005; 44: 587–96.
Albrecht HP, Hiller D, Hornstein OP, et al. Microcirculatory functions in systemic sclerosis:additional parameters for therapeutic concepts? J Invest Dermatol 1993; 101: 211–5.
Gevatter M, Keller J, Hornstein OP. The thermoregulatory effectiveness of calcitonin in progressive scleroderma. Z Hautkr 1989; 64: 507–10.
Masi AT, Rodnan GP, Medsger TA, et al. Preliminary criteria for the classification of systemic sclerosis (scleroderma). Arthritis Rheum 1980; 23: 581–90.
Hunzelmann N, Genth E, Krieg T, et al. The registry of the German Network for Systemic Scleroderma:frequency of disease subsets and patterns of organ involvement. Rheumatology 2008; 47: 1185–92.
Lambova SN, Müller–Ladner U. New lines in therapy of Raynaud’s phenomenon. Rheumatol Int 2009; 29: 355–63.
Clements PJ, Roth MD, Elashoff R, et al. Scleroderma lung study (SLS):differences in the presentation and course of patients with limited versus diffuse systemic sclerosis. Ann Rheum Dis 2007; 66: 1641–7.
Khanna D, Clements PJ, Furst DE, et al. Recombinant human relaxin in the treatment of systemic sclerosis with diffuse cutaneous involvement:a randomized, double–blind, placebo–controlled trial. Arthritis Rheum 2009; 60: 1102–11.
Apras S, Ertenli I, Ozbalkan Z, et al. Effects of oral cyclophosphamide and prednisolone therapy on the endothelial functions and clinical findings in patients with early diffuse systemic sclerosis. Arthritis Rheum 2003; 48: 2256–61.
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Uslu, U., Streiff, L. & Sticherling, M. Systemic therapy with calcitonin has positive clinical effects on systemic sclerosis in patients with cutaneous manifestations. Eur J Dermatol 28, 364–369 (2018). https://doi.org/10.1684/ejd.2018.3300
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1684/ejd.2018.3300