Abstract
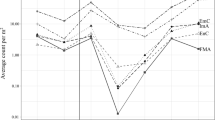
We compared growth and survival performance of the detritivorous amphipod Hyalella azteca fed lab-conditioned leaves of either Typha angustifolia (narrow-leaf cattail), Phragmites australis (common reed) sprayed with the herbicide Glypro®, and unsprayed Phragmites, in microcosms. Leaves were from plants varying in time of senescence. Herbicide application advanced plant senescence and death by ∼3 months, whereas the other two leaf types senesced naturally. Amphipods grew, on average, ∼ 100 μg d−1, and amphipod growth was positively related to fungal biomass on leaves, suggesting that fungi were a key nutritive source. Average fungal biomass was significantly greater on herbicide-treated, early sensecent Phragmites (317 ±43 μg ergosterol g−1 dry wt.; mean ±1 SE) and Typha (226 ±39 μg ergosterol g−1 dry wt.) than on naturally senescent Phragmites (114 ±21 μg ergosterol g−1 dry wt.). Leaf toughness, an indicator of unpalatability, was greatest on naturally senescent Phragmites leaves. Even so, amphipod growth, survival, and offspring numbers did not differ among litter types, probably because individuals were mainly eating surface biofilms rather than the actual leaf matter, and biofilm abundance was relatively high in all leaf treatments. We also quantified decay rates of each leaf type, and amphipod (Hyalella and Gammarus pseudolimnaeus) abundance in leaves in a Lake Erie coastal marsh (i.e., drowned river mouth) to relate amphipod performance and leaf properties in microcosms to natural patterns. Average rates of leaf breakdown (∼10.3 mg d−1 over 126 d) in coarse-mesh litterbags and amphipod numbers in litterbags were similar among leaf types. Leaf mass decreased by ∼20% during October but slowed from November to January when amphipod abundance and water temperature sharply declined. An increase in leaf loss from February to March, when Hyalella numbers and water temperatures (∼0.1 °C) were low, coincided with river channel thawing and opening, suggesting that ice and sediment movement affected litter breakdown. The ability of amphipods to perform, and fungi to grow, equally well on reed and cattail implies that leaf detritus of these macrophytes plays a similar role in marsh detrital food webs.
Similar content being viewed by others
Literature Cited
Amsberry, L., M. A. Baker, P. J. Ewanchuk, and M. D. Bertness. 2000. Clonal integration and the expansion of Phragmites australis. Ecological Applications 10: 1110–1118.
Andersen, F. Ø. 1978. Effects of nutrient level on decomposition of Phragmites communis Trin. Archive Hydrobiologie 84: 42–54.
Anesio, A. M., L. J. Tranvik, and W. Granéli. 1999. Production of inorganic carbon from aquatic macrophytes by solar radiation. Ecology 80: 1852–1859.
Angradi, T. R., S. M. Hagan, and K. W. Able. 2001. Vegetation type and the intertidal macroinvertebrate fauna of a brackish marsh: Phragmites vs. Spartina. Wetlands 21: 75–92.
Armstrong, J., F. Afreen-Zobayed, and W. Armstrong. 1996a. Phragmites die-back: sulphide- and acetic acid-induced bud and root death, lignifications, and blockages with aeration and vascular systems. New Phytologist 134: 601–614.
Armstrong, J., W. Armstrong, P. M. Beckett, J. E. Halder, S. Lythe, R. Holt, and A. Sinclair. 1996b. Pathways of aeration and the mechanisms and beneficial effects of humidity and Venturi-induced convections in Phragmites australis (Cav.) Trin. ex Steud. Aquatic Botany 54: 177–197.
Arsuffi, T. L. and K. Suberkropp. 1984. Leaf processing capabilities of aquatic hyphomycetes: Interspecific differences and influence of shredder feeding preferences. Oikos 42: 144–154.
Arsuffi, T. L. and K. Suberkropp. 1985. Selective feeding by stream caddisflies (Trichoptera) detritivores on leaves with fungal-colonized patches. Oikos 45: 50–58.
Arsuffi, T. L. and K. Suberkropp. 1986. Growth of two stream caddisflies (Trichoptera) on leaves colonized by different fungal species. Journal of the North American Benthological Society 5: 297–305.
Bärlocher, F. 1985. The role of fungi in the nutrition of stream invertebrates. Botanical Journal of the Linnean Society 91: 83–94.
Bedford, A. P. 2005. Decomposition of Phragmites australis litter in seasonally flooded and exposed areas of a managed reedbed. Wetlands 25: 713–720.
Benfield, E. F. 1996. Leaf breakdown in steam ecosystems. p. 579–589. In F. R. Hauer and G. A. Lamberti (eds.) Methods in Stream Ecology. Academic Press, San Diego, CA, USA.
Benoit, L. K. and R. A. Askins. 1999. Impact of the spread of Phragmites on the distribution of birds in Connecticut tidal marshes. Wetlands 19: 194–208.
Besitka, M. A. R. 1996. An ecological and historical study of Phragmites australis along the Atlantic Coast. M.S. Thesis. Drexel University, Philadelphia, PA, USA.
Carter, M. D. and K. Suberkropp. 2004. Respiration and annual fungal production associated with decomposing leaf litter in two streams. Freshwater Biology 49: 1112–1122.
Chambers, R. M., L. A. Meyerson, and K. Saltonstall. 1999. Expansion of Phragmites australis into tidal wetlands of North America. Aquatic Botany 64: 261–273.
Covich, A. P. and J. H. Thorp. 2001. Introduction to the subphylum Crustacea. p. 777–809. In J. H. Thorp and A. P. Covich (eds.) Ecology and Classification of North American Freshwater Invertebrates, 2nd ed., Academic Press, San Diego, CA, USA.
Delong, M. D., R. B. Summers, and J. H. Thorp. 1993. Influence of food type on the growth of a riverine amphipod, Gammarus fasciatus. Canadian Journal of Fisheries and Aquatic Sciences 50: 1891–1896.
Dokkum, H. P., D. M. E. Slijkerman, L. Rossi, and M. L. Costantini. 2002. Variation in the decomposition of Phragmites australis litter in a monomictic lake: the role of gammarids. Hydrobiologia 482: 69–77.
Enriquez, S., C. M. Duarte, and K. Sand-Jensen. 1993. Patterns in decomposition rates among photosynthetic organisms: the importance of detritus C:N:P content. Oecologia 94: 457–471.
Feeny, P. 1970. Seasonal changes in oak leaf tannins and nutrients as a cause of spring feeding by winter moth catipillars. Ecology 51: 565–581.
Fell, P. E., S. P. Weissbach, D. A. Jones, M. A. Fallon, J. A. Zeppieri, E. K. Faison, K. A. Lennon, K. J. Newberry, and L. K. Reddington. 1998. Does invasion of oligohaline tidal marshes by reed grass, Phragmites australis (Cav.) Trin.ex. Steud., affect the availability of prey resources for the mummichog, Fundulus heteroclitus L.? Journal of Experimental Marine Biology and Ecology 222: 59–77.
Findlay, S., S. Dye, and K. A. Kuehn. 2002. Microbial growth and nitrogen retention in litter of Phragmites australis compared to Typha angustifolia. Wetlands 22: 616–625.
Findlay, S., K. Howe, and K. Austin. 1990. Comparison of detritus dynamics in two tidal freshwater wetlands. Ecology 71: 288–295.
Galatowitsch, S. M., N. O. Anderson, and P. D. Ascher. 1999. Invasiveness in wetland plants in temperate North America. Wetlands 19: 733–755.
Gessner, M. O. 2000. Breakdown and nutrient dynamics of submerged Phragmites shoots in the littoral zone of a temperate hardwater lake. Aquatic Botany 66: 9–20.
Gessner, M. O., K. Suberkropp, and E. Chauvet. 1997. Decomposition of plant litter by fungi in marine and freshwater ecosystems. p. 303–322. In D. T. Wicklow and B. Söderström (eds.) The Mycota, Vol. IV, Springer Verlag, Berlin, Germany.
Graca, M. A. S., C. Cressa, M. O. Gessner, M. J. Feio, K. A. Callies, and C. Barrios. 2001. Food quality, feeding preferences, survival and growth of shredders from temperate and tropical streams. Freshwater Biology 46: 947–957.
Haley, C. J. 1997. Comparison of secondary production, life history, and mouthpart morphology between two populations of the amphipod Gammarus minus. Ph.D. Dissertation. Virginia Polytechnic Institute and State University, Blacksburg, VA, USA.
Hargrave, B. T. 1970. The utilization of benthic microflora by Hyalella azteca (Amphipoda). Journal of Animal Ecology 39: 427–437.
Hargrave, B. T. 1971. An energy budget for a deposit feeding amphipod. Limnology and Oceanography 16: 99–103.
Herdendorf, C. E. 1989. Paleogeography and Geomorphology. p. 35–70. In K. Krieger (ed.) Lake Erie and Its Estuarine Systems: Issues, Resources, Status, and Management. U.S. Department of Commerce, National Oceanic and Atmospheric Administration, Washington, DC, USA.
Herdendorf, C. E., R. C. Herdendorf, and D. M. Klarer. 2000. Catalogue of the Invertebrate Fauna of Old Woman Creek Estuary, Watershed, and Adjacent Waters of Lake Erie. Old Woman Creek National Estuarine Research Reserve and State Nature Preserve. Technical Report No.12.
Hietz, P. 1992. Decomposition and nutrient dynamics of reed (Phragmites australis (Cav.) Trin ex steud.) litter in Lake Neusiedl, Austria. Aquatic Botany 43: 211–230.
Hurlbert, S. H. 1984. Pseudoreplication and the design of ecological field experiments. Ecological Monographs 54: 187–211.
Iddles, T. L., J. Read, and G. D. Sanson. 2003. The potential contribution of biomechanical properties to anti-herbivore defence in seedlings of six Australian rainforest trees. Australian Journal of Botany 51: 119–128.
Klarer, D. M. and D. F. Millie. 1994. Regulation of phytoplankton dynamics in a Laurentian Great Lakes estuary. Hydrobiologia 482: 69–77.
Kneib, R. T. 1997. The role of tidal marshes in the ecology of estuarine nekton. Oceanography and Marine Biology Annual Review 35: 163–220.
Komínková, D., K. A. Kuehn, N. Busing, and D. Steiner. 2000. Microbial biomass, growth, and respiration associated with submerged litter of Phragmites australis decomposing in a littoral reed stand of a large lake. Aquatic Microbial Ecology 22: 271–282.
Kostalos, M. and R. L. Seymour. 1976. Role of microbial enriched detritus in the nutrition of Gammarus minus (Amphipoda). Oikos 27: 512–516.
Kuehn, K. A., M. O. Gessner, R. G. Wetzel, and K. Suberkropp. 1999. Decomposition and CO2 evolution from standing litter of the emergent macrophyte Erianthus giganteus. Microbial Ecology 38: 50–57.
Kuehn, K. A. and K. Suberkropp. 1998. Decomposition of standing litter of the freshwater emergent macrophyte Juncus effuses. Freshwater Biology 40: 717–727.
Květ, J. and D. F. Westlake. 1998. Primary production in wetlands. p. 78–268. In D. F. Westlake, J. Květ, and A. Szczepański (eds.) The Production Ecology of Wetlands. Cambridge University Press, Cambridge, UK.
Lozano, S. J., M. L. Gedeon, and P. F. Landrum. 2003. The effects of temperature and organism size on the feeding rate and modeled chemical accumulation in Diporeia spp. for Lake Michigan sediments. Journal of Great Lakes Research 29: 79–88.
Marchent, R. and H. B. N. Hynes. 1981. Field estimates of feeding rate for Gammarus pseudolimnaeus (Crustacea: Amphipoda) in the Credit River, Ontario. Freshwater Biology 11: 27–36.
Marks, M., B. Lapin, and J. Randall. 1994. Phragmites australis (P. communis): Threats, Management, and Monitoring. Natural Areas Journal 14: 285–294.
Mason, C. F. and R. J. Bryant. 1975. Production, nutrient content and decomposition of Phragmites communis Trin. and Typha latifolia L. Journal of Ecology 63: 71–95.
Meyerson, L. A., K. Saltonstall, L. Windham, E. Kiviat, and S. Findlay. 2000. A comparison of Phragmites australis in freshwater and brackish marsh environments in North America. Wetlands Ecology and Management 8: 89–103.
Mitsch, W. J. and J. G. Gosselink. 2000. Wetlands, 3rd ed., John Wiley and Sons, New York, NY, USA.
Moore, J. W. 1975. The role of algae in the diet of Asellus aquaticus L. and Gammarus pulex L. Journal of Animal Ecology 44: 719–729.
Newell, S. Y. 2001. Fungal biomass and productivity in standingdecaying leaves of black needlerush (Juncus roemerianus). Marine and Freshwater Research 52: 249–255.
Newell, S. Y. and D. Porter. 2000. Microbial secondary production from saltmarsh-grass shoots, and its known and potential fates. p. 159–185. In M. P. Weinstein and D. A. Kreeger (eds.) Concepts and Controversies in Tidal Marsh Ecology. Kluwer Academic Publishers, Dordrecht, The Netherlands.
Nilsson, L. M. 1974. Energy budget of a laboratory population of Gammarus pulex (Amphipoda). Oikos 25: 35–42.
Pennak, R. W. 1989. Freshwater Invertebrates of the United States: Protozoa to Mollusca, 3rd ed. Wiley, New York, NY, USA.
Polunin, N. V. C. 1982. Processes contributing to the decay of reed (Phragmites australis) litter in fresh water. Archive für Hydrobiologie 94: 182–209.
Polunin, N. V. C. 1984. The decomposition of emergent macrophytes in fresh water. Advances in Ecological Research 14: 115–166.
Reed, P. B. 1988. National list of plant species that occur in wetlands: Northeast (Region 1). United States Fish and Wildlife Service, Washington, DC, USA. Biological Report 88(26.1).
Rozema, J., M. Tosserams, H. J. M. Nelissen, L. van Heerwaarden, R. A. Broekman, and N. Flierman. 1997. Stratospheric ozone reduction and ecosystem processes: enhanced UV-B radiation affects chemical quality and decomposition of leaves of the dune grassland species Calamagrostis epigeios. Plant Ecology 128: 284–294.
Saltonstall, K. 2002. Cryptic invasion by a non-native genotype of the common reed, Phragmites australis, into North America. Proceedings of the National Academy of Sciences of the USA 99: 2445–2449.
Schulz, M. J. and M. N. Thormann. 2005. Functional and taxonomic diversity of saprobic filamentous fungi from Typha latifolia from central Alberta, Canada. Wetlands 25: 675–684.
Short, T. M. and J. R. Holomuzki. 1992. Indirect effects of fish on foraging behavior and leaf processing by the isopod Lirceus fontinalis. Freshwater Biology 27: 91–97.
Sokal, R. R. and F. J. Rohlf. 1995. Biometry. 3rd ed., W.H. Freeman and Company, New York, NY, USA.
Suberkropp, K. and E. Chauvet. 1995. Regulation of leaf breakdown by fungi in streams: influences of water chemistry. Ecology 76: 1433–1445.
Tanaka, Y. 1991. Microbial decomposition of reed (Phragmites communis) leaves in a saline lake. Hydrobiologia 220: 119–129.
Templer, P., S. Findlay, and C. Wigand. 1998. Sediment chemistry associated with native and non-native emergent macrophytes of a Hudson River marsh ecosystem. Wetlands 44: 719–729.
Trexell-Kroll, D. 2002. Succession of floating-leaf to emergent plant communities following reduced water levels in the Old Woman Creek Estuary. M.Sc. Thesis. Miami University, Oxford, OH, USA.
Walse, C., B. Berg, and H. Sverdrup. 1998. Review and synthesis of experimental data on organic matter decomposition with respect to the effect of temperature, moisture, and acidity. Environmental Reviews 6: 25–40.
Warren, R. S., P. E. Fell, J. L. Grimsby, E. L. Buck, G. C. Rilling, and R. A. Fertik. 2001. Rates, patterns, and impacts of Phragmites australis expansion and effects of experimental Phragmites control on vegetation, macroinvertebrates, and fish within tidelands of the lower Connecticut River. Estuaries 24: 90–107.
Webster, J. R. and E. F. Benfield. 1986. Vascular plant breakdown in freshwater ecosystems. Annual Review of Ecology and Systematics 17: 567–594.
Wetzel, R. G. and M. J. Howe. 1999. High production in a herbaceous perennial plant achieved by continuous growth and synchronized population dynamics. Aquatic Botany 64: 111–129.
Whyte, R. S. 1996. The vegetation dynamics of a freshwater estuary on Lake Erie: The Old Woman Creek State Nature Preserve and National Estuarine Research Reserve, Huron, OH. Ph.D. Dissertation. Miami University, Oxford, OH, USA.
Whyte, R. S., D. A. Franco, and D. M. Klarer. 1997. Distribution of the floating-leaf macrophyte Nelumbo lutea (American water lotus) in a coastal wetland on Lake Erie. Wetlands 17: 567–573.
Whyte, R. S., D. A. Franco, and D. M. Klarer. 2003. The aquatic vegetation of the Old Woman Creek National Estuarine Research Reserve (Huron, Ohio): a Lake Erie coastal wetland. The Michigan Botanist 42: 63–84.
Wisheu, I. C. and P. A. Keddy. 1992. Competition and centrifugal organization of plant communities: Theory and tests. Journal of Vegetation Science 3: 147–156.
Wilkinson, L. 2000. SYSTAT 9. SPSS, Chicago, IL, USA.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kulesza, A.E., Holomuzki, J.R. Amphipod performance responses to decaying leaf litter of Phragmites australis and Typha angustifolia from a Lake Erie coastal marsh. Wetlands 26, 1079–1088 (2006). https://doi.org/10.1672/0277-5212(2006)26[1079:APRTDL]2.0.CO;2
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1672/0277-5212(2006)26[1079:APRTDL]2.0.CO;2