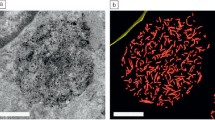
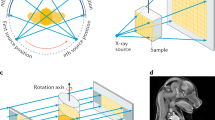
Abstract
The intricate hierarchical microstructures typical of biological materials give rise to fascinating combinations of anisotropic mechanical properties that can surpass those of man-made materials. The linking together of three-dimensional (3D) imaging techniques has the potential to reveal these structures in unprecedented detail. However, a complete understanding can only be reached if the relationship between structure and mechanical properties can be elucidated. X-ray-computed tomography (CT) is uniquely placed to image such structures across a wide range of length scales. We review recent technical advances that are leading to improved contrast and spatial resolution. We highlight how time-lapse CT 3D studies can track the response of hierarchical microstructures to mechanical loading.





Similar content being viewed by others
References
P. Fratzl, R. Weinkamer, Prog. Mater. Sci. 52, 1263 (2007).
A.H. Barber, D. Lu, N.M. Pugno, J. R. Soc. Interface 12, 20141326 (2015).
M.A. Meyers, P.-Y. Chen, M.I. Lopez, Y. Seki, A.Y.M. Lin, J. Mech. Behav. Biomed. Mater. 4, 626 (2011).
J.F.V. Vincent, Proc. Inst. Mech. Eng. H J. Eng. Med. 223, 919 (2009).
Q. Chen, N.M. Pugno, J. Mech. Behav. Biomed. Mater. 19, 3 (2013).
F. Barthelat, Int. Mater. Rev. 60, 413 (2015).
T.L. Burnett, S.A. McDonald, A. Gholinia, R. Geurts, M. Janus, T. Slater, S.J. Haigh, C. Ornek, F. Almuaili, D.L. Engelberg, G.E. Thompson, P.J. Withers, Sci. Rep. 4, 4711 (2014).
V. Ntziachristos, Nat. Methods 7, 603 (2010).
J.J. Socha, M.W. Westneat, J.F. Harrison, J.S. Waters, W.-K. Lee, BMC Biol. 5, 1 (2007).
M.R. Howells, T. Beetz, H.N. Chapman, C. Cui, J.M. Holton, C.J. Jacobsen, J. Kirz, E. Lima, S. Marchesini, H. Miao, D. Sayre, D.A. Shapiro, J.C.H. Spence, D. Starodub, J. Electron. Spectrosc. Relat. Phenom. 170, 4 (2009).
R. Carzaniga, M.-C. Domart, L.M. Collinson, E. Duke, Protoplasma 251, 449 (2014).
B. Alberto, C. Paola, S. Pekka, Phys. Med. Biol. 58, R1 (2013).
S. Zabler, P. Cloetens, J.-P. Guigay, J. Baruchel, M. Schlenker, Rev. Sci. Instrum. 76, 073705 (2005).
T. Weitkamp, A. Diaz, C. David, F. Pfeiffer, M. Stampanoni, P. Cloetens, E. Ziegler, Opt. Express 13, 6296 (2005).
A. Tkachuk, F. Duewer, H. Cui, M. Feser, S. Wang, W. Yun, Z. Kristallogr. Cryst. Mater. 222, 650 (2007).
C.K. Hagen, P. Maghsoudlou, G. Totonelli, P.C. Diemoz, M. Endrizzi, L. Rigon, R.-H. Menk, F. Arfelli, D. Dreossi, E. Brun, P. Coan, A. Bravin, P. De Coppi, A. Olivo, Sci. Rep. 5, 18156 (2015).
L.A. Walton, R.S. Bradley, P.J. Withers, V.L. Newton, R.E.B. Watson, C. Austin, M.J. Sherratt, Sci. Rep. 5, 10074 (2015).
G. Poologasundarampillai, D. Wang, S. Li, J. Nakamura, R. Bradley, P.D. Lee, M.M. Stevens, D.S. McPhail, T. Kasuga, J.R. Jones, Acta Biomater. 10, 3733 (2014).
F. Pfeiffer, T. Weitkamp, O. Bunk, C. David, Nat. Phys. 2, 258 (2006).
G. Schulz, T. Weitkamp, I. Zanette, F. Pfeiffer, F. Beckmann, C. David, S. Rutishauser, E. Reznikova, B. Müller, J. R. Soc. Interface 7, 1665 (2010).
I. Zanette, T. Zhou, A. Burvall, U. Lundström, D.H. Larsson, M. Zdora, P. Thibault, F. Pfeiffer, H.M. Hertz, Phys. Rev. Lett. 112, 253903 (2014).
H. Wang, Y. Kashyap, K. Sawhney, Sci. Rep. 6, 20476 (2016).
M. Langer, A. Pacureanu, H. Suhonen, Q. Grimal, P. Cloetens, F. Peyrin, PLoS One 7, e35691 (2012).
S. Marchesini, H.N. Chapman, S.P. Hau-Riege, R.A. London, A. Szoke, H. He, M.R. Howells, H. Padmore, R. Rosen, J.C.H. Spence, U. Weierstall, Opt. Express 11, 2344 (2003).
F. Berenguer, R.J. Bean, L. Bozec, J. Vila-Comamala, F. Zhang, C.M. Kewish, O. Bunk, J.M. Rodenburg, I.K. Robinson, Biophys. J. 106, 459 (2014).
I. Zanette, B. Enders, M. Dierolf, P. Thibault, R. Gradl, A. Diaz, M. Guizar-Sicairos, A. Menzel, F. Pfeiffer, P. Zaslansky, Sci. Rep. 5, 9210 (2015).
M. Dierolf, A. Menzel, P. Thibault, P. Schneider, C.M. Kewish, R. Wepf, O. Bunk, F. Pfeiffer, Nature 467, 436 (2010).
K. Giewekemeyer, C. Hackenberg, A. Aquila, R.N. Wilke, M.R. Groves, R. Jordanova, V.S. Lamzin, G. Borchers, K. Saksl, A.V. Zozulya, M. Sprung, A.P. Mancuso, Biophys. J. 109, 1986 (2015).
L. Shemilt, E. Verbanis, J. Schwenke, A.K. Estandarte, G. Xiong, R. Harder, N. Parmar, M. Yusuf, F. Zhang, I.K. Robinson, Biophys. J. 108, 706 (2015).
B.D. Metscher, BMC Physiol. 9, 1 (2009).
P. Das Neves Borges, A.E. Forte, T.L. Vincent, D. Dini, M. Marenzana, Osteoarthr. Cartil. 22, 1419 (2014).
T.D. Butters, S.J. Castro, T. Lowe, Y. Zhang, M. Lei, P.J. Withers, H. Zhang, PLoS One 9, e105552 (2014).
O.V. Aslanidi, T. Nikolaidou, Z. Jichao, B.H. Smaill, S.H. Gilbert, A.V. Holden, T. Lowe, P.J. Withers, R.S. Stephenson, J.C. Jarvis, J.C. Hancox, M.R. Boyett, Z. Henggui, IEEE Trans. Med. Imaging 32, 8 (2013).
T. Shearer, S. Rawson, S.J. Castro, R. Balint, R.S. Bradley, T. Lowe, J. Vila-Comamala, P.D. Lee, S.H. Cartmell, Muscles Ligaments Tendons J. 4, 238 (2014).
J.M. de Silva, I. Zanette, P.B. Noël, M.B. Cardoso, M.A. Kimm, F. Pfeiffer, Sci. Rep. 5, 14088 (2015).
L.E. Cole, R.D. Ross, J.M.R. Tilley, T. Vargo-Gogola, R.K. Roeder, Nanomedicine 10, 321 (2015).
J.C. De La Vega, U.O. Häfeli, Contrast Media Mol. Imaging 10, 81 (2015).
R. Popovtzer, A. Agrawal, N.A. Kotov, A. Popovtzer, J. Balter, T.E. Carey, R. Kopelman, Nano Lett. 8, 4593 (2008).
C. Hagen, S. Werner, S. Carregal-Romero, A.N. Malhas, B.G. Klupp, P. Guttmann, S. Rehbein, K. Henzler, T.C. Mettenleiter, D.J. Vaux, W.J. Parak, G. Schneider, K. Grunewald, Ultramicroscopy 146, 46 (2014).
P.J. Withers, Mater. Today 10, 26 (2007).
M. Bertilson, O. von Hofsten, U. Vogt, A. Holmberg, A.E. Christakou, H.M. Hertz, Opt. Lett. 36, 2728 (2011).
D.B. Carlson, J. Gelb, V. Palshin, J.E. Evans, Microsc. Microanal. 19, 22 (2013).
M. Uchida, Y. Sun, G. McDermott, C. Knoechel, M.A. Le Gros, D. Parkinson, D.G. Drubin, C.A. Larabell, Yeast 28, 227 (2011).
G. Schneider, P. Guttmann, S. Heim, S. Rehbein, F. Mueller, K. Nagashima, J.B. Heymann, W.G. Muller, J.G. McNally, Nat. Methods 7, 985 (2010).
M. Obst, G. Schmid, “3D Chemical Mapping: Application of Scanning Transmission (Soft) X-Ray Microscopy (STXM) in Combination with Angle-Scan Tomography in Bio-, Geo-, and Environmental Sciences,” in Electron Microscopy: Methods and Protocols, J. Kuo, Ed. (Humana Press, Totowa, NJ, 2014) pp. 757–781.
J. Cosmidis, K. Benzerara, N. Nassif, T. Tyliszczak, F. Bourdelle, Acta Biomater. 12, 260 (2015).
D. Hernández Cruz, M.-E. Rousseau, M.M. West, M. Pézolet, A.P. Hitchcock, Biomacromolecules 7, 836 (2006).
J. Normandeau, C. Van Kessel, D. Nicholson, B. Rahusaar Routledge, A. Fawcett, L. Lim-Cole, C. Condy, N. Sylvain, T. Walker, F. Borondics, Can. Young Sci. J. 2014, 35 (2014).
A. Kyrieleis, V. Titarenko, M. Ibison, T. Connolley, P.J. Withers, J. Microsc. 241, 69 (2011).
A. Patwardhan, A. Ashton, R. Brandt, S. Butcher, R. Carzaniga, W. Chiu, L. Collinson, P. Doux, E. Duke, M.H. Ellisman, E. Franken, K. Grunewald, J.-K. Heriche, A. Koster, W. Kuhlbrandt, I. Lagerstedt, C. Larabell, C.L. Lawson, H.R. Saibil, E. Sanz-Garcia, S. Subramaniam, P. Verkade, J.R. Swedlow, G.J. Kleywegt, Nat. Struct. Mol. Biol. 21, 841 (2014).
J. Caplan, M. Niethammer, R.M. Taylor, K.J. Czymmek, Curr. Opin. Struct. Biol. 21, 686 (2011).
B.N.G. Giepmans, Histochem. Cell Biol. 130, 211 (2008).
A. Merkle, L. Lechner, A. Steinbach, J. Gelb, M. Kienle, M. Phaneuf, D. Unrau, S. Singh, N. Chawla, Microsc. Anal. 28, S10 (2014).
S. Handschuh, N. Baeumler, T. Schwaha, B. Ruthensteiner, Front. Zool. 10, 1 (2013).
G. Schneider, P. Guttmann, S. Rehbein, S. Werner, R. Follath, J. Struct. Biol. 177, 212 (2012).
E.A. Smith, G. McDermott, M. Do, K. Leung, B. Panning, M. Le Gros, C.A. Larabell, Biophys. J. 107, 1988 (2014).
M. Liebi, M. Georgiadis, A. Menzel, P. Schneider, J. Kohlbrecher, O. Bunk, M. Guizar-Sicairos, Nature 527, 349 (2015).
P.J. Withers, M. Preuss, Annu. Rev. Mater. Res. 42, 81 (2012).
E. Maire, P.J. Withers, Int. Mater. Rev. 59, 1 (2014).
H.D. Barth, E.A. Zimmermann, E. Schaible, S.Y. Tang, T. Alliston, R.O. Ritchie, Biomaterials 32, 8892 (2011).
T. Lowe, R.J. Garwood, T.J. Simonsen, R.S. Bradley, P.J. Withers, J. R. Soc. Interface 10, 20130304 (2013).
S.M. Walker, D.A. Schwyn, R. Mokso, M. Wicklein, T. Müller, M. Doube, M. Stampanoni, H.G. Krapp, G.K. Taylor, PLoS Biol. 12, e1001823 (2014).
A.T. Jang, J.D. Lin, Y. Seo, S. Etchin, A. Merkle, K. Fahey, S.P. Ho, J. Vis. Exp. 85, e51147 (2014).
W. Wiest, S. Zabler, A. Rack, C. Fella, A. Balles, K. Nelson, R. Schmelzeisen, R. Hanke, J. Synchrotron Radiat. 22, 1492 (2015).
B. Patterson, N.L. Cordes, K. Henderson, J.C.E. Mertens, A.J. Clarke, X. Lu, R. Hartwell, B. Hornberger, A. Merkle, S. Etchin, A. Tkachuk, M. Leibowitz, D. Trapp, W. Qiu, B. Zhang, H. Bale, P.J. Withers, R.S. Bradley, Exp. Mech. (forthcoming).
U.G.K. Wegst, H. Bai, E. Saiz, A.P. Tomsia, R.O. Ritchie, Nat. Mater. 14, 23 (2015).
A. Ilnicka, J.P. Lukaszewicz, Front. Mater. 2, 20 (2015), doi:10.3389/fmats. 2015.00020.
S. Heinzer, T. Krucker, M. Stampanoni, R. Abela, E.P. Meyer, A. Schuler, P. Schneider, R. Müller, Neuroimage 32, 626 (2006).
Acknowledgments
The authors would like to thank EPSRC funding for the Henry Moseley X-Ray Imaging Facility under Grants EP/M010619, EP/K004530, EP/F007906, and EP/F028431. R.S.B. is grateful for Zeiss for funding his research fellowship.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Bradley, R.S., Withers, P.J. Correlative multiscale tomography of biological materials. MRS Bulletin 41, 549–556 (2016). https://doi.org/10.1557/mrs.2016.137
Published:
Issue Date:
DOI: https://doi.org/10.1557/mrs.2016.137