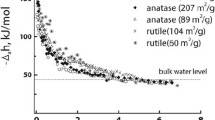
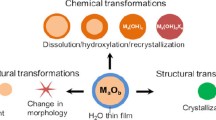
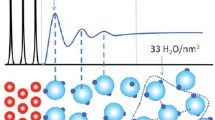
Abstract
The interaction between water and oxide surfaces plays an important role in many technological applications and environmental processes. However, gaining fundamental understanding of processes at oxide—water interfaces is challenging because of the complexity of the systems. To this end, results of experimental and computational studies utilizing well-defined oxide surfaces help to gain molecular-scale insights into the properties and reactivity of water on oxide surfaces. This is a necessary basis for the understanding of oxide surface chemistry in more complex environments. This review highlights recent advances in the fundamental understanding of oxide—water interaction using surface science experiments. In particular, we will discuss the results on crystalline and well-defined supported thin film oxide samples of the alkaline earth oxides (MgO and CaO), silica (SiO2), and magnetite (Fe3O4). Several aspects of water—oxide interactions such as adsorption modes (molecular versus dissociative), formation of long-range ordered structures, and dissolution processes will be discussed.















Similar content being viewed by others
References
P.A. Thiel and T.E. Madey: The interaction of water with solid surfaces—Fundamental aspects. Surf. Sci. Rep. 7, 211 (1987).
M.A. Henderson: The interaction of water with solid surfaces: Fundamental aspects revisited. Surf. Sci. Rep. 46, 1 (2002).
A. Verdaguer, G.M. Sacha, H. Bluhm, and M. Salmeron: Molecular structure of water at interfaces: Wetting at the nanometer scale. Chem. Rev. 106, 1478 (2006).
G.E. Ewing: Ambient thin film water on insulator surfaces. Chem. Rev. 106, 1511 (2006).
G.E. Brown, V.E. Henrich, W.H. Casey, D.L. Clark, C. Eggleston, A. Felmy, D.W. Goodman, M. Grätzel, G. Maciel, M.I. McCarthy, K.H. Nealson, D.A. Sverjensky, M.F. Toney, and J.M. Zachara: Metal oxide surfaces and their interactions with aqueous solutions and microbial organisms. Chem. Rev. 99, 77 (1999).
E. Bjornehohn, M.H. Hansen, A. Hodgson, L.M. Liu, D.T. Limmer, A. Michaelides, P. Pedevilla, J. Rossmeisl, H. Shen, G. Tocci, E. Tyrode, M.M. Walz, J. Werner, and H. Bluhm: Water at interfaces. Chem. Rev. 116, 7698 (2016).
A. Hodgson and S. Haq: Water adsorption and the wetting of metal surfaces. Surf. Sci. Rep. 64, 381 (2009).
J. Carrasco, A. Hodgson, and A. Michaelides: A molecular perspective of water at metal interfaces. Nat. Mater. 11, 667 (2012).
R.R. Rao, M.J. Kolb, J. Hwang, A.F. Pedersen, A. Mehta, H. You, K.A. Stoerzinger, Z.X. Feng, H. Zhou, H. Bluhm, L. Giordano, I.E.L. Stephens, and Y. Shao-Horn: Surface orientation dependent water dissociation on rutile ruthenium dioxide. J. Phys. Chem. C 122, 17802 (2018).
M. Schwarz, F. Faisal, S. Mohr, C. Hohner, K. Werner, T. Xu, T. Skala, N. Tsud, K.C. Prince, V. Matolin, Y. Lykhach, and J. Libuda: Structure-dependent dissociation of water on cobalt oxide. J. Phys. Chem. Lett. 9, 2763 (2018).
X.L. Hu, J. Carrasco, J. Klimes, and A. Michaelides: Trends in water monomer adsorption and dissociation on flat insulating surfaces. Phys. Chem. Chem. Phys. 13, 12447 (2011).
R.T. Mu, Z.J. Zhao, Z. Dohnalek, and J.L. Gong: Structural motifs of water on metal oxide surfaces. Chem. Soc. Rev. 46, 1785 (2017).
M. Meier, J. Hulva, Z. Jakub, J. Pavelec, M. Setvin, R. Bliem, M. Schmid, U. Diebold, C. Franchini, and G.S. Parkinson: Water agglomerates on Fe3O4(001). Proc. Natl. Acad. Sci. U. S. A 115, E5642 (2018).
R. Wlodarczyk, M. Sierka, K. Kwapien, J. Sauer, E. Carrasco, A. Aumer, J.F. Gomes, M. Sterrer, and H-J. Freund: Structures of the ordered water monolayer on MgO(001). J. Phys. Chem. C 115, 6764 (2011).
P. Fenter and N.C. Sturchio: Mineral-water interfacial structures revealed by synchrotron X-ray scattering. Prog. Surf. Sci. 77, 171 (2004).
J. Heidberg, B. Redlich, and D. Wetter: Adsorption of water vapor on the MgO(100) single-crystal surface. Ber. Bunsenges. Phys. Chem. 99, 1333 (1995).
B. Meyer, D. Marx, O. Dulub, U. Diebold, M. Kunat, D. Langenberg, and C. Wöll: Partial dissociation of water leads to stable superstructures on the surface of zinc oxide. Angew. Chem., Int. Ed. 43, 6642 (2004).
I.M. Brookes, C.A. Muryn, and G. Thornton: Imaging water dissociation on TiO2(110). Phys. Rev. Lett. 87, 266103 (2001).
J. Balajka, M.A. Hines, W.J.I. DeBenedetti, M. Komora, J. Pavelec, M. Schmid, and U. Diebold: High-affinity adsorption leads to molecularly ordered interfaces on TiO2 in air and solution. Science 361, 786 (2018).
U. Diebold: The surface science of titanium dioxide. Surf. Sci. Rep. 48, 53 (2003).
G.A. Kimmel, M. Baer, N.G. Petrik, J. VandeVondele, R. Rousseau, and C.J. Mundy: Polarization- and azimuth-resolved infrared spectroscopy of water on TiO2(110): Anisotropy and the hydrogen-bonding network. J. Phys. Chem. Lett. 3, 778 (2012).
N.G. Petrik and G.A. Kimmel: Reaction kinetics of water molecules with oxygen vacancies on rutile TiO2(110). J. Phys. Chem. C 119, 23059 (2015).
Y.M. Wang and C. Wöll: IR spectroscopic investigations of chemical and photochemical reactions on metal oxides: Bridging the materials gap. Chem. Soc. Rev. 46, 1875 (2017).
H.J. Freund, H. Kuhlenbeck, and V. Staemmler: Oxide surfaces. Rep. Prog. Phys. 59, 283 (1996).
C.T. Campbell: Ultrathin metal films and particles on oxide surfaces: Structural, electronic and chemisorption properties. Surf. Sci. Rep. 27, 1 (1997).
F.P. Netzer and A. Fortunelli, eds.: Oxide Materials at the Two-dimensional Limit, Springer Series in Materials Science, Vol. 234 (Springer, Switzerland, 2016).
N. Nilius: Properties of oxide thin films and their adsorption behavior studied by scanning tunneling microscopy and conductance spectroscopy. Surf. Sci. Rep. 64, 595 (2009).
S. Surnev, M.G. Ramsey, and F.P. Netzer: Vanadium oxide surface studies. Prog. Surf. Sci. 73, 117 (2003).
G. Kresse, M. Schmid, E. Napetschnig, M. Shishkin, L. Köhler, and P. Varga: Structure of the ultrathin aluminum oxide film on NiAl(110). Science 308, 1440 (2005).
C.R. Henry: Surface studies of supported model catalysts. Surf. Sci. Rep. 31, 231 (1998).
S. Schintke, S. Messerli, M. Pivetta, F. Patthey, L. Libioulle, M. Stengel, A. De Vita, and W-D. Schneider: Insulator at the ultrathin limit: MgO on Ag(001). Phys. Rev. Lett. 87, 276801 (2001).
M. Klaua, D. Ullmann, J. Barthel, W. Wulfhekel, J. Kirschner, R. Urban, T.L. Monchesky, A. Enders, J.F. Cochran, and B. Heinrich: Growth, structure, electronic, and magnetic properties of MgO/Fe(001) bilayers and Fe/MgO/Fe(001) trilayers. Phys. Rev. B 64, 134411 (2001).
M.C. Wu, J.S. Corneille, C.A. Estrada, J.W. He, and D.W. Goodman: Synthesis and characterization of ultra-thin MgO films on Mo(100). Chem. Phys. Lett. 182, 472 (1991).
S. Benedetti, H.M. Benia, N. Nilius, S. Valeri, and H.J. Freund: Morphology and optical properties of MgO thin films on Mo(001). Chem. Phys. Lett. 430, 330 (2006).
X. Shao, P. Myrach, N. Nilius, and H.J. Freund: Growth and morphology of calcium-oxide films grown on Mo(001). J. Phys. Chem. C 115, 8784 (2011).
N. Nilius, S. Benedetti, Y. Pan, P. Myrach, C. Noguera, L. Giordano, and J. Goniakowski: Electronic and electrostatic properties of polar oxide nanostructures: MgO(111) islands on Au(111). Phys. Rev. B 86, 205410 (2012).
J. Goniakowski, C. Finocchi, and C. Noguera: Polarity of oxide surfaces and nanostructures. Rep. Prog. Phys. 71, 016501 (2008).
F. Finocchi, A. Barbier, J. Jupille, and C. Noguera: Stability of rocksalt (111) polar surfaces: Beyond the octopole. Phys. Rev. Lett. 92, 136101 (2004).
J. Pal, M. Smerieri, E. Celasco, L. Savio, L. Vattuone, and M. Rocca: Morphology of monolayer MgO films on Ag(100): Switching from corrugated islands to extended flat terraces. Phys. Rev. Lett. 112, 126102 (2014).
J. Pal, M. Smerieri, E. Celasco, L. Savio, L. Vattuone, R. Ferrando, S. Tosoni, L. Giordano, G. Pacchioni, and M. Rocca: How growing conditions and interfacial oxygen affect the final morphology of MgO/Ag(100) films. J. Phys. Chem. C 118, 26091 (2014).
K.P. McKenna and A.L. Shluger: Electron-trapping polycrystalline materials with negative electron affinity. Nat. Mater. 7, 859 (2008).
S. Benedetti, P. Torelli, S. Valeri, H.M. Benia, N. Nilius, and G. Renaud: Structure and morphology of thin MgO films on Mo(001). Phys. Rev. B 78, 195411 (2008).
S. Benedetti, F. Stavale, S. Valeri, C. Noguera, H.J. Freund, J. Goniakowski, and N. Nilius: Steering the growth of metal ad-particles via interface interactions between a MgO thin film and a Mo support. Adv. Funct. Mater. 23, 75 (2013).
L. Giordano, F. Cinquini, and G. Pacchioni: Tuning the surface metal work function by deposition of ultrathin oxide films: Density functional calculations. Phys. Rev. B 73, 045414 (2006).
H.M. Benia, P. Myrach, N. Nilius, and H.J. Freund: Structural and electronic characterization of the MgO/Mo(001) interface using STM. Surf. Sci. 604, 435 (2010).
M. Sterrer, E. Fischbach, T. Risse, and H-J. Freund: Geometric characterization of a singly charged oxygen vacancy on a single-crystalline MgO(001) film by electron paramagnetic resonance spectroscopy. Phys. Rev. Lett. 94, 186101 (2005).
M. Sterrer, E. Fischbach, M. Heyde, N. Nilius, H.P. Rust, T. Risse, and H.J. Freund: Electron paramagnetic resonance and scanning tunneling microscopy investigations on the formation of F+ and F0 color centers on the surface of thin MgO(001) films. J. Phys. Chem. B 110, 8665 (2006).
X. Shao, N. Nilius, P. Myrach, H.J. Freund, U. Martinez, S. Prada, L. Giordano, and G. Pacchioni: Strain-induced formation of ultrathin mixed-oxide films. Phys. Rev. B 83, 245407 (2011).
Y. Cui, X. Shao, M. Baldofski, J. Sauer, N. Nilius, and H.J. Freund: Adsorption, activation, and dissociation of oxygen on doped oxides. Angew. Chem., Int. Ed. 52, 11385 (2013).
S. Sastry, P.G. Debenedetti, and F.H. Stillinger: Signatures of distinct dynamical regimes in the energy landscape of a glass-forming liquid. Nature 393, 554 (1998).
R. Zallen: The Physics of Amorphous Solids (Wiley-VCH Verlag GmbH & Co. KGaA, Weinheim, 2004).
L. Berthier and G. Biroli: Theoretical perspective on the glass transition and amorphous materials. Rev. Mod. Phys. 83, 587 (2011).
J.C. Mauro and E.D. Zanotto: Two centuries of glass research: Historical trends, current status, and grand challenges for the future. Int. J. Appl. Glass Sci. 5, 313 (2014).
W.H. Zachariasen: The atomic arrangement in glass. J. Am. Chem. Soc. 54, 3841 (1932).
L. Lichtenstein, M. Heyde, and H-J. Freund: Atomic arrangement in two-dimensional silica: From crystalline to vitreous structures. J. Phys. Chem. C 116, 20426 (2012).
D. Löffler, J.J. Uhlrich, M. Baron, B. Yang, X. Yu, L. Lichtenstein, L. Heinke, C. Büchner, M. Heyde, S. Shaikhutdinov, H-J. Freund, R. Włodarczyk, M. Sierka, and J. Sauer: Growth and structure of crystalline silica sheet on Ru(0001). Phys. Rev. Lett. 105, 146104 (2010).
L. Lichtenstein, C. Büchner, B. Yang, S. Shaikhutdinov, M. Heyde, M. Sierka, R. Włodarczyk, J. Sauer, and H-J. Freund: The atomic structure of a metal-supported vitreous thin silica film. Angew. Chem., Int. Ed. 51, 404 (2012).
L. Lichtenstein, M. Heyde, and H-J. Freund: Crystalline-vitreous interface in two dimensional silica. Phys. Rev. Lett. 109, 106101 (2012).
X. Yu, B. Yang, J.A. Boscoboinik, S. Shaikhutdinov, and H-J. Freund: Support effects on the atomic structure of ultrathin silica films on metals. Appl. Phys. Lett. 100, 151608 (2012).
P.Y. Huang, S. Kurasch, A. Srivastava, V. Skakalova, J. Kotakoski, A.V. Krasheninnikov, R. Hovden, Q. Mao, J.C. Meyer, J. Smet, D.A. Muller, and U. Kaiser: Direct imaging of a two-dimensional silica glass on graphene. Nano Lett. 12, 1081 (2012).
E.I. Altman and U.D. Schwarz: Structural and electronic heterogeneity of two-dimensional amorphous silica layers. Adv. Mater. Interfaces 1, 1400108 (2014).
R.M. Cornell and U. Schwertmann: The Iron Oxides (Wiley-VCH Verlag GmbH & Co. KGaA, Weinheim, 2004); pp. 1–7.
G. Ertl, H. Knözinger, F. Schueth, and J. Weitkamp, eds.: Handbook of Heterogeneous Catalysis, Vol. 2, compl. rev. and enlarged ed. (WILEY-VCH Verlag GmbH & Co. KGaA, Weinheim, 2008).
R. Zboril, M. Mashlan, and D. Petridis: Iron(III) oxides from thermal processes: Synthesis, structural and magnetic properties, Mössbauer spectroscopy characterization, and applications. Chem. Mater. 14, 969 (2002).
W. Weiss and W. Ranke: Surface chemistry and catalysis on well-defined epitaxial iron-oxide layers. Prog. Surf. Sci. 70, 1 (2002).
N.G. Condon, F.M. Leibsle, T. Parker, A.R. Lennie, D.J. Vaughan, and G. Thornton: Biphase ordering on Fe3O4(111). Phys. Rev. B 55, 15885 (1997).
A. Barbieri, W. Weiss, M.A.V. Hove, and G.A. Somorjai: Magnetite Fe3O4(111): Surface structure by LEED crystallography and energetics. Surf. Sci. 302, 259 (1994).
G.S. Parkinson: Iron oxide surfaces. Surf. Sci. Rep. 71, 272 (2016).
A. Sala, H. Marchetto, Z.H. Qin, S. Shaikhutdinov, T. Schmidt, and H-J. Freund: Defects and inhomogeneities in Fe3O4(111) thin film growth on Pt(111). Phys. Rev. B 86, 155430 (2012).
M. Melzer, J. Urban, H. Sack-Kongehl, K. Weiss, H-J. Freund, and R. Schlögl: Preparation of vanadium and vanadium oxide clusters by means of inert gas aggregation. Catal. Lett. 81, 219 (2002).
W. Weiss and M. Ritter: Metal oxide heteroepitaxy: Stranski-Krastanov growth of iron oxides on Pt(111). Phys. Rev. B 59, 5201 (1999).
D.T. Margulies, F.T. Parker, M.L. Rudee, F.E. Spada, J.N. Chapman, P.R. Aitchison, and A.E. Berkowitz: Origin of the anomalous magnetic behavior in single crystal Fe3O4 films. Phys. Rev. Lett. 79, 5201 (1997).
M. Ritter and W. Weiss: Fe3O4(111) surface structure determined by LEED crystallography. Surf. Sci. 432, 81 (1999).
S.K. Shaikhutdinov, M. Ritter, X.G. Wang, H. Over, and W. Weiss: Defect structures on epitaxial Fe3O4(111) films. Phys. Rev. B 60, 11062 (1999).
C. Lemire, R. Meyer, V.E. Henrich, S. Shaikhutdinov, and H.J. Freund: The surface structure of Fe3O4(111) films as studied by CO adsorption. Surf. Sci. 572, 103 (2004).
X. Li, J. Paier, J. Sauer, F. Mirabella, E. Zaki, F. Ivars-Barceló, S. Shaikhutdinov, and H.J. Freund: Surface termination of Fe3O4(111) films studied by CO adsorption revisited. J. Phys. Chem. B 122, 527 (2018).
X. Zhao, X. Shao, Y. Fujimori, S. Bhattacharya, L.M. Ghiringhelli, H-J. Freund, M. Sterrer, N. Nilius, and S.V. Levchenko: Formation of water chains on CaO(001): What drives the 1D growth? J. Phys. Chem. Lett. 6, 1204 (2015).
J. Carrasco, F. Illas, and N. Lopez: Dynamic ion pairs in the adsorption of isolated water molecules on alkaline-earth oxide(001) surfaces. Phys. Rev. Lett. 100, 016101 (2008).
Y. Fujimori, X. Zhao, X. Shao, S.V. Levchenko, N. Nilius, M. Sterrer, and H-J. Freund: Interaction of water with the CaO(001) surface. J. Phys. Chem. C 120, 5565 (2016).
D. Ferry, A. Glebov, V. Senz, J. Suzanne, J.P. Toennies, and H. Weiss: Observation of the second ordered phase of water on the MgO(100) surface: Low energy electron diffraction and helium atom scattering studies. J. Chem. Phys. 105, 1697 (1996).
D. Halwidl, B. Stöger, W. Mayr-Schmölzer, J. Pavelec, D. Fobes, J. Peng, Z.Q. Mao, G.S. Parkinson, M. Schmid, F. Mittendorfer, J. Redinger, and U. Diebold: Adsorption of water at the SrO surface of ruthenates. Nat. Mater. 15, 450 (2016).
Y.D. Kim, R.M. Lynden-Bell, A. Alavi, J. Stulz, and D.W. Goodman: Evidence for partial dissociation of water on flat MgO(100) surfaces. Chem. Phys. Lett. 352, 318 (2002).
U. Leist, W. Ranke, and K. Al-Shamery: Water adsorption and growth of ice on epitaxial Fe3O4(111), FeO(111), and Fe2O3(biphase). Phys. Chem. Chem. Phys. 5, 2435 (2003).
Y. Joseph, C. Kuhrs, W. Ranke, M. Ritter, and W. Weiss: Adsorption of water on FeO(111) and Fe3O4(111): Identification of active sites for dissociation. Chem. Phys. Lett. 314, 195 (1999).
Y. Joseph, W. Ranke, and W. Weiss: Water on FeO(111) and Fe3O4(111): Adsorption behavior on different surface terminations. J. Phys. Chem. B 104, 3224 (2000).
P.A. Redhead: Thermal desorption of gases. Vacuum 12, 203 (1962).
A.M. de Jong and J.W. Niemantsverdriet: Thermal desorption analysis: Comparative test of ten commonly applied procedures. Surf. Sci. 233, 355 (1990).
F. Mirabella, E. Zaki, F. Ivars-Barcelo, X. Li, J. Paier, J. Sauer, S. Shaikhutdinov, and H-J. Freund: Cooperative formation of long-range ordering in water ad-layers on Fe3O4(111). Angew. Chem., Int. Ed. 57, 1409 (2017).
E. Habenschaden and J. Küppers: Evaluation of flash desorption spectra. Surf. Sci. Lett. 138, L147 (1984).
S.L. Tait, Z. Dohnálek, C.T. Campbell, and B.D. Kay: n-alkanes on MgO(100). I. Coverage-dependent desorption kinetics of n-butane. J. Chem. Phys. 122, 164707 (2005).
P. Dementyev, K-H. Dostert, F. Ivars-Barceló, C.P. O’Brien, F. Mirabella, S. Schauermann, X. Li, J. Paier, J. Sauer, and H-J. Freund: Water interaction with iron oxides. Angew. Chem., Int. Ed. 54, 13942 (2015).
P. Liu, T. Kendelewicz, G.E. Brown, G.A. Parks, and P. Pianetta: Reaction of water with vacuum-cleaved CaO(100) surfaces: An X-ray photoemission spectroscopy study. Surf. Sci. 416, 326 (1998).
P. Liu, T. Kendelewicz, G.E. Gordon, and G.A. Parks: Reaction of water with MgO(100) surfaces. Part I: Synchrotron X-ray photoemission studies of low-defect surfaces. Surf. Sci. 412–13, 287 (1998).
E. Carrasco, M.A. Brown, M. Sterrer, H-J. Freund, K. Kwapien, M. Sierka, and J. Sauer: Thickness-dependent hydroxylation of MgO(001) thin films. J. Phys. Chem. C 114, 18207 (2010).
L. Savio, E. Celasco, L. Vattuone, M. Rocca, and P. Senet: MgO/Ag(100): Confined vibrational modes in the limit of ultrathin films. Phys. Rev. B 67, 075420 (2003).
F. Ringleb, Y. Fujimori, H.F. Wang, H. Ariga, E. Carrasco, M. Sterrer, H.J. Freund, L. Giordano, G. Pacchioni, and J. Goniakowski: Interaction of water with FeO(111)/Pt(111): Environmental effects and influence of oxygen. J. Phys. Chem. C 115, 19328 (2011).
L. Giordano, M. Lewandowski, I.M.N. Groot, Y.N. Sun, J. Goniakowski, C. Noguera, S. Shaikhutdinov, G. Pacchioni, and H.J. Freund: Oxygen-induced transformations of an FeO(111) film on Pt(111): A combined DFT and STM study. J. Phys. Chem. C 114, 21504 (2010).
Y.N. Sun, L. Giordano, J. Goniakowski, M. Lewandowski, Z.H. Qin, C. Noguera, S. Shaikhutdinov, G. Pacchioni, and H.J. Freund: The interplay between structure and CO oxidation catalysis on metal-supported ultrathin oxide films. Angew. Chem., Int. Ed. 49, 4418 (2010).
Y.N. Sun, Z.H. Qin, M. Lewandowski, E. Carrasco, M. Sterrer, S. Shaikhutdinov, and H.J. Freund: Monolayer iron oxide film on platinum promotes low temperature CO oxidation. J. Catal. 266, 359 (2009).
S. Shaikhutdinov and H.J. Freund: Ultrathin silica films on metals: The long and winding road to understanding the atomic structure. Adv. Mater. 25, 49 (2013).
B. Yang, E. Emmez, W.E. Kaden, X. Yu, J.A. Boscoboinik, M. Sterrer, S. Shaikhutdinov, and H-J. Freund: Hydroxylation of metal-supported sheet-like silica films. J. Phys. Chem. C 117, 8336 (2013).
X. Yu, E. Emmez, Q. Pan, B. Yang, S. Pomp, W.E. Kaden, M. Sterrer, S. Shaikhutdinov, H-J. Freund, I. Goikoetxea, R. Wlodarczyk, and J. Sauer: Electron stimulated hydroxylation of a metal supported silicate film. Phys. Chem. Chem. Phys. 18, 3755 (2016).
W.E. Kaden, S. Pomp, M. Sterrer, and H.J. Freund: Insights into silica bilayer hydroxylation and dissolution. Top. Catal. 60, 471 (2017).
L.T. Zhuravlev: The surface chemistry of amorphous silica. Zhuravlev model. Colloids Surf., A 173, 1 (2000).
B.R. Bickmore, J.C. Wheeler, B. Bates, K.L. Nagy, and D.L. Eggett: Reaction pathways for quartz dissolution determined by statistical and graphical analysis of macroscopic experimental data. Geochim. Cosmochim. Acta 72, 4521 (2008).
F. Ringleb, M. Sterrer, and H-J. Freund: Preparation of Pd—MgO model catalysts by deposition of Pd from aqueous precursor solutions onto Ag(001)-supported MgO(001) thin films. Appl. Catal., A 474, 186 (2014).
D.A. Vermilyea: Dissolution of MgO and Mg(OH)2 in aqueous solutions. J. Electrochem. Soc. 116, 1179 (1969).
O.S. Pokrovsky and J. Schott: Experimental study of brucite dissolution and precipitation in aqueous solutions: Surface speciation and chemical affinity control. Geochim. Cosmochim. Acta 68, 31 (2004).
H.F. Wang, H. Ariga, R. Dowler, M. Sterrer, and H.J. Freund: Surface science approach to catalyst preparation—Pd deposition onto thin Fe3O4(111) films from PdCl2 precursor. J. Catal. 286, 1 (2012).
H.F. Wang, W.E. Kaden, R. Dowler, M. Sterrer, and H.J. Freund: Model oxide-supported metal catalysts—Comparison of ultrahigh vacuum and solution based preparation of Pd nanoparticles on a single-crystalline oxide substrate. Phys. Chem. Chem. Phys. 14, 11525 (2012).
K.M. Burson, L. Gura, B. Kell, C. Büchner, A.L. Lewandowski, M. Heyde, and H-J. Freund: Resolving amorphous solid-liquid interfaces by atomic force microscopy. Appl. Phys. Lett. 108, 201602 (2016).
F.F. Abraham and I.P. Batra: Theoretical interpretation of atomic-force- microscope images of graphite. Surf. Sci. 209, L125 (1989).
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Sterrer, M., Nilius, N., Shaikhutdinov, S. et al. Interaction of water with oxide thin film model systems. Journal of Materials Research 34, 360–378 (2019). https://doi.org/10.1557/jmr.2018.454
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1557/jmr.2018.454