Abstract
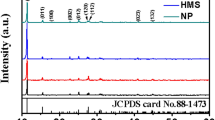
Shape control of nanocrystals has become an indispensable part in material research, such as developing new battery raw materials and synthesizing high activity catalysts. In this work, one-dimensional LiV3O8 nanorods have been fabricated by high temperature solid-state reaction using V2O5 nanowires as precursors obtained via a hydrothermal method. The as-prepared LiV3O8 nanorods were characterized by x-ray diffraction, transmission electron microscopy, scanning electron microscopy, and galvanostatic tests, compared with LiV3O8 samples synthesized by the traditional one-step solid-state method. The results show that LiV3O8 nanorods exhibited better electrochemical performance than those synthesized by the traditional method, indicating that a different shape will lead to huge distinctions in electrochemical properties. This work demonstrates that Li-insertion/deintercalation dynamics might be crystal morphology-sensitive.




Similar content being viewed by others
References
J.M. Tarascon and M. Armand: Issues and challenges facing rechargeable lithium batteries. Nature 414, 359 (2001).
X. Li, F. Cheng, B. Guo, and J. Chen: Template-synthesized LiCoO2, LiMn2O4, and LiNi0.8Co0.2O2 nanotubes as the cathode materials of lithium ion batteries. J. Phys. Chem. B 109, 14017 (2005).
K. Kang, Y.S. Meng, J. Breger, C.P. Grey, and G. Ceder: Electrodes with high power and high capacity for rechargeable lithium batteries. Science 311, 977 (2006).
M. Okubo, E. Hosono, J. Kim, M. Enomoto, N. Kojima, T. Kudo, H. Zhou, and I. Honma: Nanosize effect on high-rate Li-ion intercalation in LiCoO2 electrode. J. Am. Chem. Soc. 129, 7444 (2007).
F. Jiao, K.M. Shaju, and P.G. Bruce: Synthesis of nanowire and mesoporous low-temperature LiCoO2 by a post-templating reaction. Angew. Chem. Int. Ed. 44, 6550 (2005).
H. Chen and C.P. Grey: Molten salt synthesis and high rate performance of the “Desert-Rose” form of LiCoO2. Adv. Mater. 20, 2206 (2008).
K.S. Lee, S.T. Myung, and Y.K. Sun: Synthesis and electrochemical performances of core-shell structured Li[(M1/3Co1/3 Mn1/3)o.8(Ni1/2Mn1/2)o.2]02 cathode material for lithium ion batteries. J. Power Sources 195, 6043 (2010).
B. Kang and G. Ceder: Battery materials for ultrafast charging and discharging. Nature 458, 190 (2009).
Y. Hui, L. Juan, J.G. Zhang, and D.Z. Jia: Synthesis and properties of LiV3O8 nanomaterials as the cathode material for Li-ion battery. J. Inorg. Mater. 22, 447 (2007).
X.H. Liu, J.Q. Wang, J.Y. Zhang, and S.R. Yang: Sol-gel template synthesis of LiV3O8 nanowires. J. Mater. Sci. 42, 867 (2007).
H.W. Liu, H.M. Yang, and T. Huang: Synthesis, structure and electrochemical properties of one-dimensional nanometer materials LiV3O8. Med. Mai Infect. 37, 60 (2007).
T.J. Patey, S.H. Ng, R. Buchel, N. Tran, F. Krumeich, J. Wang, H. K. Liu, and P. Novak: Electrochemistry of LiV3O8 nanoparticles made by flame spray pyrolysis. Electrochem. Solid-State Lett. 11, A46 (2008).
D.S. Wang, T. Xie, and Y.D. Li: Nanocrystals: Solution-based synthesis and applications as nanocatalysts. Nano Res. 2, 30 (2009).
D.S. Wang, X. Wang, R. Xu, and Y.D. Li: Shape-dependent catalytic activity of CuO/MgO nanocatalysts. J. Nanosci. Nano-technol. 7, 3602 (2007).
D.S. Wang, R. Xu, X. Wang, and Y.D. Li: NiO nanorings and their unexpected catalytic property for CO oxidation. Nanotechnology 17, 979 (2006).
H. Yang, J. Li, X.G. Zhang, and Y.L. Jin: Synthesis of LiV3Og nanocrystallites as cathode materials for lithium ion batteries. J. Mater. Process. Technol. 207, 265 (2008).
K.P. Lee, K.M. Manesh, K.S. Kim, and A.Y. Gopalan: Synthesis and characterization of nanostructured wires (ID) to plates (3D) LiV3O8 combining sol-gel and electrospinning processes. J. Nanosci. Nanotechnol. 9, 417 (2009).
H.M. Liu, Y.G. Wang, K.X. Wang, Y.R. Wang, and H.S. Zhou: Synthesis and electrochemical properties of single-crystalline LiV3O8 nanorods as cathode materials for rechargeable lithium batteries. J. Power Sources 192, 668 (2009).
J.F. Liu, X. Wang, Q. Peng, and Y.D. Li: Vanadium pentoxide nanobelts: Highly selective and stable ethanol sensor materials. Adv. Mater. 17, 764 (2005).
D.S. Wang, C.H. Hao, W. Zheng, X.L. Ma, D.R. Chu, Q. Peng, and Y.D. Li: Bi2S3 nanotubes: Facile synthesis and growth mechanism. Nano Res. 2, 130 (2009).
C.H. Hao, D. Wang, W. Zheng, and Q. Peng: Growth and assembly of monodisperse Ag nanoparticles by exchanging the organic capping ligands. J. Mater. Res. 24, 352 (2009).
X.W. Lou, D. Deng, J.Y. Lee, J. Feng, and L.A. Archer: Self-supported formation of needlelike Co3O4 nanotubes and their application as lithium-ion battery electrodes. Adv. Mater. 20, 258 (2008).
Y.G. Li, B. Tan, and Y.Y. Wu: Mesoporous Co3O4 nanowire arrays for lithium ion batteries with high capacity and rate capability. Nano Lett. 8, 265 (2008).
D.K. Kim, P. Muralidharan, H.W. Lee, R. Ruffo, Y. Yang, C.K. Chan, H. Peng, R.A. Huggins, and Y. Cui: Spinel LiMn2O4 nanorods as lithium ion battery cathodes. Nano Lett. 8, 3948 (2008).
H.Y. Xu, H. Wang, Z.Q. Song, Y.W. Wang, H. Yan, and M. Yoshimura: Novel chemical method for synthesis of LiV3O8 nanorods as cathode materials for lithium ion batteries. Electro-chim. Acta 49, 349 (2004).
K. West, B. Zachau-Christiansen, S. Skaarup, Y. Saidi, J. Barker II, R. Olsen, R. Pynenburg, and R. Koksbang: Comparison of LiV3O8 cathode materials prepared by different methods. J. Electrochem. Soc. 143, 820 (1996).
X.L. Xiao, L. Wang, D.S. Wang, X.M. He, Q. Peng, and Y.D. Li: Hydrothermal synthesis of orthorhombic LiMnO2 nano-particles and LiMnO2 nanorods and comparison of their electrochemical performances. Nano Res. 2, 923 (2009).
D.S. Wang, X.L. Ma, Y.G. Wang, L. Wang, Z.Y. Wang, W. Zheng, X.M. He, J. Li, Q. Peng, and Y.D. Li: Shape control of CoO and LiCoO2 nanocrystals. Nano Res. 3, 1 (2010).
G. Pistoia, M. Pasquali, M. Tocci, R.V. Moshtev, and V. Maner: Li/ Li1+xV3O8 secondary batteries. 3. Further characterization of the mechanism of Li+ insertion and of the cycling behavior. J. Electrochem. Soc. 132, 281 (1985).
J. Kawakita, Y. Katayama, T. Miura, and T. Kishi: Lithium insertion behavior of Li1+xV3O8 prepared by precipitation technique in CH3OH. Solid State Ionics 110, 199 (1998).
J. Kawakita, T. Miura, and T. Kishi: Lithium insertion and extraction kinetics of Li1+xV3O8. J. Power Sources 83, 79 (1999).
S. Jouanneau, A. Verbaere, S. Lascaud, and D. Guyomard: Improvement of the lithium insertion properties of Lij !V3O8. Solid State Ionics 117, 311 (2006)
Author information
Authors and Affiliations
Corresponding author
Additional information
This author was an editor of this journal during the review and decision stage. For the JMR policy on review and publication of manuscripts authored by editors, please refer to http://www.mrs.org/jmr_policy
Rights and permissions
About this article
Cite this article
Zhao, P., Wang, D., Lu, J. et al. Synthesis of LiV3O8 nanorods and shape-dependent electrochemical performance. Journal of Materials Research 26, 424–429 (2011). https://doi.org/10.1557/jmr.2010.45
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1557/jmr.2010.45