Abstract
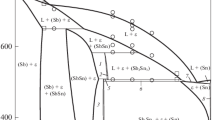
We report on Molecular Dynamics (MD) studies of free energy and vibrational entropy differences of the binary system Ag2Se-Ag2S as function of temperature. We have employed the methods of adiabatic switching and temperature integration over a reversible path to obtain the phase diagram. We discuss the nature of the entropy difference in terms of ion mobility and previous MD calculations of diffusion rates in these systems.
Similar content being viewed by others
References
R. Sadanaga and S. Sueno, Mineral. J. 5 , 124 (1967)
G. A. Wiegers, Am. Min. 56, 1882 (1971)
J. P. Rino, Y. M. M. Homos, G. A. Antonio, I. Ebbsjo, R. K. Kalia and P. Vashishta, J. Chem. Phys. 89, 7542 (1988)
J. R. Ray and P. Vashishta, J. Chem. Phys. 90, 6580 (1989)
N. E. Pingitore, B. F. Ponce and L. Estrada, J. Mater. Res. 8, 1 (1993)
P. Vashishta, I. Ebbsjö, R. Dejus and K. Sköld, J. Phys. C 18, L291 (1985)
J. G. Kirkwood, J. Chem. Phys. 3, 300 (1935)
M. Watanabe and W. P. Reinhardt, Phys. Rev. Letters, 65, 3301 (1990)
B.L. Holian, H.A. Posch and W.G. Hoover, Phys. Rev. E, 47, 3852 (1993)
K. C. Mills, Thermodynamic data for inorganic Sulfides, Selenides, and Tellurides, (Butterworth & co (Publishers) LTD, 1974), p. 734.
A. E. Sagua and J. A. Schmidt, Materials Chem. Phys. 32, 235 (1992)
F. Gr0nvold and E. F. Westrum, J. Chem. Thermodynamics 18, 381 (1986)
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Smith, A., Ravelo, R. & Pingitore, N. Thermodynamics of the System Ag2se-Ag2s: A Molecular Dynamics Study. MRS Online Proceedings Library 398, 531–536 (1995). https://doi.org/10.1557/PROC-398-531
Published:
Issue Date:
DOI: https://doi.org/10.1557/PROC-398-531