Abstract
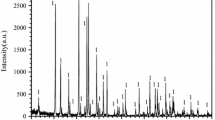
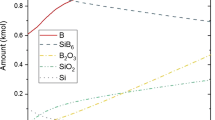
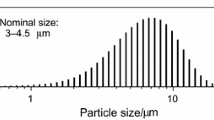
The oxidation of two α–Si3N4 powders has been studied using x-ray photoelectron spectroscopy (XPS) and Bremsstrahlung-excited Auger electron spectroscopy (AES). These two powders had comparable specific surface areas but were produced by different methods. The powders were oxidized by heating in air at temperatures between 850° and 1000 °C. The oxide thickness for each heating time and temperature was determined both from the relative O 1s and N 1s peak intensities and from the relative SiO2 and Si3N4 Si KLL peak intensities. In each case, the oxide thickness calculated from the XPS data was systematically higher than those determined from the AES data. This difference may be due to uncertainties in the data analysis procedures. The oxidation of each powder followed a linear rate law for these temperatures. On one of the powders, the XPS data showed evidence of oxynitride formation. Analysis with thermal decomposition mass spectroscopy also showed that much more hydrogen was evolved from this particular powder, particularly at temperatures >700 °C. These differences in bulk impurity levels are believed to be responsible for the differences in the activation energies for oxidation which were measured to be 104 ± 22 kJ/mol and 62 ± 4 kJ/mol for these two powders.
Similar content being viewed by others
References
G. Wotting and G. Ziegler, Interceram. 2, 32–35 (1986) and 3, 57–60 (1986).
F. F. Lange, J. Am. Ceram. Soc. 67 (2), 83 (1984).
G. Schwier, Progress in Nitrogen Ceramics, edited by F. L. Riley (Martinus Nijhoff Publishers, Boston, MA, 1983), pp. 157–166.
Anon., Advanced Ceramics Materials Technical Report, Ube Industries, 3.6000 (1987).
P.S. Wang, S.M. Hsu, and T.N. Wittberg, J. Mater. Sci. 26, 1655 (1991).
P. S. Wang, S. G. Malghan, S. M. Hsu, and T. N. Wittberg, Surf. Interf. Anal. 18, 159 (1992).
P.S. Wang, S.M. Hsu, S.G. Malghan, and T.N. Wittberg, J. Mater. Sci. 26, 3249 (1991).
J. M. Horton, J. Am. Ceram. Soc. 52, 121 (1969).
W.C. Tripp and H.C. Graham, J. Am. Ceram. Soc. 59, 399 (1976).
Certain commercial equipment, instruments, or materials are identified in this paper in order to specify adequately the experimental procedure. Such identification does not imply recommendation or endorsement by the National Institute of Standards and Technology, nor does it imply that the materials or equipment identified are necessarily the best available for the purpose.
W. H. Press, B. P. Flannery, S. A. Teukolsky, and W. T. Vetterling, Numerical Recipes (Cambridge University Press, New York, 1987), pp. 400, 407–420, 523–528.
C.D. Wagner, in Practical Surface Analysis, edited by D. Briggs and M.P. Seah (John Wiley, New York, 1983), Appendix 4.
T.A. Carlson and G.E. McGuire, J. Electron Spectrosc. 1, 161 (1972).
S. Tanuma, C. J. Powell, and D. R. Penn, Surf. Interf. Anal. 17, 927 (1991).
C.J. Powell, J. Vac. Sci. Technol. A 3, 1338 (1985).
S.R. Scholes, Modern Glass Practice, 7th ed. (CSI Publishing Co., Boston, MA, 1975), p. 387.
Handbook of Chemistry and Physics, edited by R. C. Weast, 60th ed. (Chemical Rubber, Boca Raton, FL, 1988), p. B-121.
J. H. Scofield, J. Electron. Spectrosc. 8, 129 (1976).
A.D. Katnani and K.I. Papathomas, J. Vac. Sci. Technol. A 5, 1335 (1987).
H. J. Stein and H. A. R. Wegener, J. Electrochem. Soc. 124, 908 (1977).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Wang, P.S., Malghan, S.G., Hsu, S.M. et al. Effects of α-silicon nitride powder processing on surface oxidation kinetics. Journal of Materials Research 8, 3168–3175 (1993). https://doi.org/10.1557/JMR.1993.3168
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1557/JMR.1993.3168