Abstract
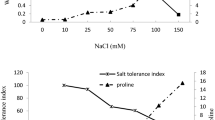
Seeds of snail medic (Medicago scutellata L.) were assessed for their response to salt at the germination and seedling stages. NaCl at concentrations 86 and 170 mM decreased the final germination percentage. Embryonic axis length, water content and dry weight of embryonic axis and cotyledons were also reduced by salt treatment. Furthermore, 28-d-old plants were grown hydroponically with different NaCl concentrations (0, 86 and 170 mM). After 7 days of treatment, growth, water content and development of the different organs of M. scutellata plant were affected especially at the highest NaCl concentration (170 mM). However, NaCl did not affect root length and the number of stem shoots but reduced stem length and total leaf area. Salt treatment increased markedly the concentration of Na+ in leaf and root tissues while reduced that of K+ only in root and stem tissues. Lipid peroxidation revealed the damage of the membranes of roots and leaves. Moreover, showed a more intense suberization and lignification at the cambial zone of roots of M. scutellata, were observed under the effect of NaCl.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
Ashraf, M., Orooj, A. (2006) Salt stress effects on growth, ion accumulation and seed oil concentration in an arid zone traditional medicinal plant ajwain (Trachyspermum ammi [L.] Sprague). J. Arid. Environ. 64, 209–220.
Asish, K. P., Anath, B. D. (2004) Salt tolerance and salinity effects on plants. Ecotox. Environ. Safe. 60, 324–349.
Attia, H., Karray, N., Rabhi, M., Lachaâl, M. (2008) Salt-imposed restrictions on the uptake of macroelements by roots of Arabidopsis thaliana. Acta Physiol. Plant. 30, 723–727.
Attia, H., Nouaili, S., Soltani, A., Lachaâl, M. (2009) Comparison of the responses to NaCl stress of two pea cultivars using split-root system. Sci. Hort. 123, 164–169.
Attia, H., Ouhibi, C., Ellili, A., Msilini, N., Bouzaïen, G., Karray, N., Lachaâl, M. (2011) Analysis of salinity effects on basil leaf surface area, photosynthetic activity, and growth. Acta Physiol. Plant. 33, 823–833.
Ben Amor, N., Hamed, K. B., Debez, A., Grignon, C., Abdelly, C. (2005) Physiological and antioxidant responses of the perennial halophyte Crithmum maritimum to salinity. Plant Sci. 168, 889–899.
Chinnusamy, V., Jagendorf, A., Zhu, J. K. (2005) Understanding and improving salt tolerance in plants. Crop Sci. 45, 437–448.
da Silva, E. C., Nogueira, R. J. M. C., Araujo, F. P., Melo, N. F., de Azevedo Neto, A. D. (2008) Physiological responses to salt stress in young umbu plants. Environ. Exp. Bot. 63, 147–157.
Dionisio-Sese, M. L., Tobita, S. (1998) Antioxidant responses of rice seedlings to salinity stress. Plant Sci. 135, 1–9.
Duzan, H. M., Zhou, X., Souleimanov, A., Smith, D. L. (2004) Perception of Bradyrhizobium japonicum Nod factor by soybean [Glycine max (L.) Merr.] root hairs under abiotic stress conditions. J. Exp. Bot. 55, 2641–2646.
El-Keblawy, A. (2004) Salinity effects on seed germination of the common desert range grass, Panicum turgidum. Seed Sci. Technol. 32, 943–948.
Fakhari, F., Sadeghi, H. (2016) Investigating the effects of pod elimination on salinity tolerance in annual Medic (Medicago scutellata L.). J. Rangeland Sci. 6, 232–241.
Garthwaite A. J., Millhollon, E. P., Lucas, M. C. (2005) Salt tolerance in wild Hordeum species is associated with restricted entry of Na+ and Cl− into the shoots. J. Exp. Bot. 56, 2365–2378.
Graham, P. H., Vance, C. P. (2003) Legumes: importance and constraints to greater use. Plant Physiol. 131, 872–877.
Heath, R. L., Packer, L. (1968) Photooxidation in isolated chloroplasts I: kinetics and stoichiometry of fatty acid peroxidation. Arch. Biochem. Biophys. 125, 189–198.
Hoagland, D. R., Arnon, D. I. (1950) The water culture method for growing plants without soil. Circ 347. Cal. Agri. Exp. Stat. Berkley.
Hu, Y., Fricke, W., Schmidhalter, U. (2005) Salinity and the growth of non-halophytic grass leaves: the role of mineral nutrient distribution. Funct. Plant Biol. 32, 973–985.
Huang, W. L., Liu, F. L. (2002) Carbohydrate metabolism in rice during callus induction and shoot regeneration induced by osmotic stress. Bot. Bull. Acad. Sinica 43, 107–113.
Jbir, N., Chaïbi, W., Ammar, S., Jemmali, A., Ayadi, A. (2001) Root growth and lignification of two wheat species differing in their sensitivity to NaCl, in response to salt stress. C.R. Acad. Sci. Paris, Sciences de la vie / Life Sciences 324, 863–868.
Lazrek, F., Roussel, V., Ronfort, J., Cardinet, G., Chardon, F., Aouani, M. E., Huguet, T. (2009) The use of neutral and non-neutral SSRs to analyse the genetic structure of a Tunisian collection of Medicago truncatula lines and to reveal associations with eco-environmental variables. Genetica 135, 391–402.
Lechno, S., Zamski, E., Telor, E. (1997) Salt stress-induced responses in cucumber plants. J. Plant Physiol. 150, 206–211.
McKinney, G. (1941) Absorption of light by chlorophyll solutions. J. Biol. Chem. 140, 315–332.
Mezni, M., Albouchi, A., Bizid, E., Hamza, M. (2002) Effet de la salinité des eaux d’irrigation sur la nutrition minérale chez trois variétés de Luzerne pérenne (Medicago sativa). Agronomie 22, 283–291.
Mittler, R. (2002) Oxidative stress, antioxidants and stress tolerance. Trends Plant Sci. 7, 406–410.
Mtimet, A. (2001) Soils of Tunisia. In: Zdruli, P., Steduto, P., Lacirignola, C., Montanarella, L. (eds) Soil resources of southern and eastern mediterranean countries. Bari, Italy, pp. 243–262.
Nasri, N., Kaddour, R., Rabhi, M., Plassard, C., Lachaâl, M. (2011) Effect of salinity on germination, phytase activity and phytate content in lettuce seedling. Acta Physiol. Plant. 33, 935–942.
Parida, A. K., Das, A. B. (2005) Salt tolerance and salinity effects on plants. Ecotox. Environ. Safe. 60, 324–349.
Piano, E., Francis, C. M. (1992) The annual species of Medicago in the Mediterranean region: ecogeography and related aspects of plant introduction and breeding. In: Proceedings of the 10th International Conference of Eucarpia Medicago spp. (Group Lodi), Italy, pp. 373–385.
Reinhards, D. H., Rost, T. L. (1995) Salinity accelerates endodermal development and induces an exodermis in cotton seedling roots. Environ. Exp. Bot. 35, 563–574.
Reinoso, H., Sosa, L., Ramírez, L., Luna, V. (2004) Salt-induced changes in the vegetative anatomy of Prosopis strombulifera (Leguminosae). Rev. Can. Bot. 82, 618–628.
Sairam, R. K., Srivastava, G. C. (2002) Changes in antioxidant activity in sub-cellular fractions of tolerant and susceptible wheat genotypes in response to long term salt stress. Plant Sci. 162, 897–904.
Shalata, A., Neumann, P. M. (2001) Exogenous ascorbic acid (vitamin C) increases resistance to salt stress and reduces lipid peroxidation. J. Exp. Bot. 52, 2207–2211.
Walker, R. R., Sedgley, M., Blesing, M. A., Douglas, T. J. (1984) Anatomy, ultrastructure and assimilate concentrations of roots of citrus genotypes differing in ability for salt exclusion. J. Exp. Bot. 35, 1481–1494.
Wenxue, W., Bilsborrow, P. E., Hooley, P., Fincham, D. A., Lombi, E., Forster, B. P. (2003) Salinity induced differences in growth, ion distribution and partitioning in barley between the cultivar Maythorpe and its derived mutant Golden Promise. Plant Soil 250, 183–191.
Zhong, H., Läuchli, A. (1993) Changes of cell wall composition and polymer size in primary roots of cotton seedlings under salt stress. J. Exp. Bot. 44, 773–778.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Attia, H., Alamer, K.H., Selmi, I. et al. Physiological and Structural Modifications in Snail Medic (Medicago scutellata L.) Plants Exposed to Salinity. BIOLOGIA FUTURA 69, 336–349 (2018). https://doi.org/10.1556/018.68.2018.3.9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1556/018.68.2018.3.9