Abstract
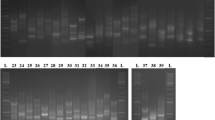
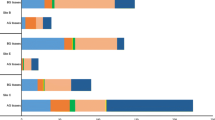
Endophytic phosphorus- and potassium-solubilizing bacteria were screened from the root, rhizome, stem, and leaves of Moso Bamboo, and their diversity was analyzed using their 16S rDNA sequences. Twenty endophytic phosphorus and potassium-solubilizing bacteria were screened from 82 bamboo plants, among which the CT-B09-2, WYS-A01-1 and JL-B06 had higher activities in decomposing organophos-phates. The three species showed a decomposition diameter/colony diameter (D/d) of 5.05, 4.19 and 2.95, respectively, and a solubilizing activity of 81.77 mg/L, 77.85 mg/L and 63.69 mg/L, respectively. JL-B06, WYS-A01-1 and CT-B09-2 had higher activities in decomposing inorganic phosphorus, with a decomposition diameter/colony diameter (D/d) of 2.34, 2.12 and 1.82, respectively, and a solubilizing activity of 30.58 mg/L, 38.89 mg/L and 48.35 mg/L, respectively. CT-B21, WYS-A03-1 and JL-B06 had higher activities in decomposing potassium, with a decomposition diameter/colony diameter (D/d) of 3.37, 4.84 and 4.33, respectively, and a solubilizing activity of 2.81 mg/L, 2.54 mg/L and 2.46 mg/L, respectively. The 16S rDNA sequence analysis showed that the 20 phosphorus- and potassium-solubilizing bacteria belong to 14 species from 10 genera, and mainly consist of Alcaligenes spp., Enterobacter spp. and Bacillus spp. Our results demonstrate the abundant diversity of endophytic phosphorus- and potassium-solubilizing bacteria in Moso Bamboo.
Article PDF
Similar content being viewed by others
References
Basak, B. B., Biswas, D. R. (2009) Influence of potassium solubilizing microorganism (Bacillus mucilaginosus) and waste mica on potassium uptake dynamics by Sudan grass (Sorghum vulgare Pers.) grown under two Alfisols. Plant Soil. 317, 235–255.
Berg, G., Krechel, A., Ditz, M., Sikora, R. A., Ulrich, A., Hallmann, J. (2005) Endophytic and exophytic potato-associated bacterial communities differ in structure and antagonistic function against plant pathogenic fungi. FEMSMicrobiol. Ecol. 51, 215–229.
Gao, Z. Q., Fu, M. Y. (2006) Characteristics of seasonal changes in soil carbon and nitrogen nutrients of different Phyllostachys pubescens stands. J. Zhejiang Coll. Forest. 23, 248–254.
Gaulner, J. M., Feldman, A. W., Zablotowiez, R. M. (1982) Identity and behavior of xylem-residing bacteria rough lemon of florida citrus trees. Appl. Environ. Microb. 43, 1335–1342.
Goenadi, D. H., Siswanto, S. Y. (2000) Bioactivation of poorly soluble phosphate rocks with a phosphorus-solubilizing fungus. Soil Sci. Soc. Am. J. 64, 927–932.
Gothwal, R. K., Nigam, V. K., Mohanl, M. K., Samal, D., Ghosh, P. (2006) Phosphate solubilization by rhizospheric bacterial isolates from economically important desert plants. Indian J. Microb. 46, 355–361.
Han, H. S., Lee, K. D. (2005) Phosphate and potassium solubilizing bacteria effect on mineral uptake, soil availability and growth of eggplant. Res. J. Agri. Bio. Sci. 1, 176–180.
Han, S., Xia, D. L., Li, L. B., Han, J. G. (2010) Diversity of the phosphate solubilizing bacteria isolated fromthe root of Phyllostachys pubescens. J. Agricul. Univer Hebei. 33, 26–31.
Huang, Q. T., Chen, A. L., He, J. (2006) Comparison of soil physical and chemical properties among various Phyllostachys pubescens plantation. J. Fujian Coll. Forest. 26, 299–302.
Illmer, P., Schinner, F. (1992) Solubilization of inorganic phosphates by microorganism isolated from forest siol. Soil. Biol. Biochem. 24, 389–395.
Ivanova, R., Bojinova, D., Nedialkova, K. (2006) Rock phosphate solubilization by soil bacteria. J. Univ. Chem. Tech. Metall. 41, 297–302.
Jha, B. K., Gandhi Pragash, M., Cletus, J., Raman, G., Sakthivel, N. (2009) Simultaneous phosphate solubilization potential and antifungal activity of new fluorescent pseudomonad strains, Pseudomonas aeruginosa, P. plecoglossicida and P. mosselii. World J. Microbiol. Biotechnol. 25, 573–581.
Kucey, R. M. N. (1988) Effect of penicillium bilaji on the solubility and uptake of P and micronutri-ents from soil by wheat. Can. J. siol Sci. 68, 261–270.
Loveleen, R., Pankaj, K., Sudhakara, M. R. (2008) Effect of carbon and nitrogen sources on phosphate solubilization by a wild-type strain and UV-induced mutants of Aspergillus tubingensis. Curr. Microbiol. 57, 401–406.
Lu, L. K. (1999) Soil Agricultural Chemical Analysis Method. Chinese agricultural Science and technology Press, Beijing.
Mano, H., Tanaka, F., Watanabe, A., Kaga, H., Okunishi, S., Morisaki, H. (2006) Culturable surface and endophytic bacterial flora of the maturing seeds of rice plants (Oryza sativa) cultivated in a paddy field. Microb. Environ. 21, 86–100.
Mathurot, C., Saisamorn, L. (2009) Phosphate solubilization potential and stress tolerance of rhizbac-teria from rice soil in Northern Thailand. World J. Microbiol. Biotechnol. 25, 305–314.
Naik, P. R., Raman, G., Narayanan, K. B., Sakthivel, N. (2008) Assessment of genetic and functional diversity of phosphate solubilizing fluorescent pseudomonads isolated from rhizospheric soil. BMC Microbiol. 8, 230–243.
Nautiyal, C. S., Bhadauria, S. S., Kumar, P., Lai, H., Mondal, R., Verma, D. (2000) Stress induced phosphate solubilization in bacteria isolated from alkaline soils. FEMSMicrobiol. Lett. 182, 291–296.
Oliveira, C. A., Alves, V. M. C., Marriel, I. E., Gomes, E. A., Scotti, M. R., Carneiro, N. P., Guimaraes, C. T., Schaffert, R. E., Sa, N. M. H. (2009) Phosphate solubilizing microorganisms isolated from rhizosphere of maize cultivated in an oxisol of the Brazilian Cerrado Biome. Soil Biol. Biochem. 41, 1782–1787.
Pandey, A., Trivedi, P., Kumar, B., Palni, L. M. (2006) Characterization of a phosphate solubilizing and antagonistic strain of Pseudomonas putida (B0) isolated from a sub-alpine location in the Indian Central Himalaya. Curr. Microbiol. 53, 102–107.
Rashid, M., Khalil, S., Ayub, N., Alam, S., Latif F. (2004) Organic acids production and phosphate solubilization by phosphate solubilizing microorganisms (PSM) under in vitro conditions. Pak. J. Biol. Sci. 7, 187–196.
Ryan, R. P., Germaine, K., Franks, A., Ryan, D. J., Dowling, D. N. (2008) Bacterial endophytes:recent developments and application. FEMS Microbiol. Lett. 278, 1–9.
Selvarajm, P., Munusamy, M., Tongmin, S. (2008) Isolation and identification of phosphate solubilizing bacteria from Chinese cabbage and their effect on growth and phosphorus utilization of plants. J. Microbiol. Biotechnol. 18, 113–411.
Souchie, E. L., Abboud, A. C. D. (2007) Phosphate solubilization by microorganisms from the rhizosphere of Pigeonpea genotypes grown in different soil classes. Semina-Ciencias Agrarias 28, 11–18.
Sridevi, M., Mallaiah, K. V. (2009) Phosphate solubilization by rhizobium strains. Indian J. Microbiol. 149, 98–102.
Wang, G. H., Zhou, D. R., Yang, Q., Jin, J., Liu, X. B. (2005) Solubilization of rock phosphate in liquid culture by fungal isolates from rhizosphere soil. Pedosphere 15, 532–538.
Xu, Y. J. (2011) Research progress on pesources diversity of plant endophytes. Guangdong Agricul. Sci. 4, 149–152.
Zhang, L. Z., Fan, J. J., Niu, W., Li, T., Wu, R. H., Jin, Y. J. (2011) Isolation of phosphate solubilizing fungus (Aspergillus niger) from Caragana rhizosphere and its potential for phosphate solubilization. Acta. Ecol. Sin. 31, 7571–7578.
Zhao, X. R., Lin, Q. M., Sun, Y. X., Yao, J., Zhang, Y. S. (2001) The methods for quantifying capacity of bacteria in dissolving P compounds. Microbiology 28, 1–4.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Yuan, ZS., Liu, F. & Zhang, GF. Characteristics and Biodiversity of Endophytic Phosphorus- and Potassium-Solubilizing Bacteria in Moso Bamboo (Phyllostachys Edulis). BIOLOGIA FUTURA 66, 449–459 (2015). https://doi.org/10.1556/018.66.2015.4.9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1556/018.66.2015.4.9