Abstract
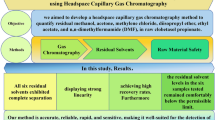
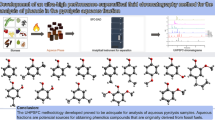
This paper reports a headspace analysis technique for the determination of products, i.e., cyclohexanone (CE) and cyclohexanol (CL), of phenol hydrogenation in a supercritical water reaction system (SWRS) with water removal by hydrate formation. An addition of anhydrous calcium chloride leads to water absorption resulting in crystal water; thus, the samples can be quantitatively measured without the influence of water. After achieving equilibrium at 150°C and maintaining it for 5 min, the obtained results showed a relative standard deviation of less than 5.3% and the recovery ranged from 93% to 104%. The presented method is simple and accurate for the analysis of CL, CE and phenol in samples from phenol conversion in SWRS.
Similar content being viewed by others
References
Behrendt, F., Neubauer, Y., Oevermann, M., Wilmes, B., & Zobel, N. (2008). Direct liquefaction of biomass. Chemical Engineering & Technology, 31, 667–677. DOI: 10.1002/ceat.200800077.
Buszewski, B., Olszowy, P., Szultka, M., & Jezewska, A. (2012). New approaches to extraction techniques in determination of 4,4′-methylenebis(2-chloroaniline) in air and water solutions. Talanta, 93, 117–121. DOI: 10.1016/j.talanta.2012.01.057.
Chai, X. S., Hou, Q. X., & Schork, F. J. (2004). Determination of residual monomer in polymer latex by full evaporation headspace gas chromatography. Journal of Chromatography A, 1040, 163–167. DOI: 10.1016/j.chroma.2004.04.024.
Guan, Q. G., Wei, C. H., & Savage, P. E. (2012). Hydrothermal gasification of Nannochloropsis sp. with Ru/C. Energy &Fuels, 26, 4575–4582. DOI: 10.1021/ef3007992.
Hu, H. C., & Chai, X. S. (2012). Determination of methanol in pulp washing filtrates by desiccated full evaporation headspace gas chromatography. Journal of Chromatography A, 1222, 1–4. DOI: 10.1016/j.chroma.2011.11.045.
Hu, H. C., Chai, X. S., Wei, C. H., & Barnes, D. (2014). Increasing the sensitivity of headspace analysis of low volatility solutes through water removal by hydrate formation. Journal of Chromatography A, 1343, 42–46. DOI: 10.1016/j.chroma.2014.03.066.
Huelsman, C. M., & Savage, P. E. (2012). Intermediates and kinetics for phenol gasification in supercritical water. Physical Chemistry Chemical Physics, 14, 2900–2910. DOI: 10.1039/c2cp23910h.
Huelsman, C. M., & Savage, P. E. (2013). Reaction pathways and kinetic modeling for phenol gasification in supercritical water. The Journal of Supercritical Fluids, 81, 200–209. DOI: 10.1016/j.supflu.2013.05.012.
Kang, S. M., Li, X. L., Fan, J., & Chang, J. (2013). Hydrothermal conversion of lignin: A review. Renewable and Sustainable Energy Reviews, 27, 546–558. DOI: 10.1016/j.rser.2013.07.013.
Li, H. L., Zhan, H. Y., Fu, S. Y., Liu, M. R., & Chai, X. S. (2007). Rapid determination of methanol in black liquors by full evaporation headspace gas chromatography. Journal of Chromatography A, 1175, 133–136. DOI: 10.1016/j.chroma.2007.10.040.
Li, H. L., Chai, X. S., Zhan, H. Y., & Fu, S. Y. (2010). Rapid determination of furfural in biomass hydrolysate by full evaporation headspace gas chromatography. Journal of Chromatography A, 1217, 7616–7619. DOI: 10.1016/j.chroma.2010.09.073.
Pandey, M. P., & Kim, C. S. (2011). Lignin depolymerization and conversion: A review of thermochemical methods. Chemical Engineering & Technology, 34, 29–41. DOI: 10.1002/ceat.201000270.
Park, C., & Keane, M. A. (2003). Catalyst support effects: gas-phase hydrogenation of phenol over palladium. Journal of Colloid and Interface Science, 266, 183–194. DOI: 10.1016/s0021-9797(03)00171-1.
Pérez, Y., Fajardo, M., & Corma, A. (2011). Highly selective palladium supported catalyst for hydrogenation of phenol in aqueous phase. Catalysis Communications, 12, 1071–1074. DOI: 10.1016/j.catcom.2011.03.026.
Reddy, S. N., Nanda, S., Dalai, A. K., & Kozinski, J. A. (2014). Supercritical water gasification of biomass for hydrogen production. International Journal of Hydrogen Energy, 39, 6912–6926. DOI: 10.1016/j.ijhydene.2014.02.125.
Shore, S. G., Ding, E., Park, C., & Keane, M. A. (2002). Vapor phase hydrogenation of phenol over supported Pd and Pd—Yb catalysts. Catalysis Communications, 3, 77–84. DOI: 10.1016/s1566-7367(02)00052-3.
Tabrizi, A. B. (2007). Development of a cloud point extractionspectrofluorimetric method for trace copper(II) determination in water samples and parenteral solutions. Journal of Hazardous Mater, 139, 260–264. DOI: 10.1016/j.jhazmat.2006.06.024.
Yoon, Y., Rousseau, R., Weber, R. S., Mei, D. H., & Lercher, J. A. (2014). First-principles study of phenol hydrogenation on Pt and Ni catalysts in aqueous phase. Journal of the American Chemical Society, 136, 10287–10298. DOI: 10.1021/ja501592y.
Zhang, Q. L., Qiu, C. T., Xu, H. D., Lin, T., Lin, Z., Gong, M. C., & Chen, Y. Q. (2011). Low-temperature selective catalytic reduction of NO with NH3 over monolith catalyst of MnO x /CeO2—ZrO2—Al2O3. Catalysis Today, 175, 171–176. DOI: 10.1016/j.cattod.2011.05.009.
Zhong, J. F., Chai, X. S., Qin, X. L., & Fu, S. Y. (2011). A full evaporation headspace gas chromatographic method for determination of monomer conversion in cellulose graft polymethyl methacrylate. Carbohydrate Polymers, 86, 367–370. DOI: 10.1016/j.carbpol.2011.04.050.
Zhou, N., Luo, H. D., Li, N., Jia, Y. Z., & Li, Y. Q. (2011). Highly sensitive and selective spectrofluorimetric approach for the rapid determination of trace benzo[α]pyrene in drinking water and in solutions leached from disposable paper cups. Luminescence, 26, 35–43. DOI: 10.1002/bio.1183.
Zhu, J. F., Tao, G. H., Liu, H. Y., He, L., Sun, Q. H., & Liu, H. C. (2014). Aqueous-phase selective hydrogenation of phenol to cyclohexanone over soluble Pd nanoparticles. Green Chemistry, 16, 2664–2669. DOI: 10.1039/c3gc42408a.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Guan, QQ., Huang, XD., Zeng, YH. et al. Rapid determination of products of phenol hydrogenation in a supercritical water system using headspace gas chromatography. Chem. Pap. 69, 662–667 (2015). https://doi.org/10.1515/chempap-2015-0081
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1515/chempap-2015-0081