Abstract
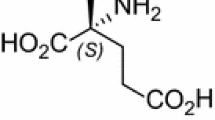
The practical synthetic preparation of glutaric acid has remained a major challenge to date. In the present study, glutaric acid was synthesised by way of one-pot diazoniation/hydrogenation of the readily available l-glutamic acid under aqueous conditions on a gram-scale with good yields. This is the first example of the deamination of the aliphatic primary amine via diazoniation and could afford a practical approach to the production of glutaric acid.
Similar content being viewed by others
References
Adams, R., & Kornblum, N. (1941). Stereochemistry of biphenyls. LI. Resolution of diphenic acids having many-membered bridges across the 5,5′-positions: A novel type of restricted rotation. Journal of the American Chemical Society, 63, 188–200. DOI: 10.1021/ja01846a044.
Antonelli, E., D’Aloisio, R., Gambaro, M., Fiorani, T., & Venturello, C. (1998). Efficient oxidative cleavage of olefins to carboxylic acids with hydrogen peroxide catalyzed by methyltrioctylammonium tetrakis(oxodiperoxotungsto)phosphate(3−) under two-phase conditions. Synthetic aspects and investigation of the reaction course. The Journal of Organic Chemistry, 63, 7190–7206. DOI: 10.1021/jo980481t.
Bacherikov, V. A., Wang, M. J., Cheng, S. Y., Chen, C. H., Chen, K. T., & Su, T. L. (2004). Ethyl acetate as a proreducing agent in an one-pot reductive deamination of nitroanilines. Bulletin of the Chemical Society of Japan, 77, 1027–1028. DOI: 10.1246/bcsj.77.1027.
Balaraman, E., Khaskin, E., Leitus, G., & Milstein, D. (2013). Catalytic transformation of alcohols to carboxylic acid salts and H2 using water as the oxygen atom source. Nature Chemistry, 5, 122–125. DOI: 10.1038/nchem.1536.
Baqi, Y., & Müller, C. E. (2012). Efficient and mild deamination procedure for 1-aminoanthraquinones yielding a diverse library of novel derivatives with potential biological activity. Tetrahedron Letters, 53, 6739–6742. DOI: 10.1016/j.tetlet.2012.09.011.
Besson, M., Gauthard, F., Horvath, B., & Gallezot, P. (2005). Catalytic oxidation with air of cyclohexanone to dicarboxylic acids on synthetic carbons. Effect of supported metals and solvents. The Journal of Physical Chemistry B, 109, 2461–2467. DOI: 10.1021/jp0459662.
Bigelow, L. A., Johnson, J. R., & Sandborn, L. T. (1926). m-Bromotoluene. Organic Syntheses, 6, 16–18. DOI: 10.15227/orgsyn.006.0016.
Bogert, M. T., & Mandelbaum, M. R. (1923). The action of sulfur upon para-toluidine in the presence of litharge. Thiopara-toluidine, its constitution and some new derivatives. Journal of the American Chemical Society, 45, 3045–3055. DOI: 10.1021/ja01665a034.
Brewster, R. Q., & Poje, J. A. (1939). Reduction of diazonium salts to hydrocarbons with alkaline formaldehyde. Journal of the American Chemical Society, 61, 2418–2419. DOI: 10.1021/ja01878a044.
Castellan, A., Bart, J. C. J., & Cavallaro, S. (1991). Industrial production and use of adipic acid. Catalysis Today, 9, 237–254. DOI: 10.1016/0920-5861(91)80049-f.
Che, C. M., Yip, W. P., & Yu, W. Y. (2006). Ruthenium-catalyzed oxidation of alkenes, alkynes, and alcohols to organic acids with aqueous hydrogen peroxide. Chemistry — An Asian Journal, 1, 453–458. DOI: 10.1002/asia.200600091.
Chen, H., Dai, W., Yang, X., Gao, R., Cao, Y., & Fan, K. (2006a). Green catalytic process for synthesis of glutaric acid by selective oxidation of cyclopentene. Shiyou Huagong/Petrochemical Technology, 35, 118–121. DOI: 10.3321/j.issn:1000-8144.2006.02.003. (in Chinese)
Chen, H., Dai, W. L., Yang, X. L., Gao, R., Cao, Y., Li, H., & Fan, K. (2006b). Studies on the structural change of a reaction-controlled phase-transfer [π-C5H5NC16H33]3PO4 [WO3]4 catalyst during the selective oxidation of cyclopentene to glutaric acid with aqueous H2O2. Applied Catalysis A: General, 309, 62–69. DOI: 10.1016/j.apcata.2006.04.037.
Chen, H., Dai, W. L., Gao, R., Cao, Y., Li, H., & Fan, K. (2007). New green catalytic manufacture of glutaric acid from the oxidation of cyclopentane-1,2-diol with aqueous hydrogen peroxide. Applied Catalysis A: General, 328, 226–236. DOI: 10.1016/j.apcata.2007.06.021.
Choudary, B. M., Valli, V. L. K., & Prasad, A. D. (1991). A novel montmorillonite-KMnO4 system for the oxidation of alkenes under triphase conditions. Synthetic Communications, 21, 2007–2013. DOI: 10.1080/00397919108019806.
Chu, X., Zhu, Q., Dai, W. L., & Fan, K. (2012). Excellent catalytic performance of graphite oxide in the selective oxidation of glutaraldehyde by aqueous hydrogen peroxide. RSC Advances, 2, 7135–7139. DOI: 10.1039/c2ra21068a.
Clarke, H. T., & Taylor, E. R. (1923). m-Nitrotoluene. Organic Syntheses, 3, 91–92. DOI: 10.15227/orgsyn.003.0091.
Coleman, G. H., & Talbot, W. F. (1933). sym-Tribromobenzene. Organic Syntheses, 13, 96–99. DOI: 10.15227/orgsyn.013.0096.
DeTar, D. F., & Kosuge, T. (1958). Mechanisms of diazonium salt reactions. VI. The reactions of diazonium salts with alcohols under acidic conditions; evidence for hydride transfer. Journal of the American Chemical Society, 80, 6072–6079. DOI: 10.1021/ja01555a044.
Doldouras, G. A., & Kollonitsch, J. (1978). A direct, selective, and general method for reductive deamination of primary amines. Journal of the American Chemical Society, 100, 341–342. DOI: 10.1021/ja00469a088.
English, J., Jr., & Dayan, J. E. (1957). Glutaric acid and glutarimide [II. From dihydropyran]. Organic Syntheses, 37, 48–52. DOI: 10.15227/orgsyn.037.0047.
Gao, R., Chen, H., Le, Y., Dai, W. L., & Fan, K. (2009). Highly active and selective Cs2.5H0.5PW12O40/SBA-15 composite material in the oxidation of cyclopentane-1,2-diol to glutaric acid by aqueous H2O2. Applied Catalysis A: General, 352, 61–65. DOI: 10.1016/j.apcata.2008.09.031.
Geoffroy, O. J., Morinelli, T. A., & Meier, G. P. (2001). Chemoselective one-pot reductive deamination of aryl amines. Tetrahedron Letters, 42, 5367–5369. DOI: 10.1016/s0040-4039(01)01027-9.
Golubev, N. S., Asfin, R. E., Smirnov, S.N., & Tolstoi, P. M. (2006). Study of hydrogen bonds of hypophosphorous acid by 1H, 2H, 31P, and 15N NMR spectroscopy under slow exchange conditions. Russian Journal of General Chemistry, 76, 915–924. DOI: 10.1134/s1070363206060119.
Griffith, W. P., Shoair, A. G., & Suriaatmaja, M. (2000). Ruthenium-catalysed cleavage of alkenes and alkynes to carboxylic acids. Synthetic Communications, 30, 3091–3095. DOI: 10.1080/00397910008086915.
Griffith, W. P., & Kwong, E. (2003). Alkene and alkyne oxidative cleavage catalyzed by RuO4 in environmentally acceptable solvents. Synthetic Communications, 33, 2945–2951. DOI: 10.1081/scc-120022466.
He, L., Qiu, G., Gao, Y., & Wu, J. (2014). Removal of amino groups from anilines through diazonium salt-based reactions. Organic & Biomolecular Chemistry, 12, 6965–6971. DOI: 10.1039/c4ob01286k.
Hendrickson, J. B. (1961). Reduction of diazonium borofluorides by sodium borohydrides. Journal of the American Chemical Society, 83, 1251–1251. DOI: 10.1021/ja01466a055.
Iwahama, T., Yoshino, Y., Keitoku, T., Sakaguchi, S., & Ishii, Y. (2000). Efficient oxidation of alcohols to carbonyl compounds with molecular oxygen catalyzed by N-hydroxyphthalimide combined with a Co species. The Journal of Organic Chemistry, 65, 6502–6507. DOI: 10.1021/jo000760s.
Johnson, R. W., Pollock, C. M., & Cantrell, R. R. (1993). Dicarboxylic acids. In J. I. Kroschwitz (Ed.), Kirk-Othmer encyclopedia of chemical technology (4th ed., Vol. 8, pp. 614–628). New York, NY, USA: Wiley.
Kolitz, M, Cohen-Arazi, N, Hagag, I, Katzhendler, J, & Domb, A. J. (2009). Biodegradable polyesters derived from amino acids. Macromolecules, 42, 4520–4530. DOI: 10.1021/ma900464g.
KÖlker, S., Ahlemeyer, B., Krieglstein, J., & Hoffmann, G. F. (1999). 3-Hydroglutaric and glutaric acids are neurotoxic through NMDA receptors in vitro. Journal of Inherited Metabolic Disease, 22, 259–262. DOI: 10.1023/a:1005577920954.
Kornblum, N. (1941). 3,3′-Dimethoxybiphenyl and 3,3′-dimethylbiphenyl. Organic Syntheses, 21, 30–35. DOI: 10.15227/orgsyn.021.0030.
Kornblum, N. (1944). Replacement of the aromatic primary amino group by hydrogen. In R. Adams (Ed.), Organic reactions (Vol. 2, pp. 262–340). New York, NY, USA: Wiley.
Kornblum, N., & Iffland, D. C. (1949). The selective replacement of the aromatic primary amino group by hydrogen in aromatic-aliphatic diamines. Journal of the American Chemical Society, 71, 2137–2143. DOI: 10.1021/ja01174a064.
Kornblum, N., Cooper, G. D., & Taylor, J. E. (1950). The chemistry of diazo compounds. II. Evidence for a free radical chain mechanism in the reduction of diazonium salts by hypophosphorous acid. Journal of the American Chemical Society, 72, 3013–3021. DOI: 10.1021/ja01163a060.
Kornblum, N., Kelley, A. E., & Cooper, G. D. (1952). The chemistry of diazo compounds. III. The reduction of diazonium salts by phosphorous acid. Journal of the American Chemical Society, 74, 3074–3076. DOI: 10.1021/ja01132a036.
Lyalin, B. V., & Petrosyan, V. A. (2009). Electrosynthesis of glutaric acid and regularities of electrocatalytic oxidation of cycloalkanones at a NiOOH anode in aqueous NaOH. Russian Chemical Bulletin, International Edition, 58, 2426–2431. DOI: 10.1007/s11172-009-0339-1.
Marques, F. O., Hagen, M. E. K., Pederzolli, C. D., Sgaravatti, A. M., Durigon, K., Testa, C. G., Wannmacher, C. M. D., Wyse, A. T. S., Wajner, M., & Dutra-Filho, C. S. (2003). Glutaric acid induces oxidative stress in brain of young rats. Brain Research, 964, 153–158. DOI: 10.1016/s0006-8993(02)04118-5.
Marvel, C. S., & Tuley, W. F. (1925). Glutaric acid. Organic Syntheses, 5, 69–72. DOI: 10.15227/orgsyn.005.0069.
Meng, X. Y., & Cai, C. (2005). Deamination in ethyl acetate media. Huaxue Shiji/Chemical Reagents, 27, 625–626. DOI: 10.3969/j.issn.0258-3283.2005.10.018. (in Chinese)
Mitsuhashi, H., Kawakami, T., & Suzuki, H. (2000). A mild one-pot deamination of aromatic amines bearing electron-withdrawing groups. Calcium hypophosphite as a dediazonation reagent in nonaqueous media. Tetrahedron Letters, 41, 5567–5569. DOI: 10.1016/s0040-4039(00)00883-2.
Mo, F., Dong, G., Zhang, Y., & Wang, J. (2013). Recent applications of arene diazonium salts in organic synthesis. Organic & Biomolecular Chemistry, 11, 1582–1593. DOI: 10.1039/c3ob27366k.
Muühlhausen, C., Burckhardt, B. C., Hagos, Y., Burckhardt, G., Keyser, B., Lukacs, Z., Ullrich, K., & Braulke, T. (2008). Membrane translocation of glutaric acid and its derivatives. Journal of Inherited Metabolic Disease, 31, 188–193. DOI: 10.1007/s10545-008-0825-x.
Nickon, A., & Hill, A. S. (1964). A direct method for reductive deamination of aliphatic amines. Journal of the American Chemical Society, 86, 1152–1158. DOI: 10.1021/ja01060a040.
Orita, H., Hayakawa, T., & Takehira, K. (1986). Oxidation of cyclopentene by RuCl3—NaOCl catalyst. Bulletin of the Chemical Society of Japan, 59, 2637–2638. DOI: 10.1246/bcsj.59.2637.
Paris, G., Berlinguet, L., & Gaudry, R. (1957). Glutaric acid and glutarimide [1. Fromγ-butyrolactone]. Organic Syntheses, 37, 47–48. DOI: 10.15227/orgsyn.037.0047.
Pazo-Llorente, R., Maskill, H., Bravo-Diaz, C., & Gonzalez-Romero, E. (2006). Dediazaoniation of 4-nitrobenzenediazonium ions in acidic MeOH/H2O mixtures: Role of acidity and MeOH concentration on the formation of transient diazo ethers that initiate hemolytic dediazoniation. European Journal of Organic Chemistry, 2006, 2201–2209. DOI: 10.1002/ejoc.200500946.
Porciúncula, L. O., Dal-Pizzol, A., Jr., Coitinho, A. S., Emanuelli, T., Souza, D. O., & Wajner, M. (2000). Inhibition of synaptosomal [3H]glutamate uptake and [3H]glutamate binding to plasma membranes from brain of young rats by glutaric acid in vitro. Journal of the Neurological Sciences, 173, 93–96. DOI: 10.1016/s0022-510X(99)00307-x.
Rieker, A., Niederer, P., & Leibfritz, D. (1969). Chemisch induzierte dynamische kernpolarisation im system phenyldiazoniumtetrafluoroborat/NaBH4 nachweis des arylradikals bei reaktionen vom typ der gomberg-arylierung. Tetrahedron Letters, 10, 4287–4290. DOI: 10.1016/s0040-4039(01)886767. (in German)
Robison, M. M., & Robison, B. L. (1956). 2,4,6-Tribromobenzoic acid. Organic Syntheses, 36, 94–97. DOI: 10.15227/orgsyn.036.0094.
Roe, A., & Graham, J. R. (1952). Replacement of the primary aromatic amino group by hydrogen using diazonium fluoborates. Journal of the American Chemical Society, 74, 6297–6298. DOI: 10.1021/ja01144a523.
Romanova, N. V., & Demidenko, N. V. (1975). Hypophosphorous acid and its salts. Russian Chemical Reviews, 44, 1036–1047. DOI: 10.1070/rc1975v044n12abeh002547.
Rosa, R. B., Dalcin, K. B., Schmidt, A. L., Gerhardt, D., Ribeiro, C. A. J., Ferreira, G. C., Schuck, P. F., Wyse, A. T. S., Porciúncula, L. O., Wofchuk, S., Salbego, C. G., Souza, D. O., & Wajner, M. (2007). Evidence that glutaric acid reduces glutamate uptake by cerebral cortex of infant rats. Life Sciences, 81, 1668–1676. DOI: 10.1016/j.lfs.2007.09.021.
Saedi, Z., Tangestaninejad, S., Moghadam, M., Mirkhani, V., & Mohammadpoor-Baltork, I. (2012a). MIL-101 metal-organic framework: A highly efficient heterogeneous catalyst for oxidative cleavage of alkenes with H2O2. Catalysis Communications, 17, 18–22. DOI: 10.1016/j.catcom.2011.10.005.
Saedi, Z., Tangestaninejad, S., Moghadam, M., Mirkhani, V., & Mohammadpoor-Baltork, I. (2012b). The effect fo encapsulated Zn-POM on the catalytic activity of MIL-101 in the oxidation of alkenes with hydrogen peroxide. Journal of Coordination Chemistry, 65, 463–473. DOI: 10.1080/00958972.2011.648929.
Shapiro, R., Cohen, B. I., & Servis, R. E. (1970). Specific deamination of RNA by sodium bisulphite. Nature, 227, 1047–1048. DOI: 10.1038/2271047a0.
Shoair, A. G. F., & Mohamed, R. H. (2006). Improved procedure for ruthenium-catalyzed oxidative cleavage of alkenes with IO(OH)5. Synthetic Communications, 36, 59–64. DOI: 10.1080/00397910500328944.
Shono, T., Matsumura, Y., & Tsubata, K. (1979). One of the simplest methods for the replacement of diazonium groups by hydrogen or deuterium. Chemistry Letters, 8, 1051–1054. DOI: 10.1246/cl.1979.1051.
Silva, C. G., Silva, A. R., Ruschel, C., Helegda, C., Wyse, A. T. S., Wannmacher, C. M. D., Dutra-Filho, C. S., & Wajner, M. (2000). Inhibition of energy production in vitro by glutaric acid in cerebral cortex of young rats. Metabolic Brain Disease, 15, 123–131. DOI: 10.1007/bf02679979.
Skrunts, L. K., Kiprianova, L. A., Levit, A. F., Gragerov, I. P., Gordina, T. A., & Mkhitarov, R. A. (1983). Kinetics and mechanism of the reduction of diazonium salts by hypophosphite. Theoretical and Experimental Chemistry, 19, 98–101. DOI: 10.1007/bf00516867.
Smith, A. B. III, & Scarborough, R. M., Jr. (1980). Ruthenium tetroxide oxidation of simple ethers: A systematic study. Synthetic Communications, 10, 205–211. DOI: 10.1080/00397918008064223.
Tachikawa, Y., Cui, L., Matsusaki, Y., Tada, N., Miura, T., & Itoh, A. (2013). Aerobic photooxidative cleavage of 1,3-diketones to carboxylic acids using 2-chloroanthraquinone. Tetrahedron Letters, 54, 6218–6221. DOI: 10.1016/j.tetlet.2013.09.015.
Travis, B. R., Narayan, R. S., & Borhan, B. (2002). Osmium tetroxide-promoted catalytic oxidative cleavage of olefins: An organometallic ozonolysis. Journal of the American Chemical Society, 124, 3824–3825. DOI: 10.1021/ja017295g.
Tullo, A. (2002). Glutaric acid debuts. Chemical & Engineering News, 80, 13–13. DOI: 10.1021/cen-v080n016.p013.
Wallingford, V. H., & Krueger, P. A. (1939). m-Iodobenzoic acid. Organic Syntheses, 19, 57–59. DOI: 10.15227/orgsyn.019.0057.
Wang, W. J., & Dai, Q. H. (2002). Carbon-carbon double bond cleavage by silica-supported potassium permanganate under conditions free of organic solvent. Chinese Journal of Synthetic Chemistry, 10, 422–424. DOI: 10.3969/j.issn.1005-1511.2002.05.010.
Wassmundt, F. W., & Kiesman, W. F. (1997). Detection of aryl radicals in hydrodediazoniations. The Journal of Organic Chemistry, 62, 8304–8308. DOI: 10.1021/jo962128y.
Yuan, Y., Ji, X., & Zhao, D. (2010). Efficient oxidative cleavage of 1,3-dicarbonyl derivatives with hydrogen peroxide catalyzed by quaternary ammounium iodide. European Journal of Organic Chemistry, 2010, 5274–5278. DOI: 10.1002/ejoc.201000666.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zhang, W., Rao, MY., Cheng, ZJ. et al. Efficient synthesis of glutaric acid from l-glutamic acid via diazoniation/hydrogenation sequence. Chem. Pap. 69, 716–721 (2015). https://doi.org/10.1515/chempap-2015-0067
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1515/chempap-2015-0067