Abstract
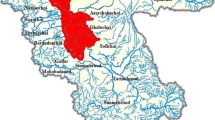
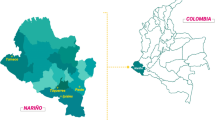
This research was undertaken to study the molecular detection and characterization of Cryptosporidium spp. in environmental water sources at Samsun and Giresun Provinces of The Black Sea in Turkey. Two-hundred forty and one-hundred eighty environmental samples were collected from a total of twenty and twenty-five sampling sites of Giresun and Samsun Provinces. One hundred twenty untreated drinking water samples were also detected for Cryptosporidium spp. in both investigated areas. 101 (%42), 92 (%38.3) of 240 and 74 (41.1%), 70 (38.8%) of 180 environmental samples have been found positive for Cryptosporidium spp. by Loop mediated isothermal amplification (LAMP) targeting the S-adenosyl-L-methionine synthetase (SAM) gene and nested PCR targeting small subunit (SSU)rRNA gene in Samsun and Giresun Provinces, respectively. Of the tested untreated drinking water samples collected from the investigated area, one sample was positive for Cryptosporidium spp. Six and twelve samples were clearly sequenced for the Cryptosporidium (SSU)rRNA gene among the highest positive samples selected from each of the twenty and twenty-five sampling sites of Giresun and Samsun Provinces, respectively. Genetic characterization of Cryptosporidium isolates from water samples represented Cryptosporidium bovis for five samples, Cryptosporidium parvum for six samples and one sample for Cryptosporidium felis in Samsun Province, where C. parvum for five samples and C. bovis for one sample were sequenced in Giresun Province. According to accessible information sources, this is the first research about genotyping of Cryptosporidium spp. in water samples collected from Samsun and Giresun Provinces of Turkey.
Similar content being viewed by others
References
Bakheit M.A., Palomino L., Thekisoe O.M.M., Mate P.A., Ongerth J., Karanis P. 2008: Sensitive and specific detection of Cryptosporidium species in PCR-negative samples by loop-mediated isothermal DNA amplification and confirmation of generated LAMP products by sequencing. Veterinary Parasitology, 158, 11–22. DOI: 10.1016/j.vetpar.2008.09.012
Caccio S.M., Ryan U. 2008. Molecular epidemiology of giardiasis. Molecular and Biochemical Parasitology, 160, 75–80. DOI: 10.1016/j.molbiopara.2008.04.006
Castro-Hermida J.A., Garci’a-Presedo I., Almeida A., Gonza’lez-Warleta V.J., Correia Da Costa M., Mezo V. 2008. Contribution of treated wastewater to the contamination of recreational river areas with Cryptosporidium spp. and Giardia duodenalis. Water Research, 42, 3528–3538. DOI:10.1016/j.watres.2008.05.001
Fall A., Thompson R., Hobbs R., Ryan U. 2003. Morphology is not a reliable tool for delineating species within Cryptosporidium. Journal of Parasitology, 89, 399–402.DOI: 10.1645/0022-3395(2003)089[0399:MINART]2.0.CO;2
Francy D.S., Bushon R.N., Brady A.M.G., Bertke E.E., Kephart C.M., Likiardopulos C.A., Maillot B.E., Schaefer F.W., Lindquist A.H.D. 2009. Performance of traditional and molecular methods for detecting biological agents in drinking water. US Geological Survey Scientific Investigations Report 2009–5097
Glaberman S., Moore J.E., Lowery C.J., Chalmers R.M., Sulaiman I., Elwin K., Rooney P.J., Millar B.C., Dooley J.S.G., Lal A.A., Xiao L. 2002. Three drinking-water-associated cryptosporidiosis outbreaks, Northern Ireland. Emerging Infectious Diseases, 8, 631–633. DOI: 10.3201/eid0806.010368
Hall TA. 1999. BioEdit: a user-friendly biological sequence alignment editor and analysis program for Windows 95/98/NT. Nucleic Acids Symposium Series. 41: 95–98
Jiang J., Xiao L. 2003. An evaluation of molecular diagnostic tools for thedetection and differentiation of human pathogenic Cryptosporidium spp. Journal of Eukaryotic Microbiology, 50, 542–547. DOI: 10.1111/j.1550-7408.2003.tb00623.x
Josephine N.G., Yang R., Whiffin V., Cox P., Ryan U. 2011. Identification of zoonotic Cryptosporidium and Giardia genotypes infecting animals in Sydney’s water catchments. Experimental Parasitology, 128, 138–144
Karanis P., Thekisoe O., Kiouptsi K., Ongerth J., Igarashi I., Inoue N. 2007. Development and preliminary evaluation of loop-mediated isothermal amplification (LAMP) for sensitive detection of Cryptosporidium oocysts in fecal and water samples. Applied and Environmental Microbiology, 73, 5660–5662. DOI: 10.1128/AEM.01152-07
Koksal F. 2002. Investigation of Source Waters for Giardia and Cryptosporidium. Journal of Turkish Society of Microbiology, 32, 275–277
Koloren Z., Delioglu B.K. 2011. Prevalence of Cryptosporidium species in water supplies of Amasya, Middle Black Sea, by Acid-Fast staining methods. Journal of Applied Biological Sciences, 5, 81–84
Koloren Z., Karanis P., Sotiriadou I. 2011. Investigations and Comparative Detection of Cryptosporidium Species by Microscopy, Nested PCR and LAMP in Water Supplies of Ordu, Middle Black Sea, Turkey. Annals of Tropical Medicine and Parasitology, 105, 607–615. DOI: 10.1179/2047773211Y.0000000011
Koloren Z., Kaya D., Avçar C. 2013. Detection of Cryptosporidium species in the sea and tap water samples of Black Sea, Turkey. Journal ofParasitology, 99, 554–557. DOI: 10.1645/GE-3232.1
Lobo M.L., Xiao L., Antunes F., Matos O. 2009. Occurrence of Cryptosporidium and Giardia genotypes and subtypes in raw and treated water in Portugal. Letters in Applied Microbiology, 48, 732–737. DOI: 10.1111/j.1472-765X.2009.02605.x
McLauchlin J., Amar C., Pedraza-Diaz S., Nichols G.L. 2000. Molecular epidemiological analysis of Cryptosporidium spp. in the United Kingdom: results of genotyping Cryptosporidium spp. in fecal samples from humans and fecal samples from livestock animals. Journal of Clinical Microbiology, 38, 3984–3990
Minarovicová J., Kachkova E., Krascsenicsova K., Siekel P., Kuchta T. 2009. A single-tube nested real-time polymerase chain reaction for sensitive contained detection of Cryptosporidium parvum. Letters in Applied Microbiology, 49, 568–572. DOI:10.1111/j.1472-765X.2009.02708.x
Monis P.T., Saint C.P. 2001. Development of a nested-PCR assay for the detection of Cryptosporidium parvum in finished water. Water Research, 35, 1641–1648. DOI: 10.1016/S0043-1354(00)00426-7
Nichols R.A.B., Campbell B.M., Smith H.V. 2003. Identification of Cryptosporidium oocysts in United Kingdom noncarbonated natural mineral waters and drinking waters by using a modified nested PCR-restriction fragment length polymorphism assay. Applied and Environmental Microbiology, 69, 4183–4189. DOI: 10.1128/AEM.69.7.4183-4189.2003
Plutzer J., Karanis P., Domokos K., Torokne A., Marialigeti K. 2008. Detection and characterisation of Giardia and Cryptosporidium in Hungarian raw, surface and sewage water samples by IFT, PCR and sequence analysis of the SSUrRNA and GDH genes. International Journal of Hygiene and Environmental Health, 211, 524–533. DOI: 10.1016/j.ijheh.2008.04.004
Ryan U., Read C., Hawkins P., Warnecke M., Swanson P., Griffith M., Deere D., Cunningham M., Cox P. 2005. Genotypes of Cryptosporidium from Sydney water catchment areas. Journal of Applied Microbiology, 98, 1221–1229. DOI: 10.1111/j.1365-2672.2005.02562.x
Smith H.V., Caccio S.M., Tait A., Mc Lauchlin J., Thompson R.C.A. 2006. Tools for investigating the environmental transmission of Cryptosporidium and Giardia infections in humans. Trends in Parasitology, 22, 160–167. DOI: 10.1016/j.pt.2006.02.009
Spano F., Crisanti A. 2000. Cryptosporidiumparvum: The many secrets of a small genome. International Journal for Parasitology, 30, 553–565. DOI: 10.1016/S0020-7519(99)00188-5
Terzi G. 2005. Importance of Food-Borne Protozoal Infections for Public Health. Yuzuncu Yil University. Journal of Veterinary Medicine Faculty, 16, 47–55
Xiao L. 2010. Molecular epidemiology of cryptosporidiosis: an update. Experimental Parasitology, 124, 80–89. DOI: 10.1016/j.exppara.2009.03.018
Xiao L., Alderisio K., Limor J., Royer M., Lal A.A. 2000. Identification of species and sources of Cryptosporidium oocysts in storm waters with a small-subunit rRNA-based diagnostic and genotyping tool. Applied and Environmental Microbiology, 66, 5492–5498. DOI: 10.1128/AEM.66.12.5492-5498.2000
Xiao L., Fayer R. 2008. Molecular characterization of species and genotypes of Cryptosporidium and Giardia and assessment of zoonotic transmission. International Journal for Parasitology, 38, 1239–1255. DOI: 10.1016/j.ijpara.2008.03.006
Xiao L., Fayer R., Ryan U., Upton S.J. 2004. Cryptosporidium taxonomy: recent advances and implications for public health. Critical Reviews in Microbiology, 17, 72–97. DOI: 10.1128/CMR.17.1.72-97.2004
Xiao L., Ryan U.M. 2006. Molecular epidemiology. In: (Eds. Fayer, R. and Xiao, L.) Cryptosporidium and Cryptosporidiosis. CRS Press, Taylor & Francis Group, Boca Raton, 119–171
Xiao L., Singh A., Limor J., Graczyk T.K., Gradus S., Lal A.A. 2001. Molecular characterization of Cryptosporidium oocysts in samples of raw surface water and wastewater. Applied and Environmental Microbiology, 67, 1097–1101. DOI: 10.1128/ AEM.67.3.1097-1101.2001
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Koloren, Z., Ayaz, E. Genotyping of Cryptosporidium spp. in environmental water in Turkey. Acta Parasit. 61, 671–679 (2016). https://doi.org/10.1515/ap-2016-0094
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1515/ap-2016-0094