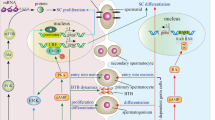
Abstract
Spermatogenesis is contingent upon hormones and growth factors acting through endocrine and paracrine pathways either in vivo or in vitro. Sertoli cells (SCs) furnish essential factors for the successful advancement of spermatogenesis and spermiogenesis. Moreover, receptors for follicle stimulating hormone (FSH) and testosterone, which are the main hormonal regulators of spermatogenesis, are identified on SCs. Testosterone, FSH and luteinizing hormone are known to determine the destiny of germ cells and in their absence germ cells undergo apoptosis. Bcl-2 family proteins determine one signaling pathway which seems to be crucial for the homeostasis of male gametes. In addition to paracrine signals, germ cell development also relies on signals generated by SCs via direct membrane contact. The regulatory peptide somatostatin has an important role in the regulation of the proliferation of the male germ cells. Activin A, follistatin and FSH control germ cell development. In vitro culture systems have provided initial evidence supporting the achievement of the completion of the first and second male meiotic division in vitro. This review article provides an overview of the literature regarding the hormonal pathways governing spermatogenesis and spermiogenesis.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
de Rooij DG, 2009 The spermatogonial stem cell niche. Microsc Res Tech 72: 580–585.
Stukenborg JB, Wistuba J, Luetjens CM, et al, 2008 Coculture of spermatogonia with somatic cells in a novel three-dimensional soft-agar-culture-system. J Androl 29: 312–329.
Kanatsu-Shinohara M, Toyokuni S, Morimoto T, Matsui S, Honjo T, Shinohara T, 2003 Functional assessment of self-renewal activity of male germline stem cells following cytotoxic damage and serial transplantation. Biol Reprod 68: 1801–1807.
Ogawa T, Ohmura M, Yumura Y, Sawada H, Kubota Y, 2003 Expansion of murine spermatogonial stem cells through serial transplantation. Biol Reprod 68: 316–322.
Vergouwen RP, Jacobs SG, Huiskamp R, Davids JA, de Rooij DG, 1991 Proliferative activity of gonocytes, Sertoli cells and interstitial cells during testicular development in mice. J Reprod Fertil 93: 233–243.
Kierszenbaum AL, 1994 Mammalian spermatogenesis in vivo and in vitro: a partnership of spermatogenic and somatic cell lineages. Endocr Rev 15: 116–134.
Yamashita YM, Jones DL, Fuller MT, 2003 Orientation of asymmetric stem cell division by the APC tumor suppressor and centrosome. Science 301: 1547–1550.
Kiger AA, Jones DL, Schulz C, Rogers MB, Fuller MT, 2001 Stem cell self-renewal specified by JAK-STAT activation in response to a support cell cue. Science 294: 2542–2545.
Tulina N, Matunis E, 2001 Control of stem cell self-renewal in Drosophila spermatogenesis by JAK-STAT signaling. Science 294: 2546–2549.
Sefton L, Timmer JR, Zhang Y, Beranger F, Cline TW, 2000 An extracellular activator of the Drosophila JAK/STAT pathway is a sex-determination signal element. Nature 405: 970–973.
Binari R, Perrimon N, 1994 Stripe-specific regulation of pair-rule genes by hopscotch, a putative Jak family tyrosine kinase in Drosophila. Genes Dev 8: 300–312.
Hou XS, Melnick MB, Perrimon N, 1996 Marelle acts downstream of the Drosophila HOP/JAK kinase and encodes a protein similar to the mammalian STATs. Cell 84: 411–419.
Aponte PM, van Bragt MP, de Rooij DG, van Pelt AM, 2005 Spermatogonial stem cells: characteristics and experimental possibilities. APMIS 113: 727–742.
Clermont Y, 1972 Kinetics of spermatogenesis in mammals: seminiferous epithelium cycle and spermatogonial renewal. Physiol Rev 52: 198–236.
Ehmcke J, Schlatt S, 2006 A revised model for spermatogonial expansion in man: lessons from non-human primates. Reproduction 132: 673–680.
Ehmcke J, Wistuba J, Schlatt S, 2006 Spermatogonial stem cells: questions, models and perspectives. Hum Reprod Update 12: 275–282.
Roosen-Runge EC, Barlow FD, 1953 Quantitative studies on human spermatogenesis. I. Spermatogonia. Am J Anat 93: 143–169.
Amann RP, 2008 The cycle of the seminiferous epithelium in humans: a need to revisit? J Androl 29: 469–487.
McLachlan RI, O’Donnell L, Meachem SJ, et al, 2002 Identification of specific sites of hormonal regulation in spermatogenesis in rats, monkeys, and man. Recent Prog Horm Res 57: 149–179.
Yoshida S, Sukeno M, Nabeshima Y, 2007 A vasculature-associated niche for undifferentiated spermatogonia in the mouse testis. Science 317: 1722–1726.
Griswold MD, 1998 The central role of Sertoli cells in spermatogenesis. Semin Cell Dev Biol 9: 411–416.
Meachem S, von Schonfeldt V, Schlatt S, 2001 Spermatogonia: stem cells with a great perspective. Reproduction 121: 825–834.
Hess RA, Cooke PS, Bunick D, Kirby JD, 1993 Adult testicular enlargement induced by neonatal hypothyroidism is accompanied by increased Sertoli and germ cell numbers. Endocrinology 132: 2607–2613.
Orth JM, Gunsalus GL, Lamperti AA, 1988 Evidence from Sertoli cell-depleted rats indicates that spermatid number in adults depends on numbers of Sertoli cells produced during perinatal development. Endocrinology 122: 787–794.
Simorangkir DR, de Kretser DM, Wreford NG, 1995 Increased numbers of Sertoli and germ cells in adult rat testes induced by synergistic action of transient neonatal hypothyroidism and neonatal hemicastration. J Reprod Fertil 104: 207–213.
Debeljuk L, Rao JN, Bartke A, 2003 Tachykinins and their possible modulatory role on testicular function: a review. Int J Androl 26: 202–210.
Allard EK, Blanchard KT, Boekelheide K, 1996 Exogenous stem cell factor (SCF) compensates for altered endogenous SCF expression in 2,5-hexanedione-induced testicular atrophy in rats. Biol Reprod 55: 185–193.
Blanchard KT, Lee J, Boekelheide K, 1998 Leuprolide, a gonadotropin-releasing hormone agonist, reestablishes spermatogenesis after 2,5-hexanedione-induced irreversible testicular injury in the rat, resulting in normalized stem cell factor expression. Endocrinology 139: 236–244.
Marziali G, Lazzaro D, Sorrentino V, 1993 Binding of germ cells to mutant Sld Sertoli cells is defective and is rescued by expression of the transmembrane form of the c-kit ligand. Dev Biol 157: 182–190.
Wu JC, Gregory CW, DePhilip RM, 1993 Expression of E-cadherin in immature rat and mouse testis and in rat Sertoli cell cultures. Biol Reprod 49: 1353–1361.
Mruk DD, Cheng CY, 2004 Sertoli-Sertoli and Sertoli-germ cell interactions and their significance in germ cell movement in the seminiferous epithelium during spermatogenesis. Endocr Rev 25: 747–806.
Erkkila K, Aito H, Aalto K, Pentikainen V, Dunkel L, 2002 Lactate inhibits germ cell apoptosis in the human testis. Mol Hum Reprod 8: 109–117.
Riera MF, Meroni SB, Gomez GE, Schteingart HF, Pellizzari EH, Cigorraga SB, 2001 Regulation of lactate production by FSH, iL1beta, and TNFalpha in rat Sertoli cells. Gen Comp Endocrinol 122: 88–97.
Riera MF, Meroni SB, Schteingart HF, Pellizzari EH, Cigorraga SB, 2002 Regulation of lactate production and glucose transport as well as of glucose transporter 1 and lactate dehydrogenase A mRNA levels by basic fibroblast growth factor in rat Sertoli cells. J Endocrinol 173: 335–343.
Alves MG, Dias TR, Silva BM, Oliveira PF, 2014 Metabolic cooperation in testis as a pharmacological target: from disease to contraception. Curr Mol Pharmacol 7: 83–95.
Robinson R, Fritz IB, 1981 Metabolism of glucose by Sertoli cells in culture. Biol Reprod 24: 1032–1041.
Oliveira PF, Martins AD, Moreira AC, Cheng CY, Alves MG, 2015 The Warburg effect revisited—lesson from the Sertoli cell. Med Res Rev 35: 126–151.
Boussouar F, Benahmed M, 2004 Lactate and energy metabolism in male germ cells. Trends Endocrinol Metab 15: 345–350.
Mullaney BP, Rosselli M, Skinner MK, 1994 Developmental regulation of Sertoli cell lactate production by hormones and the testicular paracrine factor, PModS. Mol Cell Endocrinol 104: 67–73.
Dias TR, Alves MG, Silva BM, Oliveira PF, 2014 Sperm glucose transport and metabolism in diabetic individuals. Mol Cell Endocrinol 396: 37–45.
Rato L, Alves MG, Socorro S, Carvalho RA, Cavaco JE, Oliveira PF, 2012 Metabolic modulation induced by oestradiol and DHT in immature rat Sertoli cells cultured in vitro. Biosci Rep 32: 61–69.
Oliveira PF, Alves MG, Rato L, et al, 2012 Effect of insulin deprivation on metabolism and metabolism-associated gene transcript levels of in vitro cultured human Sertoli cells. Biochim Biophys Acta 1820: 84–89.
Oliveira PF, Alves MG, Rato L, et al, 2011 Influence of 5alpha-dihydrotestosterone and 17beta-estradiol on human Sertoli cells metabolism. Int J Androl 34: e612–620.
Alves MG, Rato L, Carvalho RA, Moreira PI, Socorro S, Oliveira PF, 2013 Hormonal control of Sertoli cell metabolism regulates spermatogenesis. Cell Mol Life Sci 70: 777–793.
Martins AD, Alves MG, Simoes VL, et al, 2013 Control of Sertoli cell metabolism by sex steroid hormones is mediated through modulation in glycolysis-related transporters and enzymes. Cell Tissue Res 354: 861–868.
De Gendt K, Swinnen JV, Saunders PT, et al, 2004 A Sertoli cell-selective knockout of the androgen receptor causes spermatogenic arrest in meiosis. Proc Natl Acad Sci U S A 101: 1327–1332.
Zhou Q, Nie R, Prins GS, Saunders PT, Katzenellenbogen BS, Hess RA, 2002 Localization of androgen and estrogen receptors in adult male mouse reproductive tract. J Androl 23: 870–881.
Holdcraft RW, Braun RE, 2004 Androgen receptor function is required in Sertoli cells for the terminal differentiation of haploid spermatids. Development 131: 459–467.
Free MJ, Jaffe RA, 1979 Collection of rete testis fluid from rats without previous efferent duct ligation. Biol Reprod 20: 269–278.
Hess RA, 2000 Oestrogen in fluid transport in efferent ducts of the male reproductive tract. Rev Reprod 5: 84–92.
Carreau S, Delalande C, Galeraud-Denis I, 2009 Mammalian sperm quality and aromatase expression. Microsc Res Tech 72: 552–557.
Li H, Papadopoulos V, Vidic B, Dym M, Culty M, 1997 Regulation of rat testis gonocyte proliferation by platelet-derived growth factor and estradiol: identification of signaling mechanisms involved. Endocrinology 138: 1289–1298.
O’Donnell L, Robertson KM, Jones ME, Simpson ER, 2001 Estrogen and spermatogenesis. Endocr Rev 22: 289–318.
Cheng CY, Boettcher B, 1979 The effect of steroids on the in vitro migration of washed human spermatozoa in modified Tyrode’s solution or in fasting human blood serum. Fertil Steril 32: 566–570.
Revelli A, Massobrio M, Tesarik J, 1998 Nongenomic actions of steroid hormones in reproductive tissues. Endocr Rev 19: 3–17.
Hicks JJ, Pedron N, Rosado A, 1972 Modifications of human spermatozoa glycolysis by cyclic adenosine monophosphate (cAMP), estrogens, and follicular fluid. Fertil Steril 23: 886–893.
Mishra DP, Shaha C, 2005 Estrogen-induced spermatogenic cell apoptosis occurs via the mitochondrial pathway: role of superoxide and nitric oxide. J Biol Chem 280: 6181–6196.
Mruk DD, Cheng CY, 2010 Tight junctions in the testis: new perspectives. Philos Trans R Soc Lond B Biol Sci 365: 1621–1635.
Russell L, 1977 Movement of spermatocytes from the basal to the adluminal compartment of the rat testis. Am J Anat 148: 313–328.
Fijak M, Bhushan S, Meinhardt A, 2011 Immunoprivileged sites: the testis. Methods Mol Biol 677: 459–470.
Setchell BP, Scott TW, Voglmayr JK, Waites GM, 1969 Characteristics of testicular spermatozoa and the fluid which transports them into the epididymis. Biol Reprod 1: Suppl 1: 40–66.
Setchell BP, Voglmayr JK, Waites GM, 1969 A blood-testis barrier restricting passage from blood into rete testis fluid but not into lymph. J Physiol 200: 73–85.
Alves MG, Martins AD, Cavaco JE, Socorro S, Oliveira PF, 2013 Diabetes, insulin-mediated glucose metabolism and Sertoli/blood-testis barrier function. Tissue Barriers 1: e23992.
Rato L, Alves MG, Socorro S, Duarte AI, Cavaco JE, Oliveira PF, 2012 Metabolic regulation is important for spermatogenesis. Nat Rev Urol 9: 330–338.
Rebourcet D, O’Shaughnessy PJ, Pitetti JL, et al, 2014 Sertoli cells control peritubular myoid cell fate and support adult Leydig cell development in the prepubertal testis. Development 141: 2139–2149.
Kumar TR, Wang Y, Lu N, Matzuk MM, 1997 Follicle stimulating hormone is required for ovarian follicle maturation but not male fertility. Nat Genet 15: 201–204.
Tapanainen JS, Aittomaki K, Min J, Vaskivuo T, Huhtaniemi IT, 1997 Men homozygous for an inactivating mutation of the follicle-stimulating hormone (FSH) receptor gene present variable suppression of spermatogenesis and fertility. Nat Genet 15: 205–206.
Dierich A, Sairam MR, Monaco L, et al, 1998 Impairing follicle-stimulating hormone (FSH) signaling in vivo: targeted disruption of the FSH receptor leads to aberrant gametogenesis and hormonal imbalance. Proc Natl Acad Sci U S A 95: 13612–13617.
Marshall GR, Zorub DS, Plant TM, 1995 Follicle-stimulating hormone amplifies the population of differentiated spermatogonia in the hypophysectomized testosterone-replaced adult rhesus monkey (Macaca mulatta). Endocrinology 136: 3504–3511.
Erkkila K, Henriksen K, Hirvonen V, et al, 1997 Testosterone regulates apoptosis in adult human seminiferous tubules in vitro. J Clin Endocrinol Metab 82: 2314–2321.
Tesarik J, Greco E, Mendoza C, 2001 Assisted reproduction with in-vitro-cultured testicular spermatozoa in cases of severe germ cell apoptosis: a pilot study. Hum Reprod 16: 2640–2645.
Lee J, Richburg JH, Younkin SC, Boekelheide K, 1997 The Fas system is a key regulator of germ cell apoptosis in the testis. Endocrinology 138: 2081–2088.
Dimitriadis F, Tsambalas S, Tsounapi P, et al, 2010 Effects of phosphodiesterase-5 inhibitors on Leydig cell secretory function in oligoasthenospermic infertile men: a randomized trial. BJU Int 106: 1181–1185.
Dimitriadis F, Tsampalas S, Tsounapi P, et al, 2012 Effects of phosphodiesterase-5 inhibitor vardenafil on testicular androgen-binding protein secretion, the maintenance of foci of advanced spermatogenesis and the sperm fertilising capacity in azoospermic men. Andrologia 44: Suppl 1: 144–153.
Wayman C, Phillips S, Lunny C, et al, 2005 Phosphodiesterase 11 (PDE11) regulation of spermatozoa physiology. Int J Impot Res 17: 216–223.
Melino G, Knight RA, Nicotera P, 2005 How many ways to die? How many different models of cell death? Cell Death Differ 12: Suppl 2: 1457–1462.
Lockshin RA, Zakeri Z, 2004 Caspase-independent cell death? Oncogene 23: 2766–2773.
Shaha C, 2007 Modulators of spermatogenic cell survival. Soc Reprod Fertil Suppl 63: 173–186.
Sinha Hikim AP, Swerdloff RS, 1999 Hormonal and genetic control of germ cell apoptosis in the testis. Rev Reprod 4: 38–47.
Show MD, Folmer JS, Anway MD, Zirkin BR, 2004 Testicular expression and distribution of the rat bcl2 modifying factor in response to reduced intratesticular testosterone. Biol Reprod 70: 1153–1161.
Mayer F, Honecker F, Looijenga LH, Bokemeyer C, 2003 Towards an understanding of the biological basis of response to cisplatin-based chemotherapy in germ-cell tumors. Ann Oncol 14: 825–832.
Hikim AP, Wang C, Lue Y, Johnson L, Wang XH, Swerdloff RS, 1998 Spontaneous germ cell apoptosis in humans: evidence for ethnic differences in the susceptibility of germ cells to programmed cell death. J Clin Endocrinol Metab 83: 152–156.
Print CG, Loveland KL, 2000 Germ cell suicide: new insights into apoptosis during spermatogenesis. Bioessays 22: 423–430.
Gnessi L, Fabbri A, Spera G, 1997 Gonadal peptides as mediators of development and functional control of the testis: an integrated system with hormones and local environment. Endocr Rev 18: 541–609.
Schlatt S, Meinhardt A, Nieschlag E, 1997 Paracrine regulation of cellular interactions in the testis: factors in search of a function. Eur J Endocrinol 137: 107–117.
Zou H, Henzel WJ, Liu X, Lutschg A, Wang X, 1997 Apaf-1, a human protein homologous to C. elegans CED-4, participates in cytochrome c-dependent activation of caspase-3. Cell 90: 405–413.
Ross AJ, Waymire KG, Moss JE, et al, 1998 Testicular degeneration in Bclw-deficient mice. Nat Genet 18: 251–256.
Yan W, Samson M, Jegou B, Toppari J, 2000 Bcl-w forms complexes with Bax and Bak, and elevated ratios of Bax/Bcl-w and Bak/Bcl-w correspond to spermatogonial and spermatocyte apoptosis in the testis. Mol Endocrinol 14: 682–699.
Erkkila K, Pentikainen V, Wikstrom M, Parvinen M, Dunkel L, 1999 Partial oxygen pressure and mitochondrial permeability transition affect germ cell apoptosis in the human testis. J Clin Endocrinol Metab 84: 4253–4259.
Adams JM, Cory S, 1998 The Bcl-2 protein family: arbiters of cell survival. Science 281: 1322–1326.
Green DR, 2000 Apoptotic pathways: paper wraps stone blunts scissors. Cell 102: 1–4.
Oldereid NB, Angelis PD, Wiger R, Clausen OP, 2001 Expression of Bcl-2 family proteins and spontaneous apoptosis in normal human testis. Mol Hum Reprod 7: 403–408.
Honarpour N, Du C, Richardson JA, et al, 2000 Adult Apaf-1-deficient mice exhibit male infertility. Dev Biol 218: 248–258.
Narisawa S, Hecht NB, Goldberg E, Boatright KM, Reed JC, Millan JL, 2002 Testis-specific cytochrome c-null mice produce functional sperm but undergo early testicular atrophy. Mol Cell Biol 22: 5554–5562.
Kimura M, Itoh N, Takagi S, et al, 2003 Balance of apoptosis and proliferation of germ cells related to spermatogenesis in aged men. J Androl 24: 185–191.
Watanabe-Fukunaga R, Brannan CI, Itoh N, et al, 1992 The cDNA structure, expression, and chromosomal assignment of the mouse Fas antigen. J Immunol 148: 1274–1279.
Suda T, Takahashi T, Golstein P, Nagata S, 1993 Molecular cloning and expression of the Fas ligand, a novel member of the tumor necrosis factor family. Cell 75: 1169–1178.
Bellgrau D, Gold D, Selawry H, Moore J, Franzusoff A, Duke RC, 1995 A role for CD95 ligand in preventing graft rejection. Nature 377: 630–632.
Sofikitis N, Kaponis A, Mio Y, et al, 2003 Germ cell transplantation: a review and progress report on ICSI from spermatozoa generated in xenogeneic testes. Hum Reprod Update 9: 291–307.
Nagata S, Golstein P, 1995 The Fas death factor. Science 267: 1449–1456.
Pentikainen V, Erkkila K, Dunkel L, 1999 Fas regulates germ cell apoptosis in the human testis in vitro. Am J Physiol 276: E310–316.
Sugihara A, Saiki S, Tsuji M, et al, 1997 Expression of Fas and Fas ligand in the testes and testicular germ cell tumors: an immunohistochemical study. Anticancer Res 17: 3861–3865.
Francavilla S, D’Abrizio P, Rucci N, et al, 2000 Fas and Fas ligand expression in fetal and adult human testis with normal or deranged spermatogenesis. J Clin Endocrinol Metab 85: 2692–2700.
Giebel LB, Strunk KM, Holmes SA, Spritz RA, 1992 Organization and nucleotide sequence of the human KIT (mast/stem cell growth factor receptor) protooncogene. Oncogene 7: 2207–2217.
Vandenbark GR, deCastro CM, Taylor H, Dew-Knight S, Kaufman RE, 1992 Cloning and structural analysis of the human c-kit gene. Oncogene 7: 1259–1266.
Flanagan JG, Chan DC, Leder P, 1991 Transmembrane form of the kit ligand growth factor is determined by alternative splicing and is missing in the Sld mutant. Cell 64: 1025–1035.
Manova K, Nocka K, Besmer P, Bachvarova RF, 1990 Gonadal expression of c-kit encoded at the W locus of the mouse. Development 110: 1057–1069.
Orth JM, Jester WF Jr., Qiu J, 1996 Gonocytes in testes of neonatal rats express the c-kit gene. Mol Reprod Dev 45: 123–131.
Rossi P, Albanesi C, Grimaldi P, Geremia R, 1991 Expression of the mRNA for the ligand of c-kit in mouse Sertoli cells. Biochem Biophys Res Commun 176: 910–914.
Mauduit C, Hamamah S, Benahmed M, 1999 Stem cell factor/c-kit system in spermatogenesis. Hum Reprod Update 5: 535–545.
Korbutt GS, Elliott JF, Rajotte RV, 1997 Cotransplantation of allogeneic islets with allogeneic testicular cell aggregates allows long-term graft survival without systemic immunosuppression. Diabetes 46: 317–322.
Riccioli A, Salvati L, D’Alessio A, et al, 2003 The Fas system in the seminiferous epithelium and its possible extra-testicular role. Andrologia 35: 64–70.
Dias TR, Rato L, Martins AD, et al, 2013 Insulin deprivation decreases caspase-dependent apoptotic signaling in cultured rat Sertoli cells. ISRN Urol 2013: 970370.
Simoes VL, Alves MG, Martins AD, et al, 2013 Regulation of apoptotic signaling pathways by 5alpha-dihydrotestosterone and 17beta-estradiol in immature rat Sertoli cells. J Steroid Biochem Mol Biol 135: 15–23.
Cande C, Cohen I, Daugas E, et al, 2002 Apoptosis-inducing factor (AIF): a novel caspase-independent death effector released from mitochondria. Biochimie 84: 215–222.
Zou H, Yang R, Hao J, et al, 2003 Regulation of the Apaf-1/caspase-9 apoptosome by caspase-3 and XIAP. J Biol Chem 278: 8091–8098.
Page ST, 2011 Physiologic role and regulation of intratesticular sex steroids. Curr Opin Endocrinol Diabetes Obes 18: 217–223.
Lim P, Allan CM, Notini AJ, et al, 2008 Oestradiol-induced spermatogenesis requires a functional androgen receptor. Reprod Fertil Dev 20: 861–870.
Andersson AM, Jorgensen N, Frydelund-Larsen L, Rajpert-De Meyts E, Skakkebaek NE, 2004 Impaired Leydig cell function in infertile men: a study of 357 idiopathic infertile men and 318 proven fertile controls. J Clin Endocrinol Metab 89: 3161–3167.
Bianco-Rodriguez J, Martinez-Garcia C, 1996 Induction of apoptotic cell death in the seminiferous tubule of the adult rat testis: assessment of the germ cell types that exhibit the ability to enter apoptosis after hormone suppression by oestradiol treatment. Int J Androl 19: 237–247.
Pentikainen V, Erkkila K, Suomalainen L, Parvinen M, Dunkel L, 2000 Estradiol acts as a germ cell survival factor in the human testis in vitro. J Clin Endocrinol Metab 85: 2057–2067.
Sofikitis N, Giotitsas N, Tsounapi P, Baltogiannis D, Giannakis D, Pardalidis N, 2008 Hormonal regulation of spermatogenesis and spermiogenesis. J Steroid Biochem Mol Biol 109: 323–330.
Patel YC, Greenwood MT, Panetta R, Demchyshyn L, Niznik H, Srikant CB, 1995 The somatostatin receptor family. Life Sci 57: 1249–1265.
Reubi JC, 1997 Relevance of Somatostatin Receptors and Other Peptide Receptors in Pathology. Endocr Pathol 8: 11–20.
Vasankari T, Kujala U, Taimela S, Torma A, Irjala K, Huhtaniemi I, 1995 Effects of a long acting somatostatin analog on pituitary, adrenal, and testicular function during rest and acute exercise: unexpected stimulation of testosterone secretion. J Clin Endocrinol Metab 80: 3298–3303.
Baou N, Bouras M, Droz JP, Benahmed M, Krantic S, 2000 Evidence for a selective loss of somatostatin receptor subtype expression in male germ cell tumors of seminoma type. Carcinogenesis 21: 805–810.
Bruno JF, Xu Y, Song J, Berelowitz M, 1993 Tissue distribution of somatostatin receptor subtype messenger ribonucleic acid in the rat. Endocrinology 133: 2561–2567.
Rohrer L, Raulf F, Bruns C, Buettner R, Hofstaedter F, Schule R, 1993 Cloning and characterization of a fourth human somatostatin receptor. Proc Natl Acad Sci U S A 90: 4196–4200.
Zhu LJ, Krempels K, Bardin CW, O’Carroll AM, Mezey E, 1998 The localization of messenger ribonucleic acids for somatostatin receptors 1, 2, and 3 in rat testis. Endocrinology 139: 350–357.
Goddard I, Bauer S, Gougeon A, et al, 2001 Somatostatin inhibits stem cell factor messenger RNA expression by Sertoli cells and stem cell factor-induced DNA synthesis in isolated seminiferous tubules. Biol Reprod 65: 1732–1742.
Grataroli R, Vindrieux D, Selva J, et al, 2004 Characterization of tumour necrosis factor-alpha-related apoptosis-inducing ligand and its receptors in the adult human testis. Mol Hum Reprod 10: 123–128.
Cooke JE, Heasman J, Wylie CC, 1996 The role of interleukin-4 in the regulation of mouse primordial germ cell numbers. Dev Biol 174: 14–21.
Dolci S, Williams DE, Ernst MK, et al, 1991 Requirement for mast cell growth factor for primordial germ cell survival in culture. Nature 352: 809–811.
Matsui Y, Zsebo K, Hogan BL, 1992 Derivation of pluripotential embryonic stem cells from murine primordial germ cells in culture. Cell 70: 841–847.
Baloh RH, Tansey MG, Lampe PA, et al, 1998 Artemin, a novel member of the GDNF ligand family, supports peripheral and central neurons and signals through the GFRalpha3-RET receptor complex. Neuron 21: 1291–1302.
Kotzbauer PT, Lampe PA, Heuckeroth RO, et al, 1996 Neurturin, a relative of glial-cell-line-derived neurotrophic factor. Nature 384: 467–470.
Lin LF, Doherty DH, Lile JD, Bektesh S, Collins F, 1993 GDNF: a glial cell line-derived neurotrophic factor for midbrain dopaminergic neurons. Science 260: 1130–1132.
Milbrandt J, de Sauvage FJ, Fahrner TJ, et al, 1998 Persephin, a novel neurotrophic factor related to GDNF and neurturin. Neuron 20: 245–253.
Trupp M, Ryden M, Jornvall H, et al, 1995 Peripheral expression and biological activities of GDNF, a new neurotrophic factor for avian and mammalian peripheral neurons. J Cell Biol 130: 137–148.
Meng X, Lindahl M, Hyvonen ME, et al, 2000 Regulation of cell fate decision of undifferentiated spermatogonia by GDNF. Science 287: 1489–1493.
Antoniades HN, Hunkapiller MW, 1983 Human platelet-derived growth factor (PDGF): amino-terminal amino acid sequence. Science 220: 963–965.
Heldin CH, Westermark B, 1999 Mechanism of action and in vivo role of platelet-derived growth factor. Physiol Rev 79: 1283–1316.
Basciani S, Mariani S, Arizzi M, et al, 2002 Expression of platelet-derived growth factor-A (PDGF-A), PDGF-B, and PDGF receptor-alpha and -beta during human testicular development and disease. J Clin Endocrinol Metab 87: 2310–2319.
Hasthorpe S, Barbic S, Farmer PJ, Hutson JM, 2000 Growth factor and somatic cell regulation of mouse gonocyte-derived colony formation in vitro. J Reprod Fertil 119: 85–91.
Hasthorpe S, 2003 Clonogenic culture of normal spermatogonia: in vitro regulation of postnatal germ cell proliferation. Biol Reprod 68: 1354–1360.
Nagano M, Ryu BY, Brinster CJ, Avarbock MR, Brinster RL, 2003 Maintenance of mouse male germ line stem cells in vitro. Biol Reprod 68: 2207–2214.
Ogawa T, Arechaga JM, Avarbock MR, Brinster RL, 1997 Transplantation of testis germinal cells into mouse seminiferous tubules. Int J Dev Biol 41: 111–122.
Meehan T, Schlatt S, O’Bryan MK, de Kretser DM, Loveland KL, 2000 Regulation of germ cell and Sertoli cell development by activin, follistatin, and FSH. Dev Biol 220: 225–237.
Toebosch AM, Robertson DM, Trapman J, et al, 1988 Effects of FSH and IGF-I on immature rat Sertoli cells: inhibin alpha- and beta-subunit mRNA levels and inhibin secretion. Mol Cell Endocrinol 55: 101–105.
Boitani C, Stefanini M, Fragale A, Morena AR, 1995 Activin stimulates Sertoli cell proliferation in a defined period of rat testis development. Endocrinology 136: 5438–5444.
Mather JP, Artie KM, Woodruff TK, Rice GC, Phillips DM, 1990 Activin stimulates spermatogonial proliferation in germ-Sertoli cell cocultures from immature rat testis. Endocrinology 127: 3206–3214.
Haneji T, Koide SS, Tajima Y, Nishimune Y, 1991 Differential effects of epidermal growth factor on the differentiation of type A spermatogonia in adult mouse cryptorchid testes in vitro. J Endocrinol 128: 383–388.
Wahab-Wahlgren A, Martinelle N, Holst M, Jahnukainen K, Parvinen M, Soder O, 2003 EGF stimulates rat spermatogonial DNA synthesis in seminiferous tubule segments in vitro. Mol Cell Endocrinol 201: 39–46.
Dirami G, Ravindranath N, Pursei V, Dym M, 1999 Effects of stem cell factor and granulocyte macrophage-colony stimulating factor on survival of porcine type A spermatogonia cultured in KSOM. Biol Reprod 61: 225–230.
Nakayama Y, Yamamoto T, Abe SI, 1999 IGF-I, IGF-II and insulin promote differentiation of spermatogonia to primary spermatocytes in organ culture of newt testes. Int J Dev Biol 43: 343–347.
Tajima Y, Watanabe D, Koshimizu U, Matsuzawa T, Nishimune Y, 1995 Insulin-like growth factor-I and transforming growth factor-alpha stimulate differentiation of type A spermatogonia in organ culture of adult mouse cryptorchid testes. Int J Androl 18: 8–12.
Sousa M, Cremades N, Alves C, Silva J, Barros A, 2002 Developmental potential of human spermatogenic cells co-cultured with Sertoli cells. Hum Reprod 17: 161–172.
Tesarik J, Greco E, Rienzi L, et al, 1998 Differentiation of spermatogenic cells during in-vitro culture of testicular biopsy samples from patients with obstructive azoospermia: effect of recombinant follicle stimulating hormone. Hum Reprod 13: 2772–2781.
Tesarik J, Guido M, Mendoza C, Greco E, 1998 Human spermatogenesis in vitro: respective effects of follicle-stimulating hormone and testosterone on meiosis, spermiogenesis, and Sertoli cell apoptosis. J Clin Endocrinol Metab 83: 4467–4473.
Martin-du Pan RC, Campana A, 1993 Physiopathology of spermatogenic arrest. Fertil Steril 60: 937–946.
O’Donnell L, Nicholls PK, O’Bryan MK, McLachlan RI, Stanton PG, 2011 Spermiation: The process of sperm release. Spermatogenesis 1: 14–35.
Kerr JB, Millar M, Maddocks S, Sharpe RM, 1993 Stage-dependent changes in spermatogenesis and Sertoli cells in relation to the onset of spermatogenic failure following withdrawal of testosterone. Anat Rec 235: 547–559.
O’Donnell L, McLachlan RI, Wreford NG, de Kretser DM, Robertson DM, 1996 Testosterone withdrawal promotes stage-specific detachment of round spermatids from the rat seminiferous epithelium. Biol Reprod 55: 895–901.
Sofikitis N, Ono K, Yamamoto Y, Papadopoulos H, Miyagawa I, 1999 Influence of the male reproductive tract on the reproductive potential of round spermatids abnormally released from the seminiferous epithelium. Hum Reprod 14: 1998–2006.
Zirkin BR, 1998 Spermatogenesis: its regulation by testosterone and FSH. Semin Cell Dev Biol 9: 417–421.
Kim JM, Ghosh SR, Weil AC, Zirkin BR, 2001 Caspase-3 and caspase-activated deoxyribonuclease are associated with testicular germ cell apoptosis resulting from reduced intratesticular testosterone. Endocrinology 142: 3809–3816.
Puthalakath H, Villunger A, O’Reilly LA, et al, 2001 Bmf: a proapoptotic BH3-only protein regulated by interaction with the myosin V actin motor complex, activated by anoikis. Science 293: 1829–1832.
Russell LD, Weber JE, Vogl AW, 1986 Characterization of filaments within the subacrosomal space of rat spermatids during spermiogenesis. Tissue Cell 18: 887–898.
Sluka P, O’Donnell L, Barties JR, Stanton PG, 2006 FSH regulates the formation of adherens junctions and ectoplasmic specialisations between rat Sertoli cells in vitro and in vivo. J Endocrinol 189: 381–395.
Hannay T, 1995 New Japanese IVF method finally made available in Japan. Nat Med 1: 289–290.
Tesarik J, Mendoza C, Testart J, 1995 Viable embryos from injection of round spermatids into oocytes. N Engl J Med 333: 525.
Mansour RT, Aboulghar MA, Serour K, 1996 Pregnancy and delivery after intracytoplasmic injection of spermatids into human oocytes. Middle East Fertil. Soc. 1: 223–225.
Antinori S, Versaci C, Dani G, Antinori M, Pozza D, Selman HA, 1997 Fertilization with human testicular spermatids: four successful pregnancies. Hum Reprod 12: 286–291.
Vanderzwalmen P, Zech H, Birkenfeld A, et al, 1997 Intracytoplasmic injection of spermatids retrieved from testicular tissue: influence of testicular pathology, type of selected spermatids and oocyte activation. Hum Reprod 12: 1203–1213.
Amer M, Soliman E, el-Sadek M, Mendoza C, Tesarik J, 1997 Is complete spermiogenesis failure a good indication for spermatid conception? Lancet 350: 116.
Araki Y, Motoyama M, Yoshida A, Kim SY, Sung H, Araki S, 1997 Intracytoplasmic injection with late spermatids: a successful procedure in achieving childbirth for couples in which the male partner suffers from azoospermia due to deficient spermatogenesis. Fertil Steril 67: 559–561.
Yamanaka K, Sofikitis NV, Miyagawa I, et al, 1997 Ooplasmic round spermatid nuclear injection procedures as an experimental treatment for nonobstructive azoospermia. J Assist Reprod Genet 14: 55–62.
Bernabeu R, Cremades N, Takahashi K, Sousa M, 1998 Successful pregnancy after spermatid injection. Hum Reprod 13: 1898–1900.
Sofikitis N, Mantzavinos T, Loutradis D, Yamamoto Y, Tarlatzis V, Miyagawa I, 1998 Ooplasmic injections of secondary spermatocytes for non-obstructive azoospermia. Lancet 351: 1177–1178.
Sofikitis N, Miyagawa I, Yamamoto Y, Loutradis D, Mantzavinos T, Tarlatzis V, 1998 Micro- and macro-consequences of ooplasmic injections of early haploid male gametes. Hum Reprod Update 4: 197–212.
Rajfer J, Turner TT, Rivera F, Howards SS, Sikka SC, 1987 Inhibition of testicular testosterone biosynthesis following experimental varicocele in rats. Biol Reprod 36: 933–937.
Sofikitis N, Pappas E, Kawatani A, et al, 2005 Efforts to create an artificial testis: culture systems of male germ cells under biochemical conditions resembling the seminiferous tubular biochemical environment. Hum Reprod Update 11: 229–259.
Saito K, O’Donnell L, McLachlan RI, Robertson DM, 2000 Spermiation failure is a major contributor to early spermatogenic suppression caused by hormone withdrawal in adult rats. Endocrinology 141: 2779–2785.
Beardsley A, O’Donnell L, 2003 Characterization of normal spermiation and spermiation failure induced by hormone suppression in adult rats. Biol Reprod 68: 1299–1307.
Wine RN, Chapin RE, 1999 Adhesion and signaling proteins spatiotemporally associated with spermiation in the rat. J Androl 20: 198–213.
McLachlan RI, Wreford NG, de Kretser DM, Robertson DM, 1995 The effects of recombinant follicle-stimulating hormone on the restoration of spermatogenesis in the gonadotropin-releasing hormone-immunized adult rat. Endocrinology 136: 4035–4043.
Singh J, Handelsman DJ, 1996 The effects of recombinant FSH on testosterone-induced spermatogenesis in gonadotrophin-deficient (hpg) mice. J Androl 17: 382–393.
Baccetti B, Strehler E, Capitani S, et al, 1997 The effect of follicle stimulating hormone therapy on human sperm structure (Notulae seminologicae 11). Hum Reprod 12: 1955–1968.
Krishnamurthy H, Danilovich N, Morales CR, Sairam MR, 2000 Qualitative and quantitative decline in spermatogenesis of the follicle-stimulating hormone receptor knockout (FORKO) mouse. Biol Reprod 62: 1146–1159.
Dinulovic D, Radonjic G, 1990 Diabetes mellitus/male infertility. Arch Androl 25: 277–293.
Lok S, Johnston DS, Conklin D, et al, 2000 Identification of INSL6, a new member of the insulin family that is expressed in the testis of the human and rat. Biol Reprod 62: 1593–1599.
Jabbour HN, Lincoln GA, 1999 Prolactin receptor expression in the testis of the ram: localisation, functional activation and the influence of gonadotrophins. Mol Cell Endocrinol 148: 151–161.
Hondo E, Kurohmaru M, Sakai S, Ogawa K, Hayashi Y, 1995 Prolactin receptor expression in rat spermatogenic cells. Biol Reprod 52: 1284–1290.
de Winter JP, Themmen AP, Hoogerbrugge JW, Klaij IA, Grootegoed JA, de Jong FH, 1992 Activin receptor mRNA expression in rat testicular cell types. Mol Cell Endocrinol 83: R1–8.
Andersson AM, Muller J, Skakkebaek NE, 1998 Different roles of prepubertal and postpubertal germ cells and Sertoli cells in the regulation of serum inhibin B levels. J Clin Endocrinol Metab 83: 4451–4458.
Marchetti C, Hamdane M, Mitchell V, et al, 2003 Immunolocalization of inhibin and activin alpha and betaB subunits and expression of corresponding messenger RNAs in the human adult testis. Biol Reprod 68: 230–235.
Meinhardt A, O’Bryan MK, McFarlane JR, et al, 1998 Localization of follistatin in the rat testis. J Reprod Fertil 112: 233–241.
Tycko B, 1997 Post-graduate course program of The American Society of Andrology. In 22nd Annual Meeting of American Society of Andrology. Baltimore, Maryland.
Kimura Y, Yanagimachi R, 1995 Mouse oocytes injected with testicular spermatozoa or round spermatids can develop into normal offspring. Development 121: 2397–2405.
Ogura A, Matsuda J, Yanagimachi R, 1994 Birth of normal young after electrofusion of mouse oocytes with round spermatids. Proc Natl Acad Sci U S A 91: 7460–7462.
Sofikitis NV, Miyagawa I, Agapitos E, et al, 1994 Reproductive capacity of the nucleus of the male gamete after completion of meiosis. J Assist Reprod Genet 11: 335–341.
Fishel S, Aslam I, Tesarik J, 1996 Spermatid conception: a stage too early, or a time too soon? Hum Reprod 11: 1371–1375.
Kimura Y, Yanagimachi R, 1995 Development of normal mice from oocytes injected with secondary spermatocyte nuclei. Biol Reprod 53: 855–862.
Georgiou I, Syrrou M, Pardalidis N, et al, 2006 Genetic and epigenetic risks of intracytoplasmic sperm injection method. Asian J Androl 8: 643–673.
Swinnen JV, Joseph DR, Conti M, 1989 Molecular cloning of rat homologues of the Drosophila melanogaster dunce cAMP phosphodiesterase: evidence for a family of genes. Proc Natl Acad Sci U S A 86: 5325–5329.
Farooqui SM, Al-Bagdadi F, Houslay MD, et al, 2001 Surgically induced cryptorchidism-related degenerative changes in spermatogonia are associated with loss of cyclic adenosine monophosphate-dependent phosphodiesterases type 4 in abdominal testes of rats. Biol Reprod 64: 1583–1589.
Levallet G, Levallet J, Bouraima-Lelong H, Bonnamy PJ, 2007 Expression of the cAMP-phosphodiesterase PDE4D isoforms and age-related changes in follicle-stimulating hormone-stimulated PDE4 activities in immature rat Sertoli cells. Biol Reprod 76: 794–803.
D’Andrea MR, Qiu Y, Haynes-Johnson D, Bhattacharjee S, Kraft P, Lundeen S, 2005 Expression of PDE11A in normal and malignant human tissues. J Histochem Cytochem 53: 895–903.
Scipioni A, Stefanini S, Santone R, Giorgi M, 2005 Immunohistochemical localisation of PDE5 in Leydig and myoid cells of prepuberal and adult rat testis. Histochem Cell Biol 124: 401–407.
Vasta V, Shimizu-Albergine M, Beavo JA, 2006 Modulation of Leydig cell function by cyclic nucleotide phosphodiesterase 8A. Proc Natl Acad Sci U S A 103: 19925–19930.
Baxendale R, Burslem F, Phillips S, 2001 Phosphodiesterase type 11 (PDE11) cellular localization: progress towards defining a physiological role in testis and/or reproduction. J Urol 165: Suppl 5: 340.
Degerman E, Belfrage P, Manganiello VC, 1997 Structure, localization, and regulation of cGMP-inhibited phosphodiesterase (PDE3). J Biol Chem 272: 6823–6826.
Salanova M, Chun SY, Iona S, Puri C, Stefanini M, Conti M, 1999 Type 4 cyclic adenosine monophosphate-specific phosphodiesterases are expressed in discrete subcellular compartments during rat spermiogenesis. Endocrinology 140: 2297–2306.
Yan C, Zhao AZ, Sonnenburg WK, Beavo JA, 2001 Stage and cell-specific expression of calmodulin-dependent phosphodiesterases in mouse testis. Biol Reprod 64: 1746–1754.
Fisch JD, Behr B, Conti M, 1998 Enhancement of motility and acrosome reaction in human spermatozoa: differential activation by type-specific phosphodiesterase inhibitors. Hum Reprod 13: 1248–1254.
Aversa A, Mazzilli F, Rossi T, Delfino M, Isidori AM, Fabbri A, 2000 Effects of sildenafil (Viagra) administration on seminal parameters and post-ejaculatory refractory time in normal males. Hum Reprod 15: 131–134.
Lefievre L, de Lamirande E, Gagnon C, 2002 Presence of cyclic nucleotide phosphodiesterases PDE1A, existing as a stable complex with calmodulin, and PDE3A in human spermatozoa. Biol Reprod 67: 423–430.
Baxendale RW, Fraser LR, 2005 Mammalian sperm phosphodiesterases and their involvement in receptor-mediated cell signaling important for capacitation. Mol Reprod Dev 71: 495–508.
Clouthier DE, Avarbock MR, Maika SD, Hammer RE, Brinster RL, 1996 Rat spermatogenesis in mouse testis. Nature 381: 418–421.
Franca LR, Ogawa T, Avarbock MR, Brinster RL, Russell LD, 1998 Germ cell genotype controls cell cycle during spermatogenesis in the rat. Biol Reprod 59: 1371–1377.
Ogawa T, Dobrinski I, Avarbock MR, Brinster RL, 1999 Xenogeneic spermatogenesis following transplantation of hamster germ cells to mouse testes. Biol Reprod 60: 515–521.
Sofikitis N, Kaponis A, Giannakopoulos X, Miyagawa I 2004 How far are icsi procedures using human spermatozoa generated into animal testicles? micro-and macro-consequences of germ cell transplantation techniques. In: MG. Colpi (ed) Male infertility today. Fotolito e Stampa Grafiche Gelmini, Milano, Italy; pp, 5–39.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Dimitriadis, F., Tsiampali, C., Chaliasos, N. et al. The Sertoli cell as the orchestra conductor of spermatogenesis: spermatogenic cells dance to the tune of testosterone. Hormones 14, 479–503 (2015). https://doi.org/10.14310/horm.2002.1633
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.14310/horm.2002.1633