Abstract
OBJECTIVE
The combination of 32 mg naltrexone and 360 mg bupropion prolonged release (NB32) was recently approved by both the food and drug administration (FDA) and the European medicines agency (EMA) as an adjunct to a comprehensive lifestyle intervention to achieve weight loss.
DESIGN
Randomized controlled trials with naltrexone/bupropion prolonged release were selected through a search based on PubMed listings using the terms “bupropion AND naltrexone”.
RESULTS
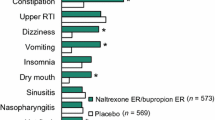
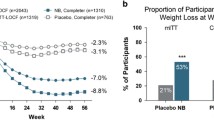
NB32 treatment resulted in 5.0–9.3% weight loss, while the placebo-subtracted weight loss was 3.2–5.2% during 56 weeks of treatment. The proportion of treated patients with ≥5% weight loss was 45–66%, while the placebo-subtracted proportion was 23–34%. NB32 was associated with a decrease in waist circumference, serum triglycerides and insulin resistance and an increase in high-density lipoprotein-cholesterol (HDL-C). Blood pressure mainly remained stable but there was a small increase in heart rate. The most common side effects were nausea, constipation, headache and vomiting. Serious adverse effects, which were very rare, included suicidal thoughts and seizures. In the majority of patients NB32 treatment was well tolerated.
CONCLUSIONS
Naltrexone/bupropion combination appears to be an effective adjunct to a comprehensive lifestyle intervention in order to achieve weight loss and treat obesity-related comorbidities.
Article PDF
Similar content being viewed by others
References
Jensen MD, Ryan DH, Apovian CM, et al, 2014 2013 AHA/ACC/TOS guideline for the management of overweight and obesity in adults: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines and The Obesity Society. Circulation 129: 25 Suppl 2: 102–138.
Kiortsis DN, Filippatos TD, Elisaf MS, 2005 The effects of orlistat on metabolic parameters and other cardiovascular risk factors. Diabetes Metab 31: 15–22.
Christopoulou FD, Kiortsis DN, 2011 An overview of the metabolic effects of rimonabant in randomized controlled trials: potential for other cannabinoid 1 receptor blockers in obesity. J Clin Pharm Ther 36: 10–18.
Filippatos TD, Kiortsis DN, Liberopoulos EN, Mikhailidis DP, Elisaf MS, 2005 A review of the metabolic effects of sibutramine. Curr Med Res Opin 21: 457–468.
Chan EW, He Y, Chui CS, Wong AY, Lau WC, Wong IC, 2013 Efficacy and safety of lorcaserin in obese adults: a meta-analysis of 1-year randomized controlled trials (RCTs) and narrative review on short-term RCTs. Obes Rev 14: 383–392.
Kiortsis DN, 2013 A review of the metabolic effects of controlled-release Phentermine/Topiramate. Hormones (Athens) 12: 507–516.
Iepsen EW, Torekov SS, Holst JJ, 2014 Therapies for inter-relating diabetes and obesity — GLP-1 and obesity. Expert Opin Pharmacother 15: 2487–2500.
Greenway FL, Fujioka K, Plodkowski RA, et al, 2010 Effect of naltrexone plus bupropion on weight loss in overweight and obese adults (COR-I): a multicentre, randomised, double-blind, placebo-controlled, phase 3 trial. Lancet 376: 595–605.
Pucci A, Finer N, 2015 New Medications for Treatment of Obesity: Metabolic and Cardiovascular Effects. Can J Cardiol 31: 142–152.
Gadde KM, Xiong GL, 2007 Bupropion for weight reduction. Expert Rev Neurother 7: 17–24.
Greenway FL, Whitehouse MJ, Guttadauria M, et al, 2009 Rational design of a combination medication for the treatment of obesity. Obesity (Silver Spring) 17: 30–39.
Greenway FL, Dunayevich E, Tollefson G, et al, 2009 Comparison of combined bupropion and naltrexone therapy for obesity with monotherapy and placebo. J Clin Endocrinol Metab 94: 4898–4906.
Smith SR, Fujioka K, Gupta AK, et al, 2013 Combination therapy with naltrexone and bupropion for obesity reduces total and visceral adiposity. Diabetes Obes Metab 15: 863–866.
Wadden TA, Foreyt JP, Foster GD, et al, 2011 Weight loss with naltrexone SR/bupropion SR combination therapy as an adjunct to behavior modification: the COR-BMOD trial. Obesity (Silver Spring) 19: 110–120.
Apovian CM, Aronne L, Rubino D, et al, 2013 A randomized, phase 3 trial of naltrexone SR/bupropion SR on weight and obesity-related risk factors (COR-II). Obesity (Silver Spring) 21: 935–943.
Hollander P, Gupta AK, Plodkowski R, et al, 2013 Effects of naltrexone sustained-release/bupropion sustained-release combination therapy on body weight and glycemic parameters in overweight and obese patients with type 2 diabetes. Diabetes Care 36: 4022–4029.
Wilcox CS, Oskooilar N, Erickson JS, et al, 2010 An open-label study of naltrexone and bupropion combination therapy for smoking cessation in overweight and obese subjects. Addict Behav 35: 229–234.
Després JP, 2012 Body fat distribution and risk of cardiovascular disease: an update. Circulation 126: 1301–1313.
Ross R, Janiszewski PM, 2008 Is weight loss the optimal target for obesity-related cardiovascular disease risk reduction? Can J Cardiol 24: Suppl D: 25D–31D.
Thase ME, Haight BR, Johnson MC, et al, 2008 A randomized, double-blind, placebo-controlled study of the effect of sustained-release bupropion on blood pressure in individuals with mild untreated hypertension. J Clin Psychopharmacol 28: 302–307.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Christou, G.A., Kiortsis, D.N. The efficacy and safety of the naltrexone/bupropion combination for the treatment of obesity: an update. Hormones 14, 370–375 (2015). https://doi.org/10.14310/horm.2002.1600
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.14310/horm.2002.1600