Abstract
Obesity and type 2 diabetes are metabolic diseases that have reached epidemic proportions worldwide. Although their etiology is complex, both result from interplay between behaviour, environment and genetic factors. Within ambient determinants, human overall gut bacteria have been identified as a crucial mediator of obesity and its consequences. Gut microbiota plays a crucial role in gastro-intestinal mucosa permeability and regulates the fermentation and absorption of dietary polyssacharides, which may explain its importance in the regulation of fat accumulation and the resultant development of obesity-related diseases. The main objective of this review is to address the pathogenic association between gut microbiota and obesity and to explore related innovative therapeutic targets. New insights into the role of the small bowel and gut microbiota in diabetes and obesity may make possible the development of integrated strategies to prevent and treat these metabolic disorders.
Article PDF
Similar content being viewed by others
References
WHO | Obesity and overweight. [cited 2014 Feb 8]; Available from: http://www.who.int/mediacentre/factsheets/fs311/en/
WHO | Diabetes. [cited 2014 Feb 8]; Available from: http://www.who.int/mediacentre/factsheets/fs312/en/
Cani PD, Delzenne NM, 2009 The role of the gut microbiota in energy metabolism and metabolic disease. Curr Pharm Des 15: 1546–1558.
Qin J, Li R, Raes J, et al, 2010 A human gut microbial gene catalogue established by metagenomic sequencing. Nature 464: 59–65.
Prakash S, Rodes L, Coussa-Charley M, Tomaro-Duchesneau C, 2011 Gut microbiota: next frontier in understanding human health and development of biotherapeutics. Biologics 5: 71–86.
Bäckhed F, Ley RE, Sonnenburg JL, Peterson DA, Gordon JI, 2005 Host-bacterial mutualism in the human intestine. Science 307: 1915–1920.
Prakash S, Rodes L, Coussa-Charley M, Tomaro-Duchesneau C, 2011 Gut microbiota: next frontier in understanding human health and development of biotherapeutics. Biologics 5: 71–86.
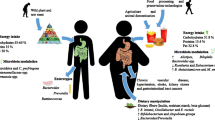
Ley RE, Bäckhed F, Turnbaugh P, Lozupone CA, Knight RD, Gordon JI, 2005 Obesity alters gut microbial ecology. Proc Natl Acad Sci U S A 102: 11070–11075.
Turnbaugh PJ, Ley RE, Mahowald MA, Magrini V, Mardis ER, Gordon JI, 2006 An obesity-associated gut microbiome with increased capacity for energy harvest. Nature 444: 1027–1031.
Ley RE, Turnbaugh PJ, Klein S, Gordon JI, 2006 Microbial ecology: human gut microbes associated with obesity. Nature 444: 1022–1023.
Furet J, Kong L, Tap J, et al, 2010 Differential Adaptation of Human Gut Microbiota to bariatric surgery-induced weight loss: links with metabolic and low-grade inflammation markers. Diabetes 59: 3049–3057.
Kong LC, Wuillemin P-H, Bastard J-P, et al, 2013 Insulin resistance and inflammation predict kinetic body weight changes in response to dietary weight loss and maintenance in overweight and obese subjects by using a Bayesian network approach. Am J Clin Nutr 98: 1385–1394.
Le Chatelier E, Nielsen T, Qin J, et al, 2013 Richness of human gut microbiome correlates with metabolic markers. Nature 500: 541–546.
Geurts L, Lazarevic V, Derrien M, et al, 2011 Altered gut microbiota and endocannabinoid system tone in obese and diabetic leptin-resistant mice: impact on apelin regulation in adipose tissue. Front Microbiol 2: 1–17.
Larsen N, Vogensen FK, van den Berg FWJ, et al, 2010 Gut microbiota in human adults with type 2 diabetes differs from non-diabetic adults. PLoS One 5: e9085.
Qin J, Li Y, Cai Z, et al, 2012 A metagenome-wide association study of gut microbiota in type 2 diabetes. Nature 490: 55–60.
Karlsson FH, Tremaroli V, Nookaew I, et al, 2013 Gut metagenome in European women with normal, impaired and diabetic glucose control. Nature 498: 99–103.
Amar J, Serino M, Lange C, et al, 2011 Involvement of tissue bacteria in the onset of diabetes in humans: evidence for a concept. Diabetologia 54: 3055–3061.
Cani PD, Amar J, Iglesias MA, et al, 2014 Metabolic endo toxemia initiates obesity and insulin resistance. Diabetes 56: 1761–1772.
Cani PD, Neyrinck AM, Fava F, et al, 2007 Selective increases of bifidobacteria in gut microflora improve high-fat-diet-induced diabetes in mice through a mechanism associated with endotoxaemia. Diabetologia 50: 2374–2383.
Laugerette F, Vors C, Géloën A, et al, 2011 Emulsified lipids increase endotoxemia: possible role in early postprandial low-grade inflammation. J Nutr Biochem 22: 53–59.
Poggi M, Bastelica D, Gual P, et al, 2007 C3H/HeJ mice carrying a toll-like receptor 4 mutation are protected against the development of insulin resistance in white adipose tissue in response to a high-fat diet. Diabetologia 50: 1267–1276.
Fasano A, 2011 Zonulin and its regulation of intestinal barrier function: the biological door to inflammation, autoimmunity, and cancer. Physiol Rev 91: 151–175.
Sonier B, Patrick C, Ajjikuttira P, Scott FW, 2009 Intestinal immune regulation as a potential diet-modifiable feature of gut inflammation and autoimmunity. Int Rev Immunol 28: 414–445.
Groschwitz KR, Hogan SP, 2009 Intestinal barrier function: molecular regulation and disease pathogenesis. J Allergy Clin Immunol 124: 3–22.
Lamprecht M, Bogner S, Schippinger G, et al, 2012 Probiotic supplementation affects markers of intestinal barrier, oxidation, and inflammation in trained men; a randomized, double-blinded, placebo-controlled trial. J Int Soc Sports Nutr 9: 45.
Xiao S, Fei N, Pang X, et al, 2014 A gut microbiota-targeted dietary intervention for amelioration of chronic inflammation underlying metabolic syndrome. FEMS Microbiol Ecol 87: 357–367.
Klaus DA, Motal MC, Burger-Klepp U, et al, 2013 Increased plasma zonulin in patients with sepsis. Biochem medica 23: 107–111.
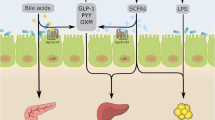
Cani PD, Bibiloni R, Knauf C, et al, 2008 Changes in gut microbiota control metabolic endotoxemia-induced inflammation in high-fat diet-induced obesity and diabetes in mice. Diabetes 57: 1470–1481.
Cani PD, Possemiers S, Van De Wiele T, et al, 2009 Changes in gut microbiota control inflammation in obese mice through a mechanism involving GLP-2-driven improvement of gut permeability. Gut 58: 1091–1103.
Estall JL, Drucker DJ, 2006 Glucagon-like Peptide-2. Annu Rev Nutr 26: 391–411.
Weisberg SP, McCann D, Desai M, Rosenbaum M, Leibel RL, Ferrante AW, 2003 Obesity is associated with macrophage accumulation in adipose tissue. J Clin Invest 112: 1796–1808.
Blüher M, Engeli S, Klöting N, et al, 2006 Dysregulation of the peripheral and adipose tissue endocannabinoid system in human abdominal obesity. Diabetes 55: 3053–3060.
Côté M, Matias I, Lemieux I, et al, 2007 Circulating endocannabinoid levels, abdominal adiposity and related cardiometabolic risk factors in obese men. Int J Obes 31: 692–699.
Silvestri C, Di Marzo V, 2013 The endocannabinoid system in energy homeostasis and the etiopathology of metabolic disorders. Cell Metab 17: 475–490.
Muccioli GG, Naslain D, Bäckhed F, et al, 2010 The endocannabinoid system links gut microbiota to adipogenesis. Mol Syst Biol 6: 392.
Alhamoruni A, Wright KL, Larvin M, O’Sullivan SE, 2012 Cannabinoids mediate opposing effects on inflammation-induced intestinal permeability. Br J Pharmacol 165: 2598–2610.
Lallès J-P, 2010 Intestinal alkaline phosphatase: multiple biological roles in maintenance of intestinal homeostasis and modulation by diet. Nutr Rev 68: 323–332.
Koyama I, Matsunaga T, Harada T, Hokari S, Komoda T, 2002 Alkaline phosphatases reduce toxicity of lipopolysaccharides in vivo and in vitro through dephosphorylation. Clin Biochem 35: 455–461.
De La Serre CB, Ellis CL, Lee J, Hartman AL, Rutledge JC, Raybould HE, 2010 Propensity to high-fat diet-induced obesity in rats is associated with changes in the gut microbiota and gut inflammation. Am J Physiol 299: G440–448.
Bates JM, Akerlund J, Mittge E, Guillemin K, 2004 Intestinal alkaline phosphatase detoxifies lipopolysaccharide and prevents inflammation in zebrafish in response to the gut microbiota. Cell Host Microbe 2: 371–382.
Ghoshal S, Witta J, Zhong J, de Villiers W, Eckhardt E, 2009 Chylomicrons promote intestinal absorption of lipopolysaccharides. J Lipid Res 50: 90–97.
Ding S, Chi MM, Scull BP, et al, 2010 High-fat diet: bacteria interactions promote intestinal inflammation which precedes and correlates with obesity and insulin resistance in mouse. PLoS One 5: e12191.
Ding H, Wang T, Hooper L V, et al, 2004 The gut microbiota as an environmental factor that regulates fat storage. Proc Natl Acad Sci U S A 101: 15718–15712.
Louis P, Scott KP, Duncan SH, Flint HJ, 2007 Understanding the effects of diet on bacterial metabolism in the large intestine. J Appl Microbiol 102:1197–1208.
Louis P, Flint HJ, 2009 Diversity, metabolism and microbial ecology of butyrate-producing bacteria from the human large intestine. FEMS Microbiol Lett 294: 1–8.
Barcenilla A, Pryde SE, Martin JC, et al, 2000 Phylogenese relationships of butyrate-producing bacteria from the human gut. Appl Environ Microbiol 66: 1654–1661.
Le Poul E, Loison C, Struyf S, et al, 2003 Functional characterization of human receptors for short chain fatty acids and their role in polymorphonuclear cell activation. J Biol Chem 278: 25481–25489.
Samuel BS, Shaito A, Motoike T, et al, 2008 Effects of the gut microbiota on host adiposity are modulated by the short-chain fatty-acid binding G protein-coupled receptor, Gpr41. Proc Natl Acad Sci U S A 105: 16767–16772.
Bjursell M, Admyre T, Göransson M, et al, 2011 Improved glucose control and reduced body fat mass in free fatty acid receptor 2-deficient mice fed a high-fat diet. Am J Physiol Endocrinol Metab 300: E211–220.
Stappenbeck TS, Hooper LV, Gordon JI, 2002 Developmental regulation of intestinal angiogenesis by indigenous microbes via Paneth cells. Proc Natl Acad Sci U S A 99: 15451–15455.
Bäckhed F, Manchester JK, Semenkovich CF, Gordon JI, 2007 Mechanisms underlying the resistance to diet-induced obesity in germ-free mice. Proc Natl Acad Sci U S A 104: 979–984.
Turnbaugh PJ, Bäckhed F, Fulton L, Gordon JI, 2008 Diet-Induced Obesity Is Linked to Marked but Reversible Alterations in the Mouse Distal Gut Microbiome. Cell Host Microbe 3: 213–223.
Vrieze A, Van Nood E, Holleman F, et al, 2012 Transfer of intestinal microbiota from lean donors increases insulin sensitivity in individuals with metabolic syndrome. Gastroenterology 143: 913–916.
Mingrone G, Panunzi S, De Gaetano A, et al, 2012 Bariatric surgery versus conventional medical therapy for type 2 diabetes. N Engl J Med 366: 1577–1585.
Schauer BR, Kashyap SR, Wolski K, et al, 2012 Bariatric Surgery versus Intensive Medical Therapy in Obese Patients with Diabetes. N Engl J Med 366: 1567–1576.
Zhang H, DiBaise JK, Zuccolo A, et al, 2009 Human gut microbiota in obesity and after gastric bypass. Proc Natl Acad Sci U S A 106: 2365–2370.
Kong L-C, Tap J, Aron-Wisnewsky J, et al, 2013 Gut microbiota after gastric bypass in human obesity: increased richness and associations of bacterial genera with adipose tissue genes. Am J Clin Nutr 98: 16–24.
Liou AP, Paziuk M, Luevano J-M, Machineni S, Turnbaugh PJ, Kaplan LM, 2013 Conserved shifts in the gut microbiota due to gastric bypass reduce host weight and adiposity. Sci Transi Med 5: 178ra41.
Gibson GR, Roberfroid MB, 1995 Dietary modulation of the human colonic microbiota: introducing the concept of prebiotics. J Nutr 125: 1401–1412.
Roberfroid M, Gibson GR, Hoyles L, et al, 2010 Pre-biotic effects: metabolic and health benefits. Br J Nutr 104: Suppl 2: 1–63.
Soldavini J, Kaunitz JD, 2013 Pathobiology and potential therapeutic value of intestinal short-chain fatty acids in gut inflammation and obesity. Dig Dis Sci 58: 2756–2766.
Parnell JA, Reimer RA, 2009 Weight loss during oligofructose supplementation is associated with decreased ghrelin and increased peptide YY in overweight and obese adults. Am J Clin Nutr 89: 1751–1759.
Cani PD, Joly E, Horsmans Y, Delzenne NM, 2006 Oligofructose promotes satiety in healthy human: a pilot study. Eur J Clin Nutr 60: 567–572.
Delzenne NM, Cani PD, Neyrinck AM, 2007 Modulation of glucagon-like peptide 1 and energy metabolism by inulin and oligofructose: Experimental Data. J Nutr 137: Suppl 11: 2547–2551.
Cani PD, 2006 Improvement of glucose tolerance and hepatic insulin sensitivity by oligofructose requires a functional glucagon-like peptide 1 receptor. Diabetes 55: 1484–1490.
Cani PD, Dewever C, Delzenne NM, 2004 Inulin-type fructans modulate gastrointestinal peptides involved in appetite regulation (glucagon-like peptide-1 and ghrelin) in rats. Br J Nutr 92: 521–526.
Cani PD, Possemiers S, Van de Wiele T, et al, 2009 Changes in gut microbiota control inflammation in obese mice through a mechanism involving GLP-2-driven improvement of gut permeability. Gut 58: 1091–1103.
Capaldo CT, Nusrat A, 2009 Cytokine regulation of tight junctions. Biochim Biophys Acta 1788: 864–871.
Parnell JA, Reimer RA, 2009 Weight loss during oligofructose supplementation is associated with decreased ghrelin and increased peptide YY in overweight and obese adults. Am J Clin Nutr 89: 1751–1759.
Sasaki M, Ogasawara N, Funaki Y, et al, 2013 Transglucosidase improves the gut microbiota profile of type 2 diabetes mellitus patients: a randomized double-blind, placebo-controlled study. BMC Gastroenterol 13: 81.
Weickert MO, Arafat AM, Blaut M, et al, 2011 Changes in dominant groups of the gut microbiota do not explain cereal-fiber induced improvement of whole-body insulin sensitivity. Nutr Metab 8: 90.
Lappi J, Salojärvi J, Kolehmainen M, et al, 2013 Intake of whole-grain and fiber-rich rye bread versus refined wheat bread does not differentiate intestinal microbiota composition in Finnish adults with metabolic syndrome. J Nutr 143: 648–655.
Isken F, Klaus S, Osterhoff M, Pfeiffer AFH, Weickert MO, 2010 Effects of long-term soluble vs. insoluble dietary fiber intake on high-fat diet-induced obesity in C57BL/6J mice. J Nutr Biochem 21: 278–284.
Johnson BR, Klaenhammer TR, 2014 Impact of genomics on the field of probiotic research: historical perspectives to modern paradigms. Antonie Van Leeuwenhoek 106: 141–156.
Chen J, Wang R, Li X-F, Wang R-L, 2012 Bifidobacterium adolescentis supplementation ameliorates visceral fat accumulation and insulin sensitivity in an experimental model of the metabolic syndrome. Br J Nutr 107: 1429–1434.
Chen JJ, Wang R, Li X, Wang R, 2011 Bifidobacterium longum supplementation improved high-fat-fed-induced metabolic syndrome and promoted intestinal Reg I gene expression. Exp Biol Med (Maywood) 236: 823–831.
Yin Y-N, Yu Q-F, Fu N, Liu X-W, Lu F-G, 2010 Effects of four Bifidobacteria on obesity in high-fat diet induced rats. World J Gastroenterol 16: 3394–3401.
Miyoshi M, Ogawa A, Higurashi S, Kadooka Y, 2014 Anti-obesity effect of Lactobacillus gasseri SBT2055 accompanied by inhibition of pro-inflammatory gene expression in the visceral adipose tissue in diet-induced obese mice. Eur J Nutr 53: 599–606.
Park D-Y, Ahn Y-T, Park S-H, et al, 2013 Supplementation of Lactobacillus curvatus HY7601 and Lactobacillus plantarum KY1032 in diet-induced obese mice is associated with gut microbial changes and reduction in obesity. PLoS One 8: e59470.
Everard A, Beizer C, Geurts L, et al, 2013 Cross-talk between Akkermansia muciniphila and intestinal epithelium controls diet-induced obesity. Proc Natl Acad Sci 110: 9066–9071.
Asemi Z, Zare Z, Shaken H, Sabihi S-S, Esmaillzadeh A, 2013 Effect of multispecies probiotic supplements on metabolic profiles, hs-CRP, and oxidative stress in patients with type 2 diabetes. Ann Nutr Metab 63: 1–9.
Ogawa A, Kadooka Y, Kato K, Shirouchi B, Sato M, 2014 Lactobacillus gasseri SBT2055 reduces postprandial and fasting serum non-esterified fatty acid levels in Japanese hypertriacylglycerolemic subjects. Lipids Health Dis 13: 36.
Kadooka Y, Sato M, Ogawa A, et al, 2013 Effect of Lactobacillus gasseri SBT2055 in fermented milk on abdominal adiposity in adults in a randomised controlled trial. Br JNutr 110: 1696–1703.
Arora T, Anastasovska J, Gibson G, et al, 2012 Effect of Lactobacillus acidophilus NCDC 13 supplementation on the progression of obesity in diet-induced obese mice. Br JNutr 108: 1382–1389.
Membrez M, Blancher F, Jaquet M, et al, 2008 Gut microbiota modulation with norfloxacin and ampicillin enhances glucose tolerance in mice. FASEB J 22: 2416–2426.
Carvalho BM, Guadagnini D, Tsukumo DML, et al, 2012 Modulation of gut microbiota by antibiotics improves insulin signalling in high-fat fed mice. Diabetologia 55: 2823–2834.
Murphy EF, Cotter PD, Hogan A, et al, 2013 Divergent metabolic outcomes arising from targeted manipulation of the gut microbiota in diet-induced obesity. Gut 62: 220–226.
Hansen CHF, Krych L, Nielsen DS, et al, 2012 Early life treatment with vancomycin propagates Akkermansia muciniphila and reduces diabetes incidence in the NOD mouse. Diabetologia 55: 2285–2294.
O’Sullivan O, Coakley M, Lakshminarayanan B, et al, 2013 Alterations in intestinal microbiota of elderly Irish subjects post-antibiotic therapy. J Antimicrob Chemother 68: 214–221.
Jernberg C, Löfmark S, Edlund C, Jansson JK, 2010 Long-term impacts of antibiotic exposure on the human intestinal microbiota. Microbiology 156: 3216–3223.
Jernberg C, Löfmark S, Edlund C, Jansson JK, 2007 Long-term ecological impacts of antibiotic administration on the human intestinal microbiota. ISME J 1: 56–66.
Thuny F, Richet H, Casalta J-P, Angelakis E, Habib G, Raoult D, 2010 Vancomycin treatment of infective endocarditis is linked with recently acquired obesity. PLoS One 5: e9074.
Ajslev TA, Andersen CS, Gamborg M, Sørensen TIA, Jess T, 2011 Childhood overweight after establishment of the gut microbiota: the role of delivery mode, pre-pregnancy weight and early administration of antibiotics. Int J Obes 35: 522–529.
Hernández E, Bargiela R, Diez MS, et al, 2013 Functional consequences of microbial shifts in the human gastrointestinal tract linked to antibiotic treatment and obesity. Gut Microbes 4: 306–315.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lau, E., Carvalho, D., Pina-Vaz, C. et al. Beyond gut microbiota: understanding obesity and type 2 diabetes. Hormones 14, 358–369 (2015). https://doi.org/10.14310/horm.2002.1571
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.14310/horm.2002.1571