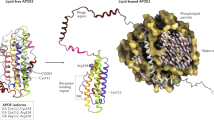
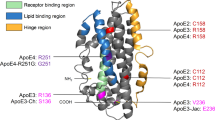
Abstract
Alzheimer’s disease (AD), the most common form of dementia, is a progressive, degenerative disorder of the central nervous system. The major hallmarks of AD include selective neuronal cell death and the presence of amyloid deposits and neurofibrillary tangles. Apolipoprotein E (ApoE) has also been shown to colocalize with these neuropathological lesions. Here is reviewed the role of ApoE in AD. The human ApoE gene has three alleles (ε2, ε3, ε4)-all products of the same gene. The ε3-allele accounts for the majority of the ApoE gene pool (∼70–80%), the ε4-allele accounts for 10–15% and the ε2 allele for 5–10%. Inheritance of the ε4-allele strongly increases the risk for developing AD at an earlier age. Functions of ApoE include cholesterol transport, neuronal repair, dendritic growth and anti-inflammatory activities. Putative pathological functions or “risk-factor activities” of ApoE-ε4 include its role in promoting amyloid accumulation, neurotoxicity, oxidative stress and neurofibrillary tangles.
ApoE mRNA is most abundant in the liver followed by the brain, where it is synthesized and secreted primarily by astrocytes. ApoE protein and mRNA are further detected in cortical and hippocampal neurons in humans. ApoE gene expression is induced by brain injury in some neurons and upregulated in astrocytes during aging. In AD, an increased ApoE mRNA was reported in the hippocampus. The risk for AD has been reported to correlate with transcriptional activity of the ApoE gene. Binding sites for putative transcriptional factors (TF), such as AP-1, AP-2 and NF-κB, are present in the ApoE promoter. The promoter also contains sites for the inflammatory response transcription factors IL-6 RE-BP, MED1, STAT1 and STAT2. A functional peroxisome-proliferator-activated receptor gamma (PPARγ) has been detected in the ApoE/ApoCI intergenic region. ApoE mRNA levels were shown to be regulated by ciglitazone, a PPARγ inducer. Certain statin drugs may also affect ApoE promoter activity. Two distal enhancers that specify ApoE gene expression in macrophages were identified. These results have implications for the regulation of ApoE gene expression, which plays an important role in the development of AD. The interaction of different transcription factors with the regulatory region of the ApoE gene is important to understand the neuroinflammatory process seen in AD. Thus, these regulatory sites can potentially be effective drug targets to control the expression of the ApoE gene.
Article PDF
Similar content being viewed by others
References
Artiga M. J., Bullido M. J., Frank A., Sastre I., Recuero M., Garcia M. A., et al. (1998) Risk for Alzheimer’s disease correlates with transcriptional activity of the APOE gene. Hum. Mol. Genet. 7(12), 1887–1892.
Bales K. R., Du Y., Holtzman D., Cordell B., and Paul S. M. (2000) Neuroinflammation and Alzheimer’s disease: critical roles for cytokine/Abeta-induced glial activation, NF-kappaB, and apolipoprotein E. Neurobiol. Aging 21(3), 427–432.
Boschert U., Merlo-Pich E., Higgins G., Roses A. D., and Catsicas S. (1999) Apolipoprotein E expression by neurons surviving excitotoxic stress. Neurobiol. Dis. 6, 508–514.
Corder E. H., Saunders A. M., Strittmatter W. J., Schmechel D. E., Gaskell P. C., Small G. W., et al. (1993) Gene dose of apolipoprotein E type 4 allele and the risk of Alzheimer’s disease in late onset families. Science 261, 921–923.
Dichtl W., Dulak J., Frick M., Alber H. F., Schwarzacher S. P., Ares M. P., et al. (2003) HMG-CoA reductase inhibitors regulate inflammatory transcription factors in human endothelial and vascular smooth muscle cells. Arterioscler. Thromb. Vasc. Biol. 23(1), 58–63.
Elshourbagy N. A., Liao W. S., Mahley R. W., and Taylor J. M. (1985) Apolipoprotein E mRNA is abundant in the brain and adrenals, as well as in the liver, and is present in other peripheral tissues of rats and marmosets. Proc. Natl. Acad. Sci. USA 82, 203–207.
Fillit H. M., O’Connell A. W., and Refolo L. M. (2002) Strategies for drug discovery for cognitive aging and Alzheimer’s disease. J. Mol. Neurosci. 19(1–2), 1–3.
Galetto R., Albajar M., Polanco J., Zakin M. M., and Rodríguez-Rey J. C. (2001) Identification of a peroxisome-proliferator-activated-receptor response element in the apolipoprotein E gene control region. Biochem. J. 357, 521–527.
Garcia M. A., Vazquez J., Gimenez C., Valdivieso F., and Zafra F. (1996) Transcription factor AP-2 regulates human apolipoprotein E gene expression in astrocytoma cells. J. Neurosci. 16(23), 7550–7556.
Grehan S., Tse E., and Taylor J. (2001) Two distal downstream enhancers direct expression of the human apolipoprotein E gene to astrocytes in the brain. J. Neurosci. 21, 812–822.
Horiuchi K., Tajima S., Menju M., and Yamamoto A. (1989) Structure and expression of mouse apolipoprotein E gene. J. Biochem. 106, 98–103.
Laffitte B. A., Repa J. J., Joseph S.B, Wilpitz D. C., Kast H. R., Mangelsdorf D. J., and Tontonoz P. (2001) LXRs control lipid-inducible expression of the apolipoprotein E gene in macrophages and adipocytes. Proc. Natl. Acad. Sci. USA 98, 507–512.
Lahiri D. K., Nall C., and Ge Y.-W. (1999) Promoter activity of the beta-amyloid precursor protein gene is negatively modulated by an upstream regulatory element. Mol. Brain Res. 71, 32–41.
Lahiri D. K., Song W., and Ge Y.-W. (2000) Analysis of the 5′-flanking region of the β-amyloid precursor protein gene that contributes to increased promoter activity in differentiated neuronal cells. Mol. Brain Res. 77, 185–198.
Lahiri D. K., Farlow M. R., Greig N. H., Giacobini E., and Schneider L. S. (2003) A critical analysis of new molecular targets and strategies for drug developments in Alzheimer’s disease. Curr. Drug Targets 4(2), 97–112.
Lahiri D. K., Alley G. M., Ge Y. W., and Du Y. (2002) Functional characterization of the 5′-regulatory region of the murine apolipoprotein gene. Ann. N. Y. Acad. Sci. 973, 340–344.
Paik Y. K., Reardon C. A., Taylor J. M., and Choi BK. (1995) Characterization of an upstream regulatory sequence and its binding protein in the mouse apolipoprotein E gene. Biochim. Biophys. Acta 1262, 124–132.
Palmer C. N., Hsu M. H., Griffin H. J., and Johnson E. F. (1995) Novel sequence determinants in peroxisome proliferator signaling. J. Biol. Chem. 270, 16114–16121.
Petanceska S. S., DeRosa S., Olm V., Diaz N., Sharma A, Thomas-Bryant T., et al. (2002) Statin therapy for Alzheimer’s disease: will it work? J. Mol. Neurosci. 19(1–2), 155–161.
Pitas R. E., Boyles J. K., Lee S. H., Hui D., and Weisgraber K. H. (1987) Lipoproteins and their receptors in the central nervous system. Characterization of the lipoproteins in cerebrospinal fluid and identification of apolipoprotein B, E(LDL) receptors in the brain. J. Biol. Chem. 262, 14352–14360.
Poirier J., Delisle M., Quirion R., Aubert I., Farlow M., Lahiri D. K., et al. (1995) Apolipoprotein E4 allele as a predictor of cholinergic deficits and treatment outcome in Alzheimer’s disease. Proc. Natl. Acad. Sci. USA 92, 122260–12264.
Rajavashisth T. B., Kaptein J. S., Reue K. L., and Lusis A. J. (1985) Evolution of apolipoprotein E: mouse sequence and evidence for an 11-nucleotide ancestral unit. Proc. Natl. Acad. Sci. USA 82, 8085–8089.
Roses A. D. (1996) Apolipoprotein E alleles as risk factors in Alzheimer’s disease. Annu. Rev. Med. 47, 387–400.
Saunders A. M., Trowers M. K., Shimkets R. A., Blakemore S., Crowther D. J., Mansfield T. A., et al. (2000) The role of apolipoprotein E in Alzheimer’s disease: pharmacogenomic target selection. Biochim. Biophys. Acta 1502(1), 85–94.
Shih S. J., Allan C., Grehan S., Tse E., Moran C., and Taylor J. M. (2000) Duplicated downstream enhancers control expression of the human apolipoprotein E gene in macrophages and adipose tissue. J. Biol. Chem. 275(41), 31567–31572.
Song W. and Lahiri D. K. (1998) Molecular cloning of the gene encoding the Rhesus monkey beta-amyloid precursor protein: structural characterization of the promoter and comparative study with other species. Gene 217, 151–164.
Song W. and Lahiri D. K. (1998) Functional identification of the promoter of the gene encoding the Rhesus monkey beta-amyloid precursor protein. Gene 217, 165–176.
Soulie C., Mitchell V., Dupont-Wallois L., Chartier-Harlin M.- C., Beauvillain J.- C., Delacourte A., and Caillet-Boudin M. L. (1999) Synthesis of apolipoprotein E (apoE) mRNA by human neuronal-type SK N SH-SY 5Y cells and its regulation by nerve growth factor and apoE. Neurosci. Lett. 265, 147–150.
Weisgraber K. H. and Mahley R. W. (1996) Human apolipoprotein E: the Alzheimer’s disease connection. FASEB J. 10(13), 1485–1494.
Xu P.- T., Gilbert J. R., Qiu H.- L., Ervin J., Rothrock-Christian T. R., Hulette C., and Schmechel D. E. (1999) Specific regional transcription of apolipoprotein E in human brain neurons. Am. J. Pathol. 154, 601–611.
Yamagata K., Urakami K., Ikeda K., Ji Y., Adachi Y., Arai H., et al. (2001) High expression of apolipoprotein EmRNA in the brains with sporadic Alzheimer’s disease. Dement. Geriatr. Cogn. Disord. 12(2), 57–62.
Zarow C. and Victoroff J. (1998) Increased apolipoprotein EmRNA in the hippocampus in Alzheimer disease and in rats after entorhinal cortex lesioning. Exp. Neurol. 149(1), 79–86.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lahiri, D.K. Apolipoprotein E as a target for developing new therapeutics for Alzheimer’s disease based on studies from protein, RNA, and regulatory region of the gene. J Mol Neurosci 23, 225–233 (2004). https://doi.org/10.1385/JMN:23:3:225
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1385/JMN:23:3:225