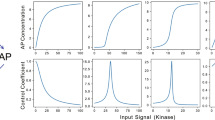
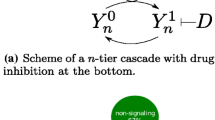
Abstract
Intracellular signal transduction pathways transmit signals from the cell surface to various intracellular destinations, such as cytoskeleton and nucleus through a cascade of protein-protein interactions and activation events, leading to phenotypic changes such as cell proliferation, differentiation, and death. Over the past two decades, numerous signaling proteins and signal transduction pathways have been discovered and characterized. There are two major classes of signaling proteins: phosphoproteins (e.g., mitogen-activated protein kinases) and guanosine triphosphatases (GTPases; e.g., Ras and G proteins). They both function as molecular switches by addition and removal of one or more high-energy phosphate groups. This review discusses developments that seek to quantify the signal transduction processes with kinetic analysis and mathematical modeling of the signaling phosphoproteins and GTPases. These studies have provided insights into the sensitivity and specificity amplification of biological signals in integrated systems.
Similar content being viewed by others
References
Gurdon, J. B., Lemaire, P., and Kato, K. (1993) Community effects and related phenomena in development. Cell 75, 831–834.
Pitcher, J. A., Freedman, N. J., and Lefkowitz, R. J. (1998) G protein-coupled receptor kinases. Annu. Rev. Biochem. 67, 653–692.
Schlessinger, J. (2000) Cell signaling by receptor tyrosine kinases. Cell 103, 211–225.
Bourne, H. R. (1997) How receptors talk to trimeric G proteins. Curr. Opin. Cell. Biol. 9, 134–142.
Cohen, P. (1992) Signal integration at the level of protein kinases, protein phosphatases and their substrates. Trends Biochem. Sci. 17, 408–413.
Pawson, T. and Nash, P. (2000) Protein-protein interactions define specificity in signal transduction. Genes. Dev. 14, 1027–1047.
Widmann, C., Gibson, S., Jarpe, M. B., and Johnson, G. L. (1999) Mitogen-activated protein kinase: conservation of a three-kinase module from yeast to human. Physiol. Rev. 79, 143–180.
Bar-Sagi, D. and Hall, A. (2000) Ras and Rho GTPases: a family reunion. Cell 103, 227–238.
Bourne, H. R., Sanders, D. A., and McCormick, F. (1990) The GTPase superfamily: a conserved switch for diverse cell functions. Nature 348, 125–132.
Gilman, A. G. (1987) G proteins: transducers of receptor-generated signals. Annu Rev Biochem 56, 615–649.
Egan, S. E., Giddings, B. W., Brooks, M. W., Buday, L., Sizeland, A. M., and Weinberg, R. A. (1993) Association of Sos Ras exchange protein with Grb2 is implicated in tyrosine kinase signal transduction and transformation. Nature 363, 45–51.
Boriack-Sjodin, P. A., Margarit, S. M., Bar-Sagi, D., and Kuriyan, J. (1998) The structural basis of the activation of Ras by Sos. Nature 394, 337–343.
Moodie, S. A., Willumsen, B. M., Weber, M. J., and Wolfman, A. (1993) Complexes of Ras-GTP with Raf-1 and mitogen-activated protein kinase. Science 260, 1658–1661.
van Aelst, L., Barr, M., Marcus, S., Polverino, A., and Wigler, M. (1993) Complex formation between RAS and RAF and other protein kinases. Proc. Natl. Acad. Sci. USA 90, 6213–6217.
Vojtek, A. B., Hollenberg, S. M., and Cooper, J. A. (1993) Mammalian ras interacts directly with the serine/threonine kinase raf. Cell 74, 205–214.
Warne, P. H., Viciana, P. R., and Downward, J. (1993) Direct interaction of Ras and the aminoterminal region of Raf-1 in vitro. Nature 364, 352–355.
Khokhlatchev, A. V., Canagarajah, B., Wilsbacher, J., Robinson, M., Atkinson, M., Goldsmith, E., and Cobb, M. H. (1998) Phosphorylation of the MAP kinase ERK2 promotes its homodimerization and nuclear translocation. Cell 93, 605–615.
Karin, M. (1995) The regulation of AP-1 activity by mitogen-activated protein kinases. J. Biol. Chem. 270, 16483–16486.
Waskiewicz, A. J. and Cooper, J. A. (1995) Mitogen and stress response pathways: MAP kinase cascades and phosphatase regulation in mammals and yeast. Curr. Opin. Cell. Biol. 7, 798–805.
Koshland, D. E., Jr. (1998) The era of pathway quantification. Science 280, 852–853.
Swain, P. S. and Siggia, E. D. (2002) The role of proofreading in signal transduction specificity. Biophys. J. 82, 2928–2933.
Ferrell, J. E., Jr., and Machleder, E. M. (1998) The biochemical basis of an all-or-none cell fate switch in Xenopus oocytes. Science 280, 895–898.
Bhalla, U. S., Ram, P. T., and Iyengar, R. (2002) MAP kinase phosphatase as a locus of flexibility in a mitogen-activated protein kinase signaling network. Science 297, 1018–1023.
Goldbeter, A. and Koshland, D. E., Jr. (1981) An amplified sensitivity arising from covalent modification in biological systems. Proc. Natl. Acad. Sci. USA 78, 6840–6844.
Baldwin, J. and Chothia, C. (1979) Haemoglobin: the structural changes related to ligand binding and its allosteric mechanism. J. Mol. Biol. 129, 175–220.
Gelin, B. R., Lee, A. W., and Karplus, M. (1983) Hemoglobin tertiary structural change on ligand binding. Its role in the co-operative mechanism. J. Mol. Biol. 171, 489–559.
Ferrell, J. E., Jr. (2002) Self-perpetuating states in signal transduction: positive feedback, double-negative feedback and bistability. Curr. Opin. Cell. Biol. 14, 140–148.
Bagowski, C. P. and Ferrell, J. E., Jr. (2001) Bistability in the JNK cascade. Curr. Biol. 11, 1176–1182.
Berg, O. G., Paulsson, J., and Ehrenberg, M. (2000) Fluctuations and quality of control in biological cells: zero-order ultrasensitivity reinvestigated. Biophys. J. 79, 1228–1236.
Qian, H. (2003) Thermodynamic and kinetic analysis of sensitivity amplification in biological signal transduction. Biophys. Chem., in press.
Hopfield, J. J. (1980) The energy relay: a proofreading scheme based on dynamic cooperativity and lacking all characteristic symptoms of kinetic proofreading in DNA replication and protein synthesis. Proc. Natl. Acad. Sci. USA 77, 5248–5252.
Wittinghofer, A. (2000) The functioning of molecular switches in three dimensions, in GTPases (Hall, A., ed.). Oxford University Press, NY, pp. 244–310.
Donovan, S., Shannon, K. M., and Bollag, G. (2002) GTPase activating proteins: critical regulators of intracellular signaling. Biochim. Biophys. Acta 1602, 23–45.
Zhang, F. L. and Casey, P. J. (1996) Protein prenylation: molecular mechanisms and functional consequences. Annu. Rev. Biochem. 65, 241–269.
Elion, E. A. (2001) The Ste5p scaffold. J. Cell. Sci. 114, 3967–3978.
Choi, K. Y., Satterberg, B., Lyons, D. M., and Elion, E. A. (1994) Ste5 tethers multiple protein kinases in the MAP kinase cascade required for mating in S. cerevisiae. Cell 78, 499–512.
Morrison, D. K. (2001) KSR: a MAPK scaffold of the Ras pathway? J. Cell. Sci. 114, 1609–1612.
Raabe, T. and Rapp, U. R. (2002) KSR—a regulator and scaffold protein of the MAPK pathway. Sci. STKE 2002, PE28.
Schaeffer, H. J., Catling, A. D., Eblen, S. T., Collier, L. S., Krauss, A., and Weber, M. J. (1998) MP1: a MEK binding partner that enhances enzymatic activation of the MAP kinase cascade. Science 281, 1668–1671.
Kornfeld, K., Hom, D. B., and Horvitz, H. R. (1995) The ksr-1 gene encodes a novel protein kinase involved in Ras-mediated signaling in C. elegans. Cell 83, 903–913.
Sundaram, M. and Han, M. (1995) The C. elegans ksr-1 gene encodes a novel Raf-related kinase involved in Ras-mediated signal transduction. Cell 83, 889–901.
Therrien, M., Chang, H. C., Solomon, N. M., Karim, F. D., Wassarman, D. A., and Rubin, G. M. (1995) KSR, a novel protein kinase required for RAS signal transduction. Cell 83, 879–888.
Jacobs, D., Glossip, D., Xing, H., Muslin, A. J., and Kornfeld, K. (1999) Multiple docking sites on substrate proteins form a modular system that mediates recognition by ERK MAP kinase. Genes Dev. 13, 163–175.
Stewart, S., Sundaram, M., Zhang, Y., Lee, J., Han, M., and Guan, K. L. (1999) Kinase suppressor of Ras forms a multiprotein signaling complex and modulates MEK localization. Mol. Cell. Biol. 19, 5523–5534.
Therrien, M., Michaud, N. R., Rubin, G. M., and Morrison, D. K. (1996) KSR modulates signal propagation within the MAPK cascade. Genes Dev. 10, 2684–2695.
Yu, W., Fantl, W. J., Harrowe, G., and Williams, L. T. (1998). Regulation of the MAP kinase pathway by mammalian Ksr through direct interaction with MEK and ERK. Curr. Biol. 8, 56–64.
Roy, F., Laberge, G., Douziech, M., Ferland-McCollough, D., and Therrien, M. (2002) KSR is a scaffold required for activation of the ERK/MAPK module. Genes Dev. 16, 427–438.
Nguyen, A., Burack, W. R., Stock, J. L., et al. (2002) Kinase suppressor of Ras (KSR) is a scaffold which facilitates mitogen-activated protein kinase activation in vivo. Mol. Cell. Biol. 22, 3035–3045.
Joneson, T., Fulton, J. A., Volle, D. J., Chaika, O. V., Bar-Sagi, D., and Lewis, R. E. (1998) Kinase suppressor of Ras inhibits the activation of extracellular ligand-regulated (ERK) mitogen-activated protein (MAP) kinase by growth factors, activated Ras, and Ras effectors. J. Biol. Chem. 273, 7743–7748.
Whitmarsh, A. J., Kuan, C. Y., Kennedy, N. J. et al. (2001) Requirement of the JIP1 scaffold protein for stress-induced JNK activation. Genes Dev. 15, 2421–2432.
Yasuda, J., Whitmarsh, A. J., Cavanagh, J., Sharma, M., and Davis, R. J. (1999) The JIP group of mitogen-activated protein kinase scaffold proteins. Mol. Cell. Biol. 19, 7245–7254.
Bray, D. and Lay, S. (1997) Computer-based analysis of the binding steps in protein complex formation. Proc. Natl. Acad. Sci. USA. 94, 13493–13498.
Levchenko, A., Bruck, J. and Sternberg, P. W. (2000) Scaffold proteins may biphasically affect the levels of mitogen-activated protein kinase signaling and reduce its threshold properties. Proc. Natl. Acad. Sci. USA. 97, 5818–5823.
Ferrell, J. E., Jr. (2000) What do scaffold proteins really do? Sci. STKE 2000, PE1.
Yablonski, D., Marbach, I. and Levitzki, A. (1996) Dimerization of Ste5, a mitogen-activated protein kinase cascade scaffold protein, is required for signal transduction. Proc. Natl. Acad. Sci. USA. 93, 13864–13869.
Derijard, B., Raingeaud, J., Barrett, T., Wu, I. H., Han, J., Ulevitch, R. J. and Davis, R. J. (1995) Independent human MAP-kinase signal transduction pathways defined by MEK and MKK isoforms. Science 267, 682–685.
Madhani, H. D., Styles, C. A. and Fink, G. R. (1997) MAP kinases with distinct inhibitory functions impart signaling specificity during yeast differentiation. Cell 91, 673–684.
Hopfield, J. J. (1974) Kinetic proofreading: a new mechanism for reducing errors in biosynthetic processes requiring high specificity. Proc. Natl. Acad. Sci. USA. 71, 4135–4139.
Ninio, J. (1975) Kinetic amplification of enzyme discrimination. Biochimie 57, 587–595.
Chong, H., Lee, J. and Guan, K. L. (2001) Positive and negative regulation of Raf kinase activity and function by phosphorylation. Embo J. 20, 3716–3727.
Li, G. and Qian, H. (2002) Kinetic timing: a novel mechanism that improves the accuracy of GTPase timers in endosome fusion and other biological processes. Traffic 3, 249–255.
Neal, S. E., Eccleston, J. F. and Webb, M. R. (1990) Hydrolysis of GTP by p21NRAS, the NRAS protooncogene product, is accompanied by a conformational change in the wildtype protein: use of a single fluorescent probe at the catalytic site. Proc. Natl. Acad. Sci. USA. 87, 3562–3565.
Hu, J. S. and Redfield, A. G. (1997) Conformational and dynamic differences between N-ras P21 bound to GTPgammaS and to GMPPNP as studied by NMR. Biochemistry 36, 5045–5052.
Gideon, P., John, J., Frech, M., Lautwein, A., Clark, R., Scheffler, J. E. and Wittinghofer, A. (1992) Mutational and kinetic analyses of the GTPase-activating protein (GAP)-p21 interaction: the C-terminal domain of GAP is not sufficient for full activity. Mol. Cell. Biol. 12, 2050–2056.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Li, G., Qian, H. Sensitivity and specificity amplification in signal transduction. Cell Biochem Biophys 39, 45–59 (2003). https://doi.org/10.1385/CBB:39:1:45
Issue Date:
DOI: https://doi.org/10.1385/CBB:39:1:45