Abstract
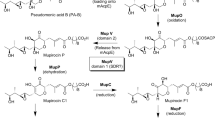
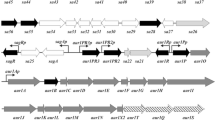
The conversion of succinyl-coenzyme A (CoA) into methylmalonyl-CoA, catalyzed by adenosylcobalamin-dependent methylmalonyl-CoA mutase (MCM), represents an important source of building blocks for rifamycin SV biosynthesis. The structural gene for MCM from rifamycin SV—producing strain Amycolatopsis mediterranei U32 was isolated by using a heterologous gene probe encoding the MCM of Streptomyces cinnamonensis. A 7.8-kbp fragment was sequenced and four complete open reading frames (ORFs) and two incomplete ORFs were found. Two central ORFs, ORF3 and ORF4, overlap by four nucleotides and were found to encode MCM small (602 residues) and large (721 residues) subunits, respectively. Comparison showed that the MCM gene of A. mediterranei U32 was quite similar to those from other sources. The functionally unknown ORF5, immediately downstream of the mut AB gene, was quite similar to the ORFs downstream of mut AB from S. cinnamonensis and Mycobacterium tuberculosis. Such a striking cross-species conservation of gene order suggested that ORF5 could also be involved in the metabolism of methylmalonyl-CoA. MCM gene was overexpressed in Escherichia coli under T7 promoter, and MCM activity could be detected in the recombinant E. coli clone harboring MCM gene after the addition of coenzyme B12. A purification procedure based on the B12 affinity column was established to purify the MCM from E. coli. The molecular weight of purified MCM from E. coli was determined by sodium dodecyl sulfate polyacrylamide gel electrophoresis, which corresponds to that calculated from the MCM protein sequence and is also the same size as that of the enzyme purified directly from A. mediterranei U32. MCM gene was overexpressed in polyketide monensin producing S. cinnamonensis, and the total monensin production was increased by 32%.
Similar content being viewed by others
References
White, R. J., Martinelli, E., Gallo, G. G., and Lancini, G. (1973), Nature (London) 243, 273–277.
Donadio, S., Stever, M. J., McAlpine, J. B., Swanson, S. J., and Katz, L. J. (1991), Science 252, 675–679.
Robinson, J. A. (1992), Philos. Trans. R. Soc. Lond. B. Biol. Sci. 332, 107–114.
Omura, S., Takeshima, H., Nakagawa, A., Miyazawa, J., Piriou, F., and Lukacs, G. (1977), Biochemistry 16, 2860–2866.
March, E. N., McKie, N., Davis, N. K., and Leadlay, P. F. (1990), Biochem. J. 260, 345–352.
Jansen, R., Kalousek, F., Fenton, W. A., Rosenberg, L. E., and Ledley, F. (1989), Genomics 4, 198–205.
Wilkemeyer, M. F., Crane, A. M., and Leadley, F. D. (1990), Biochem. J. 271, 449–455.
Charles, T. C. and Aneja, P. (1999), Gene 226, 121–127.
Jackson, C. A., Kirszbaum, L., Dashper, S., and Reynolds, E. C. (1995), Gene 167, 127–132.
Birch, A., Leiser, A., and Robinson, J. A. (1993), J. Bacteriol. 175, 3511–3519.
Roy, I. and Leadley, P. F. (1992), J. Bacteriol. 174, 5763, 5764.
Smith, L. M., Meijer, W. G., Dijkhuizen, L., and Goodwin, P. M. (1996), Microbiology 142, 675–684.
Han, L. and Reynolds, K. A. (1997), J. Bacteriol. 179, 5157–5164.
Zhang, W. and Chiao, J. S. (1996), Acta Microbiologica Sinica 36, 199–206.
Zhang, W. and Chiao, J. S. (1996), Acta Microbiologica Sinica 36, 276–282.
Zhang, W. and Chiao, J. S. (1996), Chin. J. Biochem. 22, 167–172.
Maniatis, T., Fritsch, E. F., and Sambrook, J. (1989), Molecular Cloning, A Laboratory Manual, Cold Spring Harbor Laboratory, Cold Spring, NY.
Kellermeyer, R. W. and Wood, H. G. (1969), Methods Enzymol. 13, 207–214.
Simon, E. J. and Shemin, D. (1953), J. Am. Chem. Soc. 75, 2520.
Hopwood, D. A., Bibb, M. J., Chater, K. F., Kieser, T., Bruton, C. J., Kieser, H. M., Lydiate, D. J., Smith, C. P., Ward, J. M., and Schrempf, H. (1985), Genetic Manipulation of Streptomyces: A Laboratory Manual, John Inns Foundation, Norwich, England.
Reynolds, K. A., O’Hagan, D., Gani, D., and Robinson, J. A., (1988), Chem. Soc. Perkin. Trans. 1, 3194–3207.
Beran, M. and Zima, J. (1993), Chromatographia 35, 206–208.
Bibb, M. J. and Cohen, S. N. (1982), Mol. Gen. Genetics 187, 265–277.
Strohl, W. R. (1992), Nucleic Acids Res. 20, 961–974.
Drennan, C. L., Matthews, R. G., and Ludwig, M. L. (1994), Curr. Opin. Struct. Biol. 4, 919–929.
Mancia, F., Keep, N. H., Nakagawa, A., Leadley, P. F., McSweeney, S., Rasmussen, B., Bosecke, P., Diat, O., and Even, P. R. (1996), Structure 4, 339–350.
Roy, I. (1996), FEMS Lett. 394, 126–128.
Wang, S. P., Sharma, P. L., Schoenlein, P. V., and Ely, B. (1993), Proc. Natl. Acad. Sci. USA 90, 630–634.
Author information
Authors and Affiliations
Corresponding author
Additional information
Both authors contributed equally to this paper.
Rights and permissions
About this article
Cite this article
Zhang, W., Yang, L., Jiang, W. et al. Molecular analysis and heterologous expression of the gene encoding methylmalonyl—coenzyme a mutase from rifamycin SV-producing strain Amycolatopsis mediterranei U32. Appl Biochem Biotechnol 82, 209–225 (1999). https://doi.org/10.1385/ABAB:82:3:209
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1385/ABAB:82:3:209